Critical period
In developmental psychology and developmental biology, a critical period is a maturational stage in the lifespan of an organism during which the nervous system is especially sensitive to certain environmental stimuli. If, for some reason, the organism does not receive the appropriate stimulus during this "critical period" to learn a given skill or trait, it may be difficult, ultimately less successful, or even impossible, to develop some functions later in life. Functions that are indispensable to an organism's survival, such as vision, are particularly likely to develop during critical periods. "Critical period" also relate to ability to acquire first language. Researchers found that people who passed "critical period" would not acquire first language fluently.[1]
Some researchers differentiate between 'critical' and 'sensitive' periods—defining 'sensitive' periods as more extended periods, after which learning is still possible.[2] Other researchers consider these the same phenomenon.[3]
For example, the critical period for the development of a human child's binocular vision is thought to be between three and eight months, with sensitivity to damage extending up to at least three years of age. Further critical periods have been identified for the development of hearing[4] and the vestibular system. There are critical periods during early postnatal development in which imprinting can occur, such as when a greylag goose becomes attached to a parent figure within the first 36 hours after hatching. A young chaffinch must hear an adult singing before it sexually matures, or it never properly learns the highly intricate song.
Confirming the existence of a critical period for a particular ability requires evidence that there is a point after which the associated behavior is no longer correlated with age, and ability stays at the same level. Some experimental research into critical periods has involved depriving animals of stimuli at different stages of development, while other studies have looked at children deprived of certain experiences due to illness (such as temporary blindness), or social isolation (such as feral children). Many of the studies investigating a critical period for language acquisition have focused on deaf children of hearing parents.
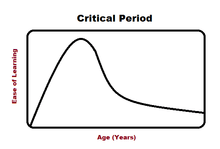
Linguistics
First language acquisition
The Critical Period Hypothesis (CPH) states that the first few years of life constitute the time during which language develops readily and after which (sometime between age 5 and puberty) language acquisition is much more difficult and ultimately less successful.[5] The hypothesis that language is acquired during a critical period was first proposed by neurologists Wilder Penfield and Lamar Roberts in 1959 and popularized by linguist Eric H. Lenneberg in 1967. Lenneberg argued for the hypothesis based on evidence that children who experience brain injury early in life develop far better language skills than adults with similar injuries.
The two most famous cases of children who failed to acquire language after the critical period are Genie and the feral child Victor of Aveyron.[6] However, the tragic circumstances of these cases and the moral and ethical impermissibility of replicating them make it difficult to draw conclusions about them. The children may have been cognitively disabled from infancy, or their inability to develop language may have resulted from the profound neglect and abuse they suffered.[5]
Many subsequent researchers have further developed the CPH, most notably Elissa Newport and Rachel Mayberry. Studies conducted by these researchers demonstrated that profoundly deaf individuals who are not exposed to a sign language as children never achieve full proficiency, even after 30 years of daily use.[7] While the effect is most profound for individuals who receive no sign language input until after the age of 12, even those deaf people who began learning a sign language at age 5 were significantly less fluent than native deaf signers (whose exposure to a sign language began at birth). Early language exposure also affects the ability to learn a second language later in life: profoundly deaf individuals with early language exposure achieve comparable levels of proficiency in a second language to hearing individuals with early language exposure. In contrast, deaf individuals without early language exposure perform far worse.[8]
Steven Pinker discusses the CPH in his book, The Language Instinct. According to Pinker, language must be viewed as a concept rather than a specific language for the sounds, grammar, meaning, vocabulary, and social norms play an important role in the acquisition of language.[9] Physiological changes in the brain are also conceivable causes for the terminus of the critical period for language acquisition.[10] As language acquisition is crucial during this phase, similarly infant-parent attachment is crucial for social development of the infant. An infant learns to trust and feel safe with the parent, but there are cases in which the infant might be staying at an orphanage where it does not receive the same attachment with their caregiver. Research shows that infants who were unable to develop this attachment had major difficulty in keeping close relationships, and had maladaptive behaviors with adopted parents.[1]
Other evidence comes from neuropsychology where it is known that adults well beyond the critical period, are more likely to suffer permanent language impairment from brain damage than are children, believed to be due to youthful resiliency of neural reorganization.[5]
Second language acquisition
The theory has often been extended to a critical period for Second Language Acquisition (SLA), which has influenced researchers in the field on both sides of the spectrum, supportive and unsupportive of CPH, to explore.[11] However, the nature of this phenomenon has been one of the most fiercely debated issues in psycholinguistics and cognitive science in general for decades.
