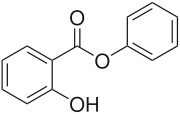
Phenyl salicylate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Phenyl 2-hydroxybenzoate | |
| Other names
Salol | |
| Identifiers | |
| 118-55-8 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:34918 |
| ChEMBL | ChEMBL1339216 |
| ChemSpider | 8058 |
| ECHA InfoCard | 100.003.873 |
| EC Number | 204-259-2 |
| KEGG | C14163 |
| MeSH | C026041 |
| PubChem | 8361 |
| |
| |
| Properties | |
| C13H10O3 | |
| Molar mass | 214.22 g/mol |
| Appearance | White solid |
| Density | 1.25 g/cm3 |
| Melting point | 41.5 °C (106.7 °F; 314.6 K) |
| Boiling point | 173 °C (343 °F; 446 K) at 12 mmHg |
| 1 g/6670 mL | |
| Refractive index (nD) |
1.615[2] |
| Pharmacology | |
| G04BX12 (WHO) | |
| Hazards | |
| Flash point | 137.3[2] °C (279.1 °F; 410.4 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Phenyl salicylate, or salol, is a chemical substance, introduced in 1886 by Marceli Nencki of Basel. It can be created by heating salicylic acid with phenol. Once used in sunscreens, phenyl salicylate is now used in the manufacture of some polymers, lacquers, adhesives, waxes and polishes.[1] It is also used frequently in school laboratory demonstrations on how cooling rates affect crystal size in igneous rocks.
Salol reaction
In the salol reaction, phenyl salicylate reacts with o-toluidine in 1,2,4-trichlorobenzene at elevated temperatures to the corresponding amide o-Salicylotoluide.[3] Salicylamides are one type of drug.
Medical
It has been used as an antiseptic[4] based on the antibacterial activity upon hydrolysis in the small intestine.
It acts as a mild analgesic.[5]
References
- 1 2 Merck Index, 11th Edition, 7282.
- 1 2 ChemBK Chemical Database http://www.chembk.com/en/chem/Phenyl%20salicylate
- ↑ Allen, C. F. H.; VanAllan, J. (1946). "SALICYL-o-TOLUIDE" (PDF). Org. Synth. 26: 92.; Coll. Vol., 3, p. 765
- ↑ Walter Sneader (2005). Drug discovery: a history. John Wiley and Sons. pp. 358–. ISBN 978-0-471-89980-8. Retrieved 28 October 2010.
- ↑ Judith Barberio (4 September 2009). Nurse's Pocket Drug Guide, 2010. McGraw Hill Professional. pp. 57–. ISBN 978-0-07-162743-6. Retrieved 28 October 2010.