Magnaporthe grisea
| Magnaporthe grisea | |
|---|---|
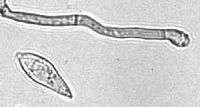 | |
| A conidium and conidiogenous cell of M. grisea | |
| Scientific classification | |
| Kingdom: | Fungi |
| Phylum: | Ascomycota |
| Class: | Sordariomycetes |
| Order: | Magnaporthales |
| Family: | Magnaporthaceae |
| Genus: | Magnaporthe |
| Species: | M. grisea |
| Binomial name | |
| Magnaporthe grisea (T.T. Hebert) M.E. Barr | |
| Synonyms | |
|
Ceratosphaeria grisea T.T. Hebert, (1971) | |
Magnaporthe grisea, also known as rice blast fungus, rice rotten neck, rice seedling blight, blast of rice, oval leaf spot of graminea, pitting disease, ryegrass blast, and Johnson spot,[1][2][3][4][5][6][7] is a plant-pathogenic fungus that causes a serious disease affecting rice. It is now known that M. grisea consists of a cryptic species complex containing at least two biological species that have clear genetic differences and do not interbreed.[8] Complex members isolated from Digitaria have been more narrowly defined as M. grisea. The remaining members of the complex isolated from rice and a variety of other hosts have been renamed Magnaporthe oryzae. Confusion on which of these two names to use for the rice blast pathogen remains, as both are now used by different authors.
Members of the Magnaporthe grisea complex can also infect other agriculturally important cereals including wheat, rye, barley, and pearl millet causing diseases called blast disease or blight disease. Rice blast causes economically significant crop losses annually. Each year it is estimated to destroy enough rice to feed more than 60 million people. The fungus is known to occur in 85 countries worldwide.[9]
Hosts and symptoms


M. grisea is an ascomycete fungus. It is an extremely effective plant pathogen as it can reproduce both sexually and asexually to produce specialized infectious structures known as appressoria that infect aerial tissues and hyphae that can infect root tissues.
Rice blast has been observed on rice strains M-201, M-202, M-204, M-205, M-103, M-104, S-102, L-204, Calmochi-101, with M-201 being the most vulnerable.[10] Initial symptoms are white to gray-green lesions or spots with darker borders produced on all parts of the shoot, while older lesions are elliptical or spindle-shaped and whitish to gray with necrotic borders. Lesions may enlarge and coalesce to kill the entire leaf. Symptoms are observed on all above-ground parts of the plant.[11] Lesions can be seen on the leaf collar, culm, culm nodes, and panicle neck node. Internodal infection of the culm occurs in a banded pattern. Nodal infection causes the culm to break at the infected node (rotten neck).[12] It also affects reproduction by causing the host to produce fewer seeds. This is caused by the disease preventing maturation of the actual grain.[9]
Disease cycle
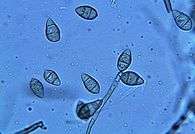
The pathogen infects as a spore that produces lesions or spots on parts of the rice plant such as the leaf, leaf collar, panicle, culm and culm nodes. Using a structure called an appressorium, the pathogen penetrates the plant. M. grisea then sporulates from the diseased rice tissue to be dispersed as conidiospores.[13] After overwintering in sources such as rice straw and stubble, the cycle repeats.[9]
A single cycle can be completed in about a week under favorable conditions where one lesion can generate up to thousands of spores in a single night. With the ability to continue to produce the spores for over 20 days, rice blast lesions can be devastating to susceptible rice crops.[14]
Environment
Rice blast is a significant problem in temperate regions and can be found in areas such as irrigated lowland and upland.[15] Conditions conducive for rice blast include long periods of free moisture where leaf wetness is required for infection and high humidity is common.[15] Sporulation increases with high relative humidity and at 77-82 degrees F, spore germination, lesion formation, and sporulation are at optimum levels.[9]
In terms of control, excessive use of nitrogen fertilization as well as drought stress increase rice susceptibility to the pathogen as the plant is placed in a weakened state and its defenses are low.[9] Extended drain periods also favor infection as they aerate the soil, converting ammonium to nitrate and thus causing stress to rice crops, as well.[9]
Management

The fungus has been able to establish resistance to both chemical treatments and genetic resistance in some types of rice developed by plant breeders. It is thought that the fungus can achieve this by genetic change through mutation. In order to most effectively control infection by M. grisea, an integrated management program should be implemented to avoid overuse of a single control method and fight against genetic resistance. For example, eliminating crop residue could reduce the occurrence of overwintering and discourage inoculation in subsequent seasons. Another strategy would be to plant resistant rice varieties that are not as susceptible to infection by M. grisea.[9] Knowledge of the pathogenicity of M. grisea and its need for free moisture suggest other control strategies such as regulated irrigation and a combination of chemical treatments with different modes of action.[9] Managing the amount of water supplied to the crops limits spore mobility thus dampening the opportunity for infection. Chemical controls such as Carpropamid have been shown to prevent penetration of the appressoria into rice epidermal cells, leaving the grain unaffected.[16]
Importance
Rice blast is the most important disease concerning the rice crop in the world. Since rice is an important food source for much of the world, its effects have a broad range. It has been found in over 85 countries across the world and reached the United States in 1996. Every year the amount of crops lost to rice blast could feed 60 million people. Although there are some resistant strains of rice, the disease persists wherever rice is grown. The disease has never been eradicated from a region.[17]
References
- ↑ Talbot, N. J. (2003). "ON THE TRAIL OF A CEREAL KILLER: Exploring the Biology of Magnaporthe grisea". Annual Review of Microbiology. 57: 177–202. doi:10.1146/annurev.micro.57.030502.090957. PMID 14527276.
