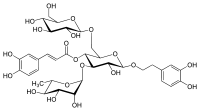
Echinacoside
 | |
| Names | |
|---|---|
| IUPAC name
[(2R,3R,4R,5R,6R)-6-[2-(3,4-dihydroxyphenyl)ethoxy]-5-hydroxy-2-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxymethyl]-4-[(2S,3R,4R,5R,6S)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxyoxan-3-yl](E)-3-(3,4-dihydroxyphenyl)prop-2-enoate | |
| Identifiers | |
| 82854-37-3 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 4445084 |
| ECHA InfoCard | 100.127.421 |
| PubChem | 5281771 |
| |
| |
| Properties | |
| C35H46O20 | |
| Molar mass | 786,73 g/mol |
| Melting point | 200 to 220 °C (392 to 428 °F; 473 to 493 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Echinacoside is a natural phenol. It is a caffeic acid glycoside from the phenylpropanoid class. It is constituted from a trisaccharide consisting of two glucose and one rhamnose moieties glycosidically linked to one caffeic acid and one dihydroxyphenylethanol (hydroxytyrosol) residue at the centrally situated rhamnose. This water-soluble glycoside is a distinctive secondary metabolite of Echinacea angustifolia and Echinacea pallida (to about 1%) but only occurs in trace amounts in Echinacea purpurea. It is also isolated from Cistanche spp.
It was first isolated by Stoll et al. in 1950 from the roots of Echinacea angustifolia. It shows weak antibiotic activity in vitro against Staphylococcus aureus and Streptococci.[1]
References
- ↑ Stoll, A.; Renz, J.; Brack, A. (1950). "Isolierung und Konstitution des Echinacosids, eines Glykosids aus den Wurzeln von Echinacea angustifolia D. C. 6. Mitteilung über antibakterielle Stoffe". Helvetica Chimica Acta. 33 (6): 1877–1893. doi:10.1002/hlca.19500330657.
This article is issued from Wikipedia - version of the 1/28/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.