Certainly, older learners of a second language rarely achieve the native-like fluency that younger learners display, despite often progressing faster than children in the initial stages. This is generally accepted as evidence supporting the CPH. Incorporating the idea, "younger equals better" by Penfield, David Singleton (1995) states that in learning a second language there are many exceptions, noting that five percent of adult bilinguals master a second language even though they begin learning it when they are well into adulthood — long after any critical period has presumably come to a close. The critical period hypothesis holds that first language acquisition must occur before cerebral lateralization completes, at about the age of puberty. One prediction of this hypothesis is that second language acquisition is relatively fast, successful, and qualitatively similar to first language only if it occurs before the age of puberty.[12] To grasp a better understanding of SLA, it is essential to consider linguistic, cognitive, and social factors rather than age alone, as they are all essential to the learner's language acquisition.[11]
Vision

In mammals, neurons in the brain, which process vision, actually develop after birth based on signals from the eyes. A landmark experiment by David H. Hubel and Torsten Wiesel (1963) showed that cats that had one eye sewn shut from birth to three months of age (monocular deprivation) only fully developed vision in the open eye. They showed that columns in the primary visual cortex receiving inputs from the other eye took over the areas that would normally receive input from the deprived eye. In general electrophysiological analyses of axons and neurons in the lateral geniculate nucleus showed that the visual receptive field properties was comparable to adult cats. However, the layers of cortex that were deprived had less activity and fewer responses were isolated. The kittens had abnormally small ocular dominance columns (part of the brain that processes sight) connected to the closed eye, and abnormally large, wide columns connected to the open eye. Because the critical period time had elapsed, it would be impossible for the kittens to alter and develop vision in the closed eye. This did not happen to adult cats even when one eye was sewn shut for a year because they had fully developed their vision during their critical period. Later experiments in monkeys found similar results.[13]
In a follow-up experiment, Hubel and Wiesel (1963) explored the cortical responses present in kittens after binocular deprivation; they found it difficult to find any active cells in the cortex, and the responses they did get were either slow-moving or fast-fatiguing. Furthermore, the cells that did respond selected for edges and bars with distinct orientation preferences. Nevertheless, these kittens developed normal binocularity. Hubel and Wiesel first explained the mechanism, known as orientation selectivity, in the mammalian visual cortex. Orientation tuning, a model that originated with their model, is a concept in which receptive fields of neurons in the LGN excite a cortical simple cell and are arranged in rows. This model was important because it was able to describe a critical period for the proper development of normal ocular dominance columns in the lateral geniculate nucleus, and thus able to explain the effects of monocular deprivation during this critical period. The critical period for cats is about three months and for monkeys, about six months.[14]
In a similar experiment, Antonini and Stryker (1993) examined the anatomical changes that can be observed after monocular deprivation. They compared geniculocortical axonal arbors in monocularly deprived animals in the long term (4- weeks) to short term (6–7 days) during the critical period established by Hubel and Wiesel (1993). They found that in the long term, monocular deprivation causes reduced branching at the end of neurons, while the amount of afferents allocated to the nondeprived eye increased. Even in the short term, Antonini and Stryker (1993) found that geniculocortical neurons were similarly affected. This supports the aforementioned concept of a critical period for proper neural development for vision in the cortex.[15]
In humans, some babies are born blind in one or both eyes, for example, due to cataracts. Even when their vision is restored later by treatment, their sight would not function in the normal way as for someone who had binocular vision from birth or had surgery to restore vision shortly after birth. Therefore, it is important to treat babies born blind soon if their condition is treatable.