- ↑ Zeigler, RS; Leong, SA; Teeng, PS (1994). "Rice Blast Disease." Wallingford: CAB International.
- ↑ Wilson, R. A.; Talbot, N. J. (2009). "Under pressure: Investigating the biology of plant infection by Magnaporthe oryzae". Nature Reviews Microbiology. 7 (3): 185–95. doi:10.1038/nrmicro2032. PMID 19219052.
- ↑ Sesma, A.; Osbourn, A. E. (2004). "The rice leaf blast pathogen undergoes developmental processes typical of root-infecting fungi". Nature. 431 (7008): 582. doi:10.1038/nature02880. PMID 15457264.
- ↑ Dean, R. A.; Talbot, N. J.; Ebbole, D. J.; Farman, M. L.; Mitchell, T. K.; Orbach, M. J.; Thon, M; Kulkarni, R; Xu, J. R.; Pan, H; Read, N. D.; Lee, Y. H.; Carbone, I; Brown, D; Oh, Y. Y.; Donofrio, N; Jeong, J. S.; Soanes, D. M.; Djonovic, S; Kolomiets, E; Rehmeyer, C; Li, W; Harding, M; Kim, S; Lebrun, M. H.; Bohnert, H; Coughlan, S; Butler, J; Calvo, S; et al. (2005). "The genome sequence of the rice blast fungus Magnaporthe grisea". Nature. 434 (7036): 980–6. doi:10.1038/nature03449. PMID 15846337.
- ↑ Couch, B. C.; Kohn, L. M. (2002). "A multilocus gene genealogy concordant with host preference indicates segregation of a new species, Magnaporthe oryzae, from M. Grisea". Mycologia. 94 (4): 683–93. doi:10.2307/3761719. PMID 21156541.
- ↑ Magnaporthe grisea at Crop Protection Compendium, CAB International
- ↑ Couch, B. C.; Fudal, I; Lebrun, M. H.; Tharreau, D; Valent, B; Van Kim, P; Nottéghem, J. L.; Kohn, L. M. (2005). "Origins of host-specific populations of the blast pathogen Magnaporthe oryzae in crop domestication with subsequent expansion of pandemic clones on rice and weeds of rice". Genetics. 170 (2): 613–30. doi:10.1534/genetics.105.041780. PMC 1450392
 . PMID 15802503.
. PMID 15802503. - 1 2 3 4 5 6 7 8 S.C. Scardaci; et al. (2003). "Rice Blast: A New Disease in California". University of California-Davis: Agronomy Fact Sheet Series 1997-2. Archived from the original on 2006-09-11. Retrieved 2014-02-25.
- ↑ Rice Blast at University of California Integrated Pest Management
- ↑ Rice Blast at the Online Information Service for Non-Chemical Pest Management in the Tropics
- ↑ Rice Blast at Factsheets on Chemical and Biological Warfare Agents
- ↑ Agrios, George N. (2005). Plant Pathology. Amsterdam: Elsevier Academic Press.
- ↑ Diagnostic Methods for Rice Blast at PaDIL Plant Biosecurity Toolbox
- 1 2 Kuyek, Devlin (2000). "Implications of corporate strategies on rice research in asia". Grain. Retrieved 2010-10-20.
- ↑ Kurahasi, Yoshio (1997). "Biological Activity of Carpropamid (KTU 3616): A new fungicide for rice blast disease". Journal of Pesticide Science. Retrieved 2014-02-25.
- ↑ Rice Blast at Cereal Knowledge Bank
Further reading
- California EPA. Rice Crop Infestation in Three Counties Leads To Emergency Burn Agreement, February 11, 1998
- Kadlec, RP. Biological Weapons for Waging Economic Warfare, Air & Space Power Chronicles
- NSF. Microbial Genome Helps Blast Devastating Rice Disease, April 21, 2005
- United States Congress. Testimony of Dr. Kenneth Alibek, 1999
External links
| Wikimedia Commons has media related to Magnaporthe oryzae. |
- GROMO - Genomic Resources of Magnaporthe oryzae
- Magnaporthe grisea Genome
- The official Website of the International Rice Blast Genome Consortium
- Index Fungorum
- Magnaporthe grisea at MetaPathogen: stages, tissues, mating types, strains, references
- Facebook page for Magnaporthe oryzae