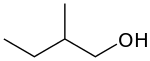
2-Methyl-1-butanol
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylbutan-1-ol | |
| Other names
2-Methyl-1-butanol Active amyl alcohol | |
| Identifiers | |
| 137-32-6 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:48945 |
| ChEMBL | ChEMBL451923 |
| ChemSpider | 8398 |
| ECHA InfoCard | 100.004.809 |
| PubChem | 8723 |
| UNII | 7VTJ239ASU |
| |
| |
| Properties | |
| C5H12O | |
| Molar mass | 88.148 g/mol |
| Appearance | colorless liquid |
| Density | 0.8152 g/cm3 |
| Melting point | −117.2 °C (−179.0 °F; 156.0 K) |
| Boiling point | 127.5 °C (261.5 °F; 400.6 K) |
| 31 g/L | |
| Solubility | miscible with ethanol, diethyl ether; very soluble in acetone |
| Vapor pressure | 3 mm Hg |
| Viscosity | 4.453 mPa·s |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH |
-356.6 kJ·mol−1 (liquid) -301.4 kJ·mol−1 (gas) |
| Hazards | |
| 385 °C (725 °F; 658 K) | |
| Related compounds | |
| Related compounds |
Amyl alcohol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
2-Methyl-1-butanol (IUPAC name, also called active amyl alcohol) is an organic chemical compound.
It is one of the components of the aroma of Tuber melanosporum, the black truffle.
Uses
It is used as a solvent and an intermediate in the manufacture of other chemicals. 2-Methyl-1-butanol is a component of many mixtures of amyl alcohols sold industrially.
Reactions
2-Methyl-1-butanol can be derived from fusel oil (because it occurs naturally in fruits such as grapes[3]) or manufactured by either the oxo process or via the halogenation of pentane.[2]
See also
References
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, FL: CRC Press, pp. 3–374, 5–42, 6–188, 8–102, 16–22, ISBN 0-8493-0594-2
- 1 2 McKetta, John J.; Cunningham, William Aaron (1977), Encyclopedia of Chemical Processing and Design, 3, Boca Raton, FL: CRC Press, pp. 279–280, ISBN 978-0-8247-2480-1, retrieved 2009-12-14
- ↑ Howard, Philip H. (1993), Handbook of Environmental Fate and Exposure Data for Organic Chemicals, 4, Boca Raton, FL: CRC Press, pp. 392–396, ISBN 978-0-87371-413-6, retrieved 2009-12-14
This article is issued from Wikipedia - version of the 9/23/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.