Heptasulfur imide
 | |
 | |
| Identifiers | |
|---|---|
| 293-42-5 | |
| Properties | |
| HNS7 | |
| Appearance | pale yellow solid |
| Density | 2.01 g/cm3 |
| Melting point | 113.5 °C (236.3 °F; 386.6 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
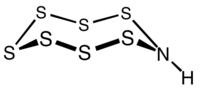
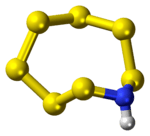
Heptasulfur imide is the inorganic compound with the formula S7NH. It is a pale yellow solid that is, like elemental sulfur, highly soluble in carbon disulfide. The compound, which is only of academic interest, is representative of a family of sulfur imides Sx(NH)y.
Synthesis and structure
It is prepared by reaction of disulfur dichloride with ammonia,[1] although other methods have been developed.[2] Together with S7NH, the reaction also produces three isomers of S6(NH)2 and two isomers of S5(NH)3.
It is an analogue of S8, with one S replaced by NH. The S2NH center is almost planar,[3] suggesting that the amine is nonbasic.
References
- ↑ Becke-Goehring, Margot; Fluck, Ekkehard "Heptasulfur imide" Inorganic Syntheses 1966, vol. 8, 103-5. doi:10.1002/9780470132395.ch26
- ↑ Bojes, J.; Chivers, T.; Drummond, I. "Heptathiazocine(heptasulfurimide) and tetrabutylammonium tetrathionitrate" Inorganic Syntheses (1978), 18, 203-6. doi:10.1002/9780470132494.ch36
- ↑ Hecht, H. J.; Reinhardt, R.; Steudel, R.; Bradaczek, H. "Redetermination of the crystal and molecular structure of heptasulfur imide, S7NH" Zeitschrift für Anorganische und Allgemeine Chemie 1976, vol. 426, pp. 43-8. doi:10.1002/zaac.19764260106
This article is issued from Wikipedia - version of the 2/12/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.