1,3,5-Triazine
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1,3,5-Triazine | |||
| Other names
sym-Triazine s-Triazine Cyanidine Hydrogen cyanide trimer Vedita | |||
| Identifiers | |||
| 290-87-9 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:30259 | ||
| ChEMBL | ChEMBL15698 | ||
| ChemSpider | 8905 | ||
| ECHA InfoCard | 100.005.481 | ||
| RTECS number | XY2957000 | ||
| |||
| |||
| Properties | |||
| C3H3N3 | |||
| Molar mass | 81.08 g/mol | ||
| Appearance | White crystalline solid | ||
| Melting point | 81 to 83 °C (178 to 181 °F; 354 to 356 K) | ||
| Structure | |||
| planar | |||
| zero | |||
| Hazards | |||
| Main hazards | Sensitive to water | ||
| Related compounds | |||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
1,3,5-triazine, also called s-triazine, is an organic chemical compound with the formula (HCN)3. It is a six-membered heterocyclic aromatic ring, one of several isomeric triazines. S-triazine and its derivatives are useful in a variety of applications.
Preparation
Symmetrical 1,3,5-triazines are prepared by trimerization of certain nitriles such as cyanogen chloride or cyanimide. Benzoguanamine (with one phenyl and 2 amino substituents) is synthesised from benzonitrile and dicyandiamide.[1] In the Pinner triazine synthesis (named after Adolf Pinner )[2] the reactants are an alkyl or aryl amidine and phosgene [3][4] Insertion of N-H moiety into hydrazide by a copper carbenoid, followed by treatment with ammonium chloride also gives the triazine core.[5]
Applications
As a reagent in organic synthesis, s-triazine is used as the equivalent of hydrogen cyanide (HCN). Being a solid (vs a gas for HCN), triazine is sometimes easier to handle in the laboratory. One application is in the Gattermann reaction, used to attach the formyl group to aromatic substrates.[6]
Triazine derivatives
N- and C-substituted triazines are used industrially. The most common derivative of 1,3,5-triazine is 2,4,6-triamino-1,3,5-triazine, commonly known as melamine or cyanuramide. Another important derivative is 2,4,6-trihydroxy-1,3,5-triazine better known as cyanuric acid.
Trichloro-1,3,5-triazine (cyanuric chloride) is the starting point for the manufacture of many herbicides such as Simazine and atrazine. Chlorinated triazines are the basis of an important family of reactive dyes, which are covalently attached to cellulosic materials.[7]
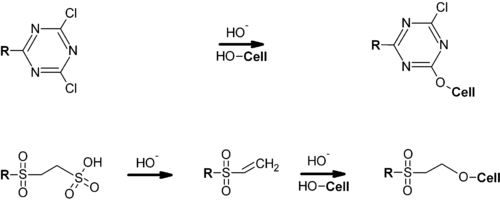
Triazines are also found in pharmaceutical products.[8]
References
- ↑ Benzoguanamine J. K. Simons and M. R. Saxton Organic Syntheses Coll. Vol. 4, p.78; Vol. 33, p.13 Article
- ↑ A. Pinner, Ber. 23, 2919 (1890)
- ↑ Name reactions and reagents in organic synthesis, Bradford P. Mundy, Michael G. Ellerd, Frank G. Favaloro
- ↑ Triazines. XIV. The Extension of the Pinner Synthesis of Monohydroxy-s-triazines to the Aliphatic Series. 2,4-Dimethyl-s-triazine1-3 Hansjuergen Schroeder, Christoph Grundmann J. Am. Chem. Soc., 1956, 78 (11), pp 2447–2451 doi:10.1021/ja01592a028
- ↑ Shi, B.; Lewis, W.; Campbell, I. B.; Moody, C. J. Org. Lett., 2009, 3686-3688 doi:10.1021/ol901502u
- ↑ Roswitha M. Böhme, Qun Dang "1,3,5-Triazine" in Encyclopedia of Reagents for Organic Synthesis 2008 John Wiley & Sons. doi:10.1002/047084289X.rt158.pub2
- ↑ Horst Tappe, Walter Helmling, Peter Mischke, Karl Rebsamen, Uwe Reiher, Werner Russ, Ludwig Schläfer and Petra Vermehren "Reactive Dyes"in Ullmann's Encyclopedia of Industrial Chemistry 2000, Wiley-VCH, Weinheim. doi:10.1002/14356007.a22_651
- ↑ Aksenov A. V., Aksenova, I. V. "Use of the ring opening reactions of 1,3,5-triazines in organic synthesis" Chemistry of Heterocyclic Cmpds. 45, pp 130-150 (2009). doi:10.1007/s10593-009-0243-5