Expression of the protein Lynx1 has been associated with the normal end of the critical period for synaptic plasticity in the visual system.[16]
Imprinting
In psychology, imprinting is any type of rapid learning that occurs in a particular life stage. While this rapid learning is independent of the behavioral outcome, it also establishes it and can effect behavioral responses to different stimuli. Konrad Lorenz is well known for his classic studies of filial imprinting in graylag geese. From 1935 to 1938, he presented himself to a group of newly hatched gosling and took note of how he was instantly accepted, followed, and called to as if he were the one who laid them himself. As the first moving object they encountered, Lorenz studied the phenomenon in how quickly the geese were able to form such an irreversible bond. Through his work he demonstrated that this only developed during a brief “critical period,” which was about a few hours after hatching.[17] Lorenz also discovered a long-lasting effect of his studies, and that was a shift in the species' sexual imprinting as a result from imprinting upon a foster mother of a second species. For certain species, when raised by a second one, they develop and retain imprinted preferences and approach the second species they were raised by rather than choose their own, if given a choice.[18]
Imprinting serves as the distinguishing factor between one's own mother and other mother figures. The mother and the infant both identify with each other, this a strong bonding moment for humans. It provides a sort of model or guide to adult behaviors in addition to other factors such as nurture, protection in infancy, guidance, and nourishment. The imprinting process, Lorenz also found, brought about a sense of familiarity for the young animals. When such a strong bond is formed at such an early stage, it creates a sense of security and comfort for the subject and actually encourages the imprinting behavior.
Pheromones play a key role in the imprinting process, they trigger a biochemical response in the recipient, leading to a confirmed identification in the other individual. If direct contact between mother and infant is not maintained during the critical imprinting period, then the mother goose may reject the infant because she is unfamiliar with her newborn's scent. If that does happen, then the infant's life would be in jeopardy unless it were claimed by a substitute mother and if it failed to imprint would, that trigger psychological trauma, possibly leading to awkward social behavior in later life.[19] In relation to humans, a newborn during the critical period identifies with its mother's and other peoples' scents since its scent is one of the most developed senses at that stage in life. The newborn uses this pheromone identification to seek the people it identifies with, when in times of distress, hunger, and discomfort as a survival skill.[20] Inferences could be made for newborns based upon Lorenz's studies. When imprinting on their mothers, newborns look to them for nourishment, a sense of security, and comfort. Human newborns are among the most helpless newborns known, and with very little that they can do for themselves the most they could do is form bonds the people that they could depend on to provide them the essentials needed. Imprinting is a crucial factor of the critical period because it facilitates the newborn's abilities to form bonds with other people, from infancy to adulthood.
Auditory processing
Many studies have supported a correlation between the type of auditory stimuli present in the early postnatal environment and the development on the topographical and structural development of the auditory system.[4]
First reports on critical periods came from deaf children and animals that received a cochlear implant to restore hearing. Approximately at the same time, both an electroencephalographic study by Sharma, Dorman and Spahr [21] and an in-vivo investigation of the cortical plasticity in deaf cats by Kral and colleagues [22] demonstrated that the adaptation to the cochlear implant is subject to an early, developmental sensitive period. The closure of sensitive periods likely involves a multitude of processes that in their combination make it difficult to reopen these behaviorally.[4] The understanding of the mechanisms behind critical periods has consequences for medical therapy of hearing loss.[23] M. Merzenich and colleagues showed that during an early critical period, noise exposure can affect the frequency organization of the auditory cortex.[24]
Recent studies have examined the possibility of a critical period for thalamocortical connectivity in the auditory system. For example, Zhou and Merzenich (2008) studied the effects of noise on development in the primary auditory cortex in rats. In their study, rats were exposed to pulsed noise during the critical period and the effect on cortical processing was measured. Rats that were exposed to pulsed noise during the critical period had cortical neurons that were less able to respond to repeated stimuli; the early auditory environment interrupted normal structural organization during development.
