WRKY transcription factor family
WRKY transcription factors are key regulators of many processes in plants. These include the responses to biotic and abiotic stresses, senescence, seed dormancy and seed germination and some developmental processes.[1] Like many transcription factors, WRKY transcription factors are defined by the presence of a DNA-binding domain, in this case called the WRKY domain (WRKY protein domain). The WRKY domain was named in 1996 after the almost invariant WRKY amino acid sequence at the N-terminus and is about 60 residues in length. In addition to containing the ‘WRKY signature’ WRKY domains also possess an atypical zinc-finger structure at the C-terminus (either Cx4-5Cx22-23HxH or Cx7Cx23HxC). Most WRKY transcription factors bind to the W-box promoter element that has a consensus sequence of TTGACC/T.
The History of WRKY Transcription Factors
In 1994 and 1995, the first two reports of WRKY transcription factors appeared. They described newly discovered but as yet ill-defined DNA binding proteins that played potential roles in the regulation of gene expression by sucrose (SPF1) [2] or during germination (ABF1 and ABF2).[3] A third report appeared in 1996 that identified WRKY1, WRKY2 and WRKY3 from parsley. The authors named the new transcription factor family the WRKY family (pronounced ‘worky’) after a conserved amino acid sequence at the N-terminus of the DNA-binding domain.[4] The parsley WRKY proteins also provided the first evidence that WRKY transcription factors play roles in regulating plant responses to pathogens. Numerous papers have now shown this to be a major function of WRKY transcription factors. Since these initial publications, it has become clear that the WRKY family is among the ten largest families of transcription factors in higher plants and that these transcription factors play key roles in regulating a number of plant processes including the responses to biotic and abiotic stresses, germination, senescence, and some developmental processes.[5]
The Structure of the WRKY Domain

As soon as the WRKY domain was characterized, it was suggested that it contained a novel zinc finger structure and the first evidence to support this came from studies with 2-phenanthroline that chelates zinc ions. Addition of 2-phenenthroline to gel retardation assays that contained E. coli expressed WRKY proteins resulted in a loss of binding to the W box target sequence. The other suggestion was that the WRKY signature amino acid sequence at the N-terminus of the WRKY domain directly binds to the W box sequence in the DNA of target promoters. These suggestions were shown to be correct by publication of the solution structure of the C-terminal WRKY domain of the Arabidopsis WRKY4 protein. The WRKY domain was found to form a four-stranded β-sheet.[6] Soon afterwards, a crystal structure of the C-terminal WRKY domain of the Arabidopsis WRKY1 protein was reported. This showed a similar result to the solution structure except that it may contain an additional β-strand at the N-terminus of the domain.[7] From these two studies it appears that the conserved WRKYGQK signature amino acid sequence enters the major groove of the DNA to bind to the W Box. Recently, the first structural determination of the WRKY domain complexed with a W Box was reported. The NMR solution structure of the WRKY DNA-binding domain of Arabidopsis WRKY4 in complex with W Box DNA revealed that part of a four-stranded β-sheet enters the major groove of DNA in an atypical mode that the authors named the β-wedge, where this sheet is almost perpendicular to the DNA helical axis. As initially predicted, amino acids in the conserved WRKYGQK signature motif contact the W Box DNA bases mainly through extensive apolar contacts with thymine methyl groups. These structural data explain the conservation of both the WRKY signature sequence at the N-terminus of the WRKY domain and the conserved cysteine and histidine residues. It also provides the molecular basis for the previously noted remarkable conservation of both the WRKY amino acid signature sequence and the W Box DNA sequence.[8]
The evolution of WRKY transcription factors
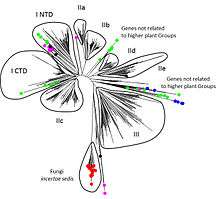
WRKY transcription factor genes are found throughout the plant lineage and also outside of the plant lineage in some diplomonads, social amoebae, fungi incertae sedis, and amoebozoa.[9] This patchy distribution suggests that lateral gene transfer is responsible. These lateral gene transfer events appear to pre-date the formation of the WRKY groups in flowering plants, where there are seven well-defined groups, Groups I + IIc, Groups IIa + IIb, Groups IId + IIe, and Group III. Flowering plants also contain proteins with domains typical for both resistance (R) proteins and WRKY transcription factors. R protein-WRKY genes have evolved numerous times in flowering plants, each type being restricted to specific flowering plant lineages. These chimeric proteins contain not only novel combinations of protein domains but also novel combinations and numbers of WRKY domains.
External Links and Useful Resources
- WRKY Transcription Factor Family at The Arabidopsis Information Resource
- The Rushton Lab
- The Somssich Lab
- The Shen Lab
- Somssich’s list of WRKY-related publications
- The Eulgem Lab
- WRKY Wide Web
References
- ↑ Rushton PJ, Somssich IE, Ringler P, Shen QJ: WRKY transcription factors. Trends in plant science 2010, 15(5):247-258
- ↑ Ishiguro, S. and Nakamura, K. (1994) Characterization of a cDNA encoding a novel DNA-binding protein, SPF1, that recognizes SP8 sequences in the 50 upstream regions of genes coding for sporamin and beta-amylase from sweet potato. Mol. Gen. Genet. 244, 563–571
- ↑ Rushton, P.J. et al. (1995) Members of a new family of DNA-binding proteins bind to a conserved cis-element in the promoters of alpha- Amy2 genes. Plant Mol. Biol. 29, 691–702
- ↑ Rushton, P.J., Torres, J.T., Parniske, M., Wernert, P., Hahlbrock, K. and Somssich, I.E. (1996) Interaction of elicitor-induced DNA-binding proteins with elicitor response elements in the promoters of parsley PR1 genes. EMBO J. 15, 5690–5700
- ↑ Eulgem, T. and Somssich, I.E. (2007) Networks of WRKY transcription factors in defense signaling. Curr. Opin. Plant Biol. 10, 366–371, Ulker, B. and Somssich, I.E. (2004) WRKY transcription factors: from DNA binding towards biological function. Curr. Opin. Plant Biol. 7, 491–498
- ↑ Yamasaki K, Kigawa T, Watanabe S, Inoue M, Yamasaki T, Seki M, Shinozaki K and Yokoyama S (2012) Structural basis for sequence-specific DNA recognition by an Arabidopsis WRKY transcription factor. J Biol Chem 287: 7683–7691
- ↑ Duan MR, Nan J, Liang YH, Mao P, Lu L, Li L, Wei C, Lai L, Li Y and Su XD (2007) DNA binding mechanism revealed by high resolution crystal structure of Arabidopsis thaliana WRKY1 protein. Nucleic Acid Res.35:1145-1154
- ↑ Eulgem T, Rushton PJ, Robatzek S and Somssich IE (2000) The WRKY superfamily of plant transcription factors. Trends Plant Sci 5:199-206
- ↑ Charles I. Rinerson, Roel C. Rabara, Prateek Tripathi Qingxi J Shen, and Paul J. Rushton (2015) Structure and evolution of WRKY transcription factors. In Plant Transcription Factors: Evolutionary, Structural and Functional Aspects Edited by: Daniel H. Gonzalez. Elsevier