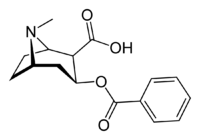
Benzoylecgonine
 | |
 | |
| Names | |
|---|---|
| IUPAC name
3-Benzoyloxy-8-methyl-8-azabicyclo[3.2.1]octane-4-carboxylic acid | |
| Identifiers | |
| 519-09-5 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:41001 |
| ChemSpider | 395095 |
| DrugBank | DB01515 |
| ECHA InfoCard | 100.007.513 |
| KEGG | C10847 |
| UNII | 5353I8I6YS |
| |
| |
| Properties | |
| C16H19NO4 | |
| Molar mass | 289.33 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Benzoylecgonine is the main metabolite of cocaine.
History
Benzoylecgonine is the main pharmaceutical ingredient in the investigational drug Esterom,[1] a topical solution used for the relief of muscle pain that is not FDA approved or on the market in the United States.[2]
Pharmacokinetics
Chemically, benzoylecgonine is ecgonine benzoate. It is a primary metabolite of cocaine.[3]
Urinalysis
Benzoylecgonine is the compound tested for in most substantive cocaine urinalyses. It is the corresponding carboxylic acid of cocaine, its methyl ester. It is formed in the liver by the metabolism of cocaine, catalysed by carboxylesterases, and subsequently excreted in the urine. It can be found in the urine for considerably longer than the cocaine itself which is generally cleared out within 5 days.
Presence in drinking water
Benzoylecgonine is sometimes found in drinking water supplies. In 2005, scientists found surprisingly large quantities of benzoylecgonine in Italy's Po River and used its concentration to estimate the number of cocaine users in the region.[4] In 2006, a similar study was performed in the Swiss ski town of Saint-Moritz using waste water to estimate the daily cocaine consumption of the population.[5] A study done in the United Kingdom found traces of benzoylecgonine in the country's drinking water supply, along with carbamazepine (an anticonvulsant) and ibuprofen (a common non-steroidal anti-inflammatory drug), although the study noted that the amount of each compound present was several orders of magnitude lower than the therapeutic dose and therefore did not pose a risk to the population.[6]
Preliminary studies on ecological systems show that benzoylecgonine has potential toxicity issues.[7] Research is being conducted on degradation options such as advanced oxidation and photocatalysis[8] for this metabolite in an effort to reduce concentrations in waste and surface waters. At environmentally relevant concentrations, benzoylecgonine has been shown to have a negative ecological impact.[7]
See also
References
- ↑ McDonald, Sarah; Lunte, Craig (Nov 2003). "Determination of the Dermal Penetration of Esterom Components Using Microdialysis Sampling". Pharm Res. 20 (11): 1827–1834. PMC 2519827
 .
. - ↑ FDA. "Website of the USFDA". US Department of Health and Human Services. Retrieved 20 January 2016.
- ↑ Schindler, Charles W; Goldberg, Steven R (2012). "Accelerating cocaine metabolism as an approach to the treatment of cocaine abuse and toxicity". Future Medicinal Chemistry. 4 (2): 163. doi:10.4155/fmc.11.181. PMID 22300096.
- ↑ "Italian river 'full of cocaine'". BBC News. 5 August 2005. Retrieved 11 May 2014.
- ↑ "Tant de coke ? Stupéfiant !". Courrier International (in French). 2 February 2006. Retrieved 11 May 2014.
- ↑ Withnall, Adam (11 May 2014). "Cocaine use in Britain so high it has contaminated our drinking water, report shows". The Independent. Retrieved 11 May 2014.
- 1 2 Binelli, A.; Marisa, I; Fedorova, M; Hoffmann, R; Riva, C (2013). "First evidence of protein profile alteration due to the main cocaine metabolite (benzoylecgonine) in a freshwater model". Aquatic Toxicology. 140-141: 268–278. doi:10.1016/j.aquatox.2013.06.013. PMID 23838174.
- ↑ Postigo, C.; Sirtori, C.; Oller, I.; Malato, S.; Maldonado, M.I.; Lopez de Alda, M.; Barcelo, D. (2011). "Solar transformation and photocatalytic treatment of cocaine in water: kinetics, characterization of major intermediate products and toxicity evaluation.". Applied Catalysis B: Environmental. 104: 37–48.