In a related study, Barkat, Polley and Hensch (2011) looked at how exposure to different sound frequencies influences the development of the tonotopic map in the primary auditory cortex and the ventral medical geniculate body. In this experiment, mice were reared either in normal environments or in the presence of 7 kHz tones during early postnatal days. They found that mice that were exposed to an abnormal auditory environment during a critical period P11- P15 had an atypical tonotopic map in the primary auditory cortex.[25] These studies support the notion that exposure to certain sounds within the critical period can influence the development of tonotopic maps and the response properties of neurons. Critical periods are important for the development of the brain for the function from a pattern of connectivity. In general, the early auditory environment influences the structural development and response specificity of the primary auditory cortex.[26]
Musical ability
Absolute pitch manifests itself almost always before adolescence and rarely if ever among individuals who are first exposed to music after mid-childhood, suggesting that exposure to music or similar phenomena (e.g., tonal languages) in early to mid-childhood is a necessary condition for its development or refinement. Studies that ask musicians and non-musicians to sing or hum well-known popular songs that have definitive recordings (and hence are sung in standardized keys) show that—on average—participants sing within a semitone of the standardized key but that outside the small subset of participants with absolute pitch there is broad variation (the "bell curve" that reflects the degree of approximation to the standard key is broad and flat).{{{1}}} These results suggest that almost all humans have some innate aptitude for absolute pitch recognition—though other factors may enhance or limit the level of that aptitude. Also, the results' conjunction with the aforementioned chronological observations suggests that early to mid-childhood exposure to environments whose interpretation depends on pitch is a developmental "trigger" for whatever aptitude an individual possesses.
Vestibular system
In our vestibular system, neurons are undeveloped at neuronal birth and mature during the critical period of the first 2-3 postnatal weeks. Hence, disruption of maturation during this period can cause changes in normal balance and movement through space. Animals with abnormal vestibular development tend to have irregular motor skills.[27] Studies have consistently shown that animals with genetic vestibular deficiencies during this critical period have altered vestibular phenotypes, most likely as a result of lack insufficient input from the semicircular canals and dopaminergic abnormalities. Moreover, exposure to abnormal vestibular stimuli during the critical period is associated with irregular motor development. Children with hypofunctioning vestibular receptors frequently have delayed motor development. The results of the studies done on ferrets and rats reinforced the idea that the vestibular system is very important to motor development during the initial neonatal period. If the vestibular receptors are present during the initial six months to a year when the infant is learning to sit and stand, then the child may develop motor control and balance normally.[28]
The vestibulo-ocular reflex (VOR) is a reflex eye movement that stabilizes images on the retina during head movement. It produces an eye movement in the direction opposite to head movement, thus preserving the image on the center of the visual field. Studies in fish and amphibians revealed a sensitivity in their VOR. They launched into space flight for 9-10, some with developing VOR's and others with already developed reflexes. The fish with developing reflexes developed an upward bend in their tails. The altered gravity resulted in a shift of orientation. Those who were already matured with the reflex were insensitive to the microgravity exposure.[29]
Memory
Recent studies also support the possibility of a critical period for the development of neurons that mediate memory processing. Experimental evidence supports that notion that young neurons in the adult dentate gyrus have a critical period (about 1–3 weeks after neuronal birth) during which they are integral to memory formation.[30] Although the exact reasoning behind this observation is uncertain, studies suggest that the functional properties of neurons at this age make them most appropriate for this purpose; these neurons: (1) Remain hyperactive during the formation of memories; (2) are more excitable; and (3) More easily depolarizable due to GABAergic effects. It is also possible that hyperplasticity makes the neurons more useful in memory formation. If these young neurons had more plasticity than adult neurons in the same context, they could be more influential in smaller numbers.[30] The role of these neurons in the adult dentate gyrus in memory processing is further supported by the fact that behavioral experiments have shown that an intact dentate gyrus is integral to hippocampal memory formation.[30] It is speculated that the dentate gyrus acts a relay station for information relating to memory storage. The likelihood of a critical period could change the way we view memory processing because it would ultimately mean that the collection of neurons present is constantly being replenished as new neurons replace old ones. If a critical period does indeed exist, this could possibly mean that: (1) Diverse populations of neurons that represent events occurring soon after one another may connect those events temporally in the memory formation and processing; OR (2) These different populations of neurons may distinguish between similar events, independent of temporal position; OR (3) Separate populations may mediate the formation of new memories when the same events occur frequently.[30]
See also
- Behavioral cusp
- Child development
- Critical period hypothesis
- Developmental psychology
- Malleable intelligence
- Universal grammar
External links
References
- 1 2 Robson, Ann L. "Critical/Sensitive Periods." Child Development. Ed. Neil J. Salkind. New York: Macmillan Reference USA, 2002. 101-103. Gale Virtual Reference Library. Web. 10 Dec. 2014.
