E-64
 | |
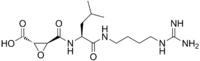 | |
| Names | |
|---|---|
| IUPAC name
(1S,2S)-2-(((S)-1-((4-Guanidinobutyl)amino)-4-methyl-1-oxopentan-2-yl)carbamoyl)cyclopropanecarboxylic acid | |
| Other names
E 64; Proteinase Inhibitor E 64; N-[N-(L-3-trans-carboxyirane-2-carbonyl)-L-leucyl]-agmatine; [1-[N-[(L-3-trans-carboxyoxirane-2-carbonyl)-L-leucyl]amino]-4-guanidinobutane] | |
| Identifiers | |
| 66701-25-5 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:30270 |
| ChEMBL | ChEMBL374508 |
| ChemSpider | 110504 |
| ECHA InfoCard | 100.130.729 |
| KEGG | C01341 |
| PubChem | 123985 |
| UNII | R76F7856MV |
| |
| |
| Properties | |
| C15H27N5O5 | |
| Molar mass | 357.41 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
E-64 is an epoxide which can irreversibly inhibit a wide range of cysteine peptidases.
The compound was first isolated and identified from Aspergillus japonicus in 1978.[1] It has since been shown to inhibit many cysteine peptidases such as papain, cathepsin B, cathepsin L, calpain and staphopain.[2]
The low toxic effects of the inhibitor, in addition to its effective mechanism of action, makes E-64 a potential template for drugs to treat diseases where high levels of a cysteine proteases are the primary cause.
Structure and mechanism of inhibition
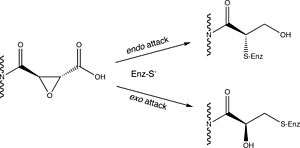
E-64 possesses a trans-epoxysuccinic acid group coupled to a modified dipeptide. The covalent attachment of E-64 to the active site cysteine occurs via nucleophillic attack from the thiol group of the cysteine on C2 of the epoxide. Early studies suggested that the amino-4-guanidinobutane bound in the S3' subsite and the leucyl group in the S2' subsite,[3] however published crystal structures of E-64 complexed with papain and actinidum indicated that E-64 binds via the S subsites.[2]
References
- ↑ Hanada K, Tamai M, Yamagishi M, Ohmura S, Sawada J, Tanaka I (1978). Agric. Biol. Chem. 42: 523–528. Missing or empty
|title=(help) - 1 2 Varughese K, Ahmed F, Carey P, Hasnain S, Huber C, Storer A (1989). "Crystal structure of a papain-E-64 complex". Biochemistry. 28 (3): 1332–2. doi:10.1021/bi00429a058. PMID 2713367.
- ↑ Barrett A, Kembhavi A, Brown M, Kirschke H, Knight C, Tamai M, Hanada K (1982). "L-trans-Epoxysuccinyl-leucylamido(4-guanidino)butane (E-64) and its analogues as inhibitors of cysteine proteinases including cathepsins B, H and L.". J. Biochem. 201 (1): 189–98. doi:10.1042/bj2010189. PMC 1163625
 . PMID 7044372.
. PMID 7044372.