PAC-1
This article refers to the anti-tumor molecule, and not the a2iib3 integrin activation specific antibody of the same name
 | |
| Identifiers | |
|---|---|
| |
| CAS Number |
315183-21-2 |
| PubChem (CID) | 1185541 |
| IUPHAR/BPS | 5238 |
| ChemSpider |
12272376 |
| UNII |
9LIS8N0B2C |
| ECHA InfoCard | 100.164.322 |
| Chemical and physical data | |
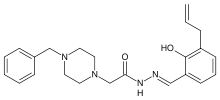
| Formula | C23H28N4O2 |
| Molar mass | 392.494 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| (verify) | |
PAC-1 (first procaspase activating compound) is a synthesized chemical compound that selectively induces apoptosis, or cell suicide, in cancerous cells. PAC-1 is known for the unique path it has taken to the clinic, as it is perhaps the only cancer drug to first be rigorously evaluated in pet dogs with spontaneous cancer as a prelude to the human clinical trial. If PAC-1 were to become an approved drug it could lead to more drug candidates being developed in this fashion. In 2010 a published study showed PAC-1 to be safe to research dogs, and a second study published later that same year reported that a PAC-1 derivative (called S-PAC-1) was well tolerated in a small Phase I Clinical Trial of pet dogs with lymphoma. More recently, in addition to this single-agent activity PAC-1 has shown to potently synergize with approved cancer drugs, for example with doxorubicin in the treatment of pet dogs with lymphoma and metastatic osteosarcoma. In 2011 Vanquish Oncology Inc. was founded to move PAC-1 forward to a human clinical trial, in 2013 Vanquish Oncology announced a multimillion-dollar angel investment into the company, in 2015 a Phase 1 clinical trial of PAC-1 opened for enrollment of cancer patients, and in 2016 it was announced that PAC-1 had been granted Orphan Drug Designation for treatment of glioblastoma by the FDA.
Function and discovery
PAC-1 (pronounced "pack one") was discovered in Professor Paul Hergenrother's laboratory at the University of Illinois at Urbana-Champaign during a process that screened many chemicals for anti-tumor potential. This molecule, when delivered to cancer cells, signals the cells to self-destruct by activating an "executioner" protein, procaspase-3. Then, the activated executioner protein begins a cascade of events that destroys the machinery of the cell.
Uses of apoptosis in the body and its irregularities
This cascade of events is named apoptosis. Apoptosis is self-induced in cells to combat infections or DNA damage. For instance, when a cell in one's body is infected with a bacterium or virus, it will self-destruct to take away the resources needed by the virus to proliferate. Apoptosis is also found to help in embryo development (destroying the webbing in between an embryo's fingers to separate the fingers) and the regular replenishment of cells that are constantly being used up or destroyed (cells that line the intestinal tract), also called homeostasis.
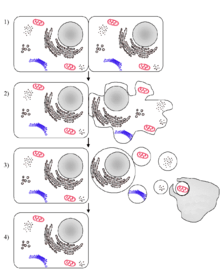
The problem lies when one part of the apoptosis pathway is broken. Normally, the balance between cell division and apoptosis is rigorously regulated to keep the integrity of organs and tissues. Examples of broken apoptosis pathways occur in many cancers. If old lung cells cannot self-destruct to make room for new lung cells, a large mass of cells form and a tumor is made.
In many cases, the apoptotic pathway is disrupted because procaspase-3, the executioner protein, cannot be activated by the cell. This is analogous to an executioner who does not have orders to kill. Without the orders, the condemned will not die. The same analogy can be made with procaspase-3. Without activated procaspase-3, the apoptotic cascade will not occur and the cell will not destroy itself no matter how necessary it may be. PAC-1 acts a replacement order that works and bypasses the lawyers, court orders, and governor's calls. It will activate procaspase-3 indiscriminately.
How PAC-1 affects the apoptotic process
In cells, the executioner protein, caspase-3, is stored in its inactive form, procaspase-3. This way, the cell can quickly undergo apoptosis by activating the protein that is already there. This inactive form is called a zymogen. Procaspase-3 is known to be inhibited by low levels of zinc. PAC-1 activates procaspase-3 by chelating zinc, thus relieving the zinc-mediated inhibition. This allows procaspase-3 to be an active enzyme, and it can then cleave another molecule of procaspase-3 to active caspase-3. Caspase-3 can further activate other molecules of procaspase-3 in the cell, causing an exponential increase in caspase-3 concentration. PAC-1 facilitates this process and causes the cell to undergo apoptosis quickly.[1]
This direct procaspase-3 activation mode-of-action for PAC-1 has been confirmed by other laboratories: In 2013 by the Megeney lab during the course of studies on the role of caspase-3 in cardiomyocytes,[2] in 2014 by the Wu lab in an extensive study on the anticancer activity and mode-of-action of PAC-1 and derivatives,[3] and in 2015 by the Gandhi lab in an exploration of the potential of PAC-1 and derivative B-PAC-1 for treatment of chronic lymphocytic leukemia (CLL).[4]
A potential selectivity problem arises because procaspase-3 is present in most cells of the body. However, it has been shown that in many cancers, including certain neuroblastomas, lymphomas, leukemias, melanomas, and liver cancers, procaspase-3 is present in higher concentrations.[1] For instance, lung cancer cells can have over 1000 times more procaspase-3 than normal cells.[1] Therefore, by controlling the dosage, one can achieve selectivity between normal and cancerous cells.