- ↑ Brainard, M. S., & Knudsen, E. I. (1998). Sensitive periods for visual calibration of the auditory space map in the barn owl optic tectum. The Journal of Neuroscience, 18(10), 3929-3942.
- ↑ Hensch, T. K. (2004). "Critical period regulation". J Neurosci. 27: 549–579. doi:10.1146/annurev.neuro.27.070203.144327.
- 1 2 3 Kral A (2013). "Auditory critical periods: A review from system's perspective". Neuroscience. 247: 117–133. doi:10.1016/j.neuroscience.2013.05.021. PMID 23707979.
- 1 2 3 Siegler, Robert (2006). How Children Develop, Exploring Child Develop Student Media Tool Kit & Scientific American Reader to Accompany How Children Develop. New York: Worth Publishers. ISBN 0-7167-6113-0.
- ↑ Curtiss, S. (1977). Genie: a psycholinguistic study of a modern-day wild child. New York: Academic Press.
- ↑ Newport, E. L. (1990). "Maturational constraints on language learning". Cognitive science. 14 (1): 11–28. doi:10.1207/s15516709cog1401_2.
- ↑ Mayberry, R. I.; Lock, E.; Kazmi, H. (2002). "Development: Linguistic ability and early language exposure". Nature. 417 (6884): 38–38. Bibcode:2002Natur.417...38M. doi:10.1038/417038a.
- ↑ Johnson, Eric. "First-Language Acquisition." Encyclopedia of Bilingual Education. Ed. Josué M. González. Vol. 1. Thousand Oaks, CA: SAGE Publications, 2008. 299-304. Gale Virtual Reference Library. Web. 22 Oct. 2014.
- ↑ Pinker, Steven (1994). The Language Instinct. New York: Morrow.
- 1 2 Jia, Li. "Learning a Language, Best Age." Encyclopedia of Bilingual Education. Ed. Josué M. González. Vol. 1. Thousand Oaks, CA: SAGE Publications, 2008. 520-523. Gale Virtual Reference Library. Web. 20 Oct. 2014.
- ↑ Snow, C.E.; Hoefnagel-Höhle, M. (December 1978). "The Critical Period for Language Acquisition: Evidence from Second-Language Learning". Child Development. 49 (4): 1114–1128. doi:10.1111/j.1467-8624.1978.tb04080.x. JSTOR 1128751.
- ↑ Wiesel, TN, Hubel DH (1963). "Effects of visual deprivation on morphology and physiology of cell in the cat's lateral geniculate body". Journal of Neurophysiology. 26 (6): 978–993. PMID 14084170.
- ↑ Experiment Module: Effects of Visual Deprivation During the Critical Period for Development of Vision. McGill University, The Brain from Top to Bottom
- ↑ Antonini A, Stryker MP (June 1993). "Rapid Remodeling of Axonal Arbors in the Visual Cortex". Science. 260 (5115): 1819–21. Bibcode:1993Sci...260.1819A. doi:10.1126/science.8511592. JSTOR 2881379.
