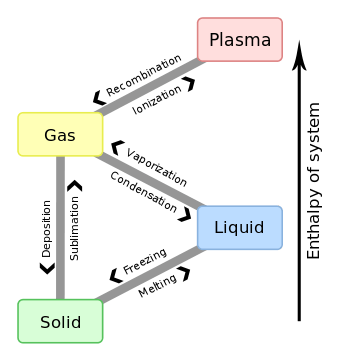
Enthalpy of sublimation
The enthalpy of sublimation, or heat of sublimation, is the heat required to change one mole of a substance from solid state to gaseous state at a given combination of temperature and pressure, usually standard temperature and pressure (STP). The heat of sublimation is usually expressed in kJ/mol, although the less customary kJ/kg is also encountered.
Sublimation enthalpy substances
| symbol | substances | Sublimation enthalpy (kJ/mol) |
|---|---|---|
| Na | sodium | 108 |
| K | potassium | 89 |
| Rb | rubidium | 82 |
| Cs | cesium | 78 |
| Mg | magnesium | 150 |
| Ca | calcium | 192 |
| Sr | strontium | 164 |
| Ba | barium | 176 |
| C10H8 | naphthalene | 72.9 |
| CO2 | carbon dioxide | 25 |
See also
This article is issued from Wikipedia - version of the 4/27/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.