In addition to its stand-alone activity, PAC-1 has also been shown to markedly synergize with a variety of approved cancer drugs, for example with BRAF and MEK inhibitors in mouse models of melanoma,[5] and with conventional chemotherapeutic agents such as doxorubicin in pet dogs with spontaneous cancers including lymphoma and metastatic osteosarcoma.[6]
Further research and human application
Thus far, PAC-1 seems promising as a new anti-tumor drug. Vanquish Oncology reported their intention to begin a Phase I human clinical trial in cancer patients to begin in early 2015, and indeed the Phase 1 trial of PAC-1 at the University of Illinois Cancer Center in Chicago and at the Sidney Kimmel Cancer Center at Johns Hopkins opened for enrollment in February 2015 (NCT02355535).
References
- 1 2 3 Putt KS, Chen GW, Pearson JM, Sandhorst JS, Hoagland MS, Kwon JT, Hwang SK, Jin H, Churchwell MI, Cho MH, Doerge DR, Helferich WG, Hergenrother PJ (2006). "Small-molecule activation of procaspase-3 to caspase-3 as a personalized anticancer strategy l". Nature Chemical Biology. 2 (10): 543–50. doi:10.1038/nchembio814. PMID 16936720.
- ↑ Putinski C, Abdul-Ghani M, Stiles R, Brunette S, Dick SA, Fernando P, Megeney LA (2013). "Intrinsic-mediated caspase activation is essential for cardiomyocyte hypertrophy". Proceedings of the National Academy of Sciences. 110 (43): E4079–87. doi:10.1073/pnas.1315587110. PMC 3808644
 . PMID 24101493.
. PMID 24101493.
- ↑ Wang F, Wang L, Zhao Y, Li Y, Ping G, Xiao S, Chen K, Zhu W, Gong P, Yang J, Wu C (2014). "A novel small-molecule activator of procaspase-3 induces apoptosis in cancer cells and reduces tumor growth in human breast, liver and gallbladder cancer xenografts". Molecular Oncology. 8 (8): 1640–1652. doi:10.1016/j.molonc.2014.06.015.
- ↑ Patel V, Balakrishnan K, Keatin MJ, Wierda WG, Gandhi V (2015). "Expression of executioner procaspases and their activation by a procaspase-activating compound in chronic lymphocytic leukemia cells". Blood. 125 (7): 1126–1136. doi:10.1182/blood-2014-01-546796.
- ↑ Peh J, Fan TM, Wycislo KL, Roth HS, Hergenrother PJ (2016). "The Combination of Vemurafenib and Procaspase-3 Activation is Synergistic in Mutant BRAF Melanomas". Molecular Cancer Therapeutics. 15: 1859–1869. doi:10.1158/1535-7163.MCT-16-0025.
- ↑ Botham RC, Roth HS, Book AP, Roady PJ, Fan TM, Hergenrother, PJ (2016). "Small-Molecule Procaspase-3 Activation Sensitizes Cancer to Treatment with Diverse Chemotherapeuticss". ACS Central Science. 2: 545–559. doi:10.1021/acscentsci.6b00165.
- Peterson, Q. P.; Goode, D. R.; West, D. C.; Ramsey, K. N.; Lee, J. J.; Hergenrother, P. J. (2009). "PAC-1 Activates Procaspase-3 in vitro Through Relief of Zinc-Mediated Inhibition". J. Mol. Biol. 388 (1): 144–158. doi:10.1016/j.jmb.2009.03.003. PMC 2714579
 . PMID 19281821.
. PMID 19281821. - Peterson, Q. P.; Hsu, D. C.; Goode, D. R.; Novotny, C. J.; Totten, R. K. Hergenrother (2009). "Procaspase-3 Activation as an Anti-Cancer Strategy: Structure-Activity Relationship of PAC-1, and its Cellular Co-Localization with Caspase-3". J. Med. Chem. 52 (18): 5721–5731. doi:10.1021/jm900722z. PMID 19708658.
- Lucas, P. W.; Schmit, J. M.; Peterson, Q. P.; West, D. C.; Hsu, D. C.; Novotny, C. J.; Dirikoul, L.; Deorge, D. R.; Garrett, L. D.; et al. (2011). "Pharmacokinetics and Derivation of an Anticancer Dosing Regimen for PAC-1, a Preferential Small Molecule Activator of Procaspase-3, in Healthy Dogs". Invest. New Drugs. 29 (5): 901–911. doi:10.1007/s10637-010-9445-z. PMID 20499133.
- Peterson, Q. P.; Hsu, D. C.; Novotny, C. J.; West, D. C.; Kim, D.; Schmit, J. M.; Dirikolu, L.; Hergenrother, P. J.; Fan, T. M.; et al. (2010). "Discovery and Canine Preclinical Assessment of a Nontoxic Procaspase-3-Activating Compound". Cancer Res. 70 (18): 7232–7241. doi:10.1158/0008-5472.can-10-0766. PMID 20823163.
- West, D. C.; Qin, Y.; Peterson, Q. P.; Thomas, D. L.; Palchaudhuri, R. P.; Morrison, K. C.; Lucas, P. W.; Palmer, A. E.; Fan, T. M.; et al. (2012). "Differential effects of procaspase-3 activating compounds in the induction of cancer cell death". Mol. Pharmaceutics. 9: 1425–1434.
- Botham, R. C.; Fan, T. M.; Im, I.; Borst, L. B; Dirikolu, L.; Hergenrother, P. J. (2014). "Dual Small-Molecule Targeting of Procaspase-3 Dramatically Enhances Zymogen Activation and Anticancer Activity". J. Am. Chem. Soc. 136 (4): 1312–1319. doi:10.1021/ja4124303. PMID 24383395.
External links
- Cancer cell 'executioner' found. BBC News 27 August 2006.
- Cancer cells 'can live forever'. BBC News 29 April 2004.
- Vanquish Oncology
- Angel investment helps move PAC-1 forward
- FDA grants PAC-1 Orphan Drug Designation for glioma
- Phase 1 clinical trial of PAC-1 in human cancer patients clinicaltrials.gov