- ↑ Higley, M. J.; Strittmatter, S. M. (2010). "Lynx for Braking Plasticity". Science. 330 (6008): 1189–1190. Bibcode:2010Sci...330.1189H. doi:10.1126/science.1198983. PMC 3244692
 . PMID 21109660.
. PMID 21109660. - ↑ Kisilevsky BS, Sylvia MJ, Xing X, Hefeung H, Hai HY, Ke Z, Zengping W (2003). "Effects of experience on fetal voice recognition". Psychological Science. 14 (3): 220–4. doi:10.1111/1467-9280.02435. PMID 12741744.
- ↑ Mertz, Leslie A. "Imprinting and Establishment of Ethology". Gale Virtual Reference Library. Neil Schlager and Josh Lauer. Retrieved 20 October 2014.
- ↑ Hollar, David Wason, Jr. "Imprinting". Salem Health: Psychology & Mental Health. 3: 980–984.
- ↑ Stone, Sherril M. "Imprinting". Gale Virtual Reference Library. SAGE Reference. Retrieved 20 October 2014.
- ↑ Sharma A, Dorman MF, Spahr AJ (2002). "A sensitive period for the development of the central auditory system in children with cochlear implants: implications for age of implantation.". Ear Hear. 23 (6): 532–29. doi:10.1097/00003446-200212000-00004.
- ↑ Kral A, Harmann R, Tillein J, Heid S, Klinke R (2002). "Hearing after congenital deafness: central auditory plasticity in deafness". Cerebral Cortex. 12 (8): 797–807. doi:10.1093/cercor/12.8.797. PMID 12122028.
- ↑ Kral A, Sharma A (2012). "Developmental neuroplasticity after cochlear implantation.". Trends Neuroscience. 35 (2): 111–122. doi:10.1016/j.tins.2011.09.004. PMC 3561718
 . PMID 22104561.
. PMID 22104561. - ↑ Nakahara; et al. (2004). "Specialization of primary auditory cortex processing by sound exposure in the "critical period"". PNAS. 101 (18): 7170–7174. Bibcode:2004PNAS..101.7170N. doi:10.1073/pnas.0401196101.
- ↑ Barkat TR, Polley DB, Hensch TK (September 2011). "A critical period for auditory thalamocortical connectivity". Nature Neuroscience. 14 (9): 1189–96. doi:10.1038/nn.2882. PMC 3419581
 . PMID 21804538.
. PMID 21804538. - ↑ Zhou X, Merzenich MM (1993). "Enduring Effects of early structured noise exposure on temporal modulation in the primary auditory cortex" Check
|url=value (help). Proceedings of the National Academy of Sciences. 105 (11): 4423–8. Bibcode:2008PNAS..105.4423Z. doi:10.1073/pnas.0800009105. - ↑ Eugéne D, Deforges S, Vibert N, Vidal PP (2009). "Vestibular Critical Period, Maturation of Central Vestibular Neurons, and Locomotor Control". Basic and Clinical Aspects of Vertigo and Dizziness. 1164 (1): 180–187. doi:10.1111/j.1749-6632.2008.03727.x.
- ↑ Van Cleave, S. "A critical period for the impact of vestibular sensation on ferret motor development". Pub Med. Pub Med Central.
- ↑ Einstein-Allee, Albert. ""Critical periods" in vestibular development or adaptation of gravity sensory systems to altered gravitational conditions?". PubMed.giv. University of Ulm.
- 1 2 3 4 Aasebø, IEJ; Blankvoort, Stefan; Tashiro, Ayumu (2011). "Critical maturational period of new neuron in adult dentate gyrus for their involvement in memory formation". European Journal of Neuroscience. 33 (6): 1094–1100. doi:10.1111/j.1460-9568.2011.07608.x. PMID 21395853.