Lithium triethylborohydride
 | |
| Names | |
|---|---|
| IUPAC name
Lithium triethylborohydride | |
| Other names
Superhydride LiTEBH | |
| Identifiers | |
| 22560-16-3 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 2006168 |
| ECHA InfoCard | 100.040.963 |
| PubChem | 2723993 |
| |
| |
| Properties | |
| Li(C2H5)3BH | |
| Molar mass | 105.95 g/mol |
| Appearance | Colorless to yellow liquid |
| Density | 0.890 g/cm3, liquid |
| Boiling point | 66 °C (151 °F; 339 K) for THF |
| reactive | |
| Hazards | |
| Main hazards | highly flammable corrosive Causes burns Probable Carcinogen |
| Safety data sheet | External MSDS |
| R-phrases | 11-14/15-19-34 |
| S-phrases | 16-26-33-36/37/39-43-45 |
| NFPA 704 | |
| Related compounds | |
| Related hydride |
sodium borohydride sodium hydride lithium aluminium hydride |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Lithium triethylborohydride is the organoboron compound with the formula LiEt3BH). Commonly referred to as LiTEBH or Superhydride, it is a powerful reducing agent used in organometallic and organic chemistry. LiBHEt3 is far more reducing than lithium borohydride and lithium aluminium hydride. It is a colorless or white solid but is typically marketed as a THF solution.[1] The related reducing agent, sodium triethylborohydride, is commercially available as toluene solutions.
Preparation
LiBHEt3 is prepared by the reaction of lithium hydride (LiH) and triethylborane (Et3B) in tetrahydrofuran (THF):
- LiH + Et3B → LiEt3BH
Its THF solutions are stable indefinitely in the absence of moisture and air.
Uses
Carbonyl substrates
LiBHEt3 reduces a wide range of functional groups, not only aldehydes, ketones, acid chlorides, but also esters and even tertiary amides. The immediate product of these reductions are lithium alkoxides, which, upon hydrolytic workup, are converted to the alcohols. The stoichiometry for the reduction of ketones is shown:
- R2CO + LiBHEt3 → R2CHOLi + BEt3
- R2CHOLi + H2O → R2CHOH + LiOH
Even sterically hindered substrates are reduced, as shown with 2,2,4,4-tetramethyl-3-pentanone.
LiBHEt3 deprotonates carboxylic acids, but does not reduce the resulting lithium carboxylates. For similar reasons, it reduces acid anhydrides to alcohols and the carboxylic acid, not to the diol. Similarly lactones reduce to diols. α,β-Enones undergo 1,4-addition to give lithium enolates. Disulfides reduce to thiols (via thiolates).
Other substrates
Alkyl halides are reduced to the alkanes by LiBHEt3.[2][1]
Epoxides undergo ring-opening upon treatment with LiBHEt3 to give the alcohol. With unsymmetrical epoxides, the reaction can proceed with high regio- and stereo- selectivity, favoring the Markovnikov alcohol.[2] For example, the 1,2-epoxybutane is reduced to 1-methylcyclohexanol:
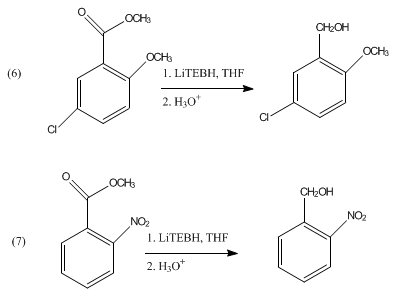
Acetals and ketals are not reduced by LiBHEt3. It can be used in the reductive cleavage of mesylates and tosylates.[3] LiBHEt3 can selectively deprotect tertiary N-acyl groups without affecting secondary amide functionality.[4] It has also shown to reduce aromatic esters to the corresponding alcohols as shown in eq 6 and 7.
LiBHEt3 also reduces pyridine and isoquinolines to piperidines and tetrahydroisoquinolines respectively.[5]
The reduction of β-hydroxysulfinyl imines with catecholborane and LiBHEt3 produces anti-1,3-amino alcohols shown in (8).[6]
Precautions
LiBHEt3 reacts exothermically, potentially violently, with water, alcohols, or acids, releasing hydrogen and the pyrophoric triethylborane.[1]
References
- 1 2 3 Marek Zaidlewicz, Herbert C. Brown "Lithium Triethylborohydride" Encyclopedia of Reagents for Organic Synthesis 2001, John Wiley & Sons. doi:10.1002/047084289X.rl148
- ↑ Brown, H.C.; Kim, S.C.; Krishnamurthy, S. "Selective reductions. 27. Reaction of alkyl halides with representative complex metal hydrides and metal hydrides. Comparison of various hydride reducing agents" J. Org. Chem. 1980, 45, 1-12. doi:10.1021/jo01293a018
- ↑ Baer, H.H.; Mekarska-Falicki, M. Can. J. Chem. 1985, 63, 3043.
- ↑ Tanaka, H.; Ogasawara, K. Tetrahedron Lett. 2002, 43, 4417.
- ↑ Blough, B.E.; Carroll, F. I. "Reduction of isoquinoline and pyridine-containing heterocycles with lithium triethylborohydride (Super-Hydride®)" Tetrahedron Lett. 1993, 34, 7239. doi:10.1016/S0040-4039(00)79297-5
- ↑ Kochi, T; Tang, T.P.; Ellman, J.A. J. Am. Chem. Soc. 2002, 124, 6518.