BD1008
 | |
| Identifiers | |
|---|---|
| |
| CAS Number | 138356-08-8 |
| PubChem (CID) | 126388 |
| ChemSpider | 112324 |
| Chemical and physical data | |
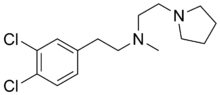
| Formula | C15H22Cl2N2 |
| Molar mass | 301.25 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
BD1008 or N-[2-(3,4-dichlorophenyl)ethyl]-N-methyl-1-pyrrolidineethanamine is a selective sigma receptor antagonist, with a reported binding affinity of Ki = 2 ± 1 nM for the sigma-1 receptor and 4 times selectivity over the sigma-2 receptor.[1]
Consistent with other reported sigma receptor antagonists, pretreating Swiss Webster mice with BD1008 significantly attenuates the behavioral toxicity of cocaine, and may be potentially useful in the development of antidotes for the treatment of cocaine overdose.[1][2][3][4]
See also
References
- 1 2 Matsumoto RR, McCracken KA, Pouw B, Miller J, Bowen WD, Williams W, De Costa BR (2001). "N-alkyl substituted analogs of the sigma receptor ligand BD1008 and traditional sigma receptor ligands affect cocaine-induced convulsions and lethality in mice". Eur. J. Pharmacol. 411 (3): 261–73. doi:10.1016/s0014-2999(00)00917-1.
- ↑ Monnet FP, de Costa BR, Bowen WD (September 1996). "Differentiation of sigma ligand-activated receptor subtypes that modulate NMDA-evoked [3H]-noradrenaline release in rat hippocampal slices". British Journal of Pharmacology. 119 (1): 65–72. doi:10.1111/j.1476-5381.1996.tb15678.x. PMC 1915737
 . PMID 8872358.
. PMID 8872358. - ↑ Matsumoto RR, McCracken KA, Friedman MJ, Pouw B, De Costa BR, Bowen WD (May 2001). "Conformationally restricted analogs of BD1008 and an antisense oligodeoxynucleotide targeting sigma1 receptors produce anti-cocaine effects in mice". European Journal of Pharmacology. 419 (2-3): 163–74. doi:10.1016/S0014-2999(01)00968-2. PMID 11426838.
- ↑ Matsumoto RR, Gilmore DL, Pouw B, Bowen WD, Williams W, Kausar A, Coop A (May 2004). "Novel analogs of the sigma receptor ligand BD1008 attenuate cocaine-induced toxicity in mice". European Journal of Pharmacology. 492 (1): 21–6. doi:10.1016/j.ejphar.2004.03.037. PMID 15145701.
This article is issued from Wikipedia - version of the 6/7/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.