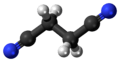Succinonitrile
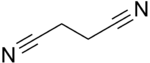 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Butanedinitrile[1] | |||
Other names
| |||
| Identifiers | |||
| 110-61-2 | |||
| 3D model (Jmol) | Interactive image | ||
| 1098380 | |||
| ChemSpider | 21106481 | ||
| ECHA InfoCard | 100.003.441 | ||
| EC Number | 203-783-9 | ||
| MeSH | succinonitrile | ||
| PubChem | 8062 | ||
| RTECS number | WN3850000 | ||
| UNII | 1R479O92DO | ||
| |||
| |||
| Properties | |||
| C4H4N2 | |||
| Molar mass | 80.09 g·mol−1 | ||
| Appearance | Colorless, waxy crystals | ||
| Odor | odorless[2] | ||
| Density | 985 mg mL−1 | ||
| Melting point | 52 to 62 °C; 125 to 143 °F; 325 to 335 K | ||
| Boiling point | 266.1 °C; 510.9 °F; 539.2 K | ||
| 130 g L−1 | |||
| Vapor pressure | 300 Pa (at 100 °C) | ||
| Thermochemistry | |||
| 145.60 J K−1 mol−1 | |||
| Std molar entropy (S |
191.59 J K−1 mol−1 | ||
| Std enthalpy of formation (ΔfH |
139.3–140.4 kJ mol−1 | ||
| Std enthalpy of combustion (ΔcH |
−2.2848–−2.2860 MJ mol−1 | ||
| Hazards | |||
| GHS pictograms |  | ||
| GHS signal word | WARNING | ||
| H302, H315, H319, H335 | |||
| P261, P305+351+338 | |||
| EU classification (DSD) |
| ||
| R-phrases | R22, R36/37/38 | ||
| S-phrases | S26, S36/37, S45 | ||
| Flash point | 113 °C (235 °F; 386 K) | ||
| Lethal dose or concentration (LD, LC): | |||
| LD50 (median dose) |
450 mg kg−1 (oral, rat) | ||
| US health exposure limits (NIOSH): | |||
| PEL (Permissible) |
none[2] | ||
| REL (Recommended) |
TWA 6 ppm (20 mg/m3)[2] | ||
| IDLH (Immediate danger) |
N.D.[2] | ||
| Related compounds | |||
| Related alkanenitriles |
|||
| Related compounds |
DBNPA | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Succinonitrile, also butanedinitrile, is a nitrile, with the formula of C2H4(CN)2. It is a colorless solid that melts at 57 °C, hence its waxy consistency.
Succinonitrile is produced by the addition of hydrogen cyanide to acrylonitrile:[3]
- CH2=CHCN + HCN → NCCH2CH2CN
Hydrogenation of succinonitrile yields putrescine (1,4-diaminobutane).
References
- ↑ "succinonitrile - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 26 March 2005. Identification. Retrieved 14 June 2012.
- 1 2 3 4 "NIOSH Pocket Guide to Chemical Hazards #0573". National Institute for Occupational Safety and Health (NIOSH).
- ↑ "Nitriles". Ullmann's Encyclopedia of Industrial Chemistry (7th ed.). Retrieved 2007-09-10.
External links
This article is issued from Wikipedia - version of the 10/25/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.