Benzenediazonium chloride
 | |
 | |
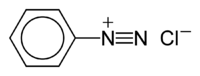
 The benzenediazonium ion | |
| Names | |
|---|---|
| IUPAC name
Benzenediazonium chloride | |
| Other names
Phenyldiazonium chloride | |
| Identifiers | |
| 100-34-5 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 54953 |
| ECHA InfoCard | 100.002.584 |
| PubChem | 60992 |
| UNII | 3U50A1GI4F |
| |
| |
| Properties | |
| C6H5ClN2 | |
| Molar mass | 140.57 g·mol−1 |
| Appearance | colorless crystals |
| Melting point | decomposes |
| Boiling point | decomposes |
| very good, hydroscopic | |
| Hazards | |
| Main hazards | unstable, possibly explosive |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Benzenediazonium chloride is an organic compound with the formula [C6H5N2]Cl. It is a salt of a diazonium cation and chloride. It exists as a colourless solid that is soluble in polar solvents including water. It is the parent member of the aryldiazonium compounds,[1] which are widely used in organic chemistry. Because the salt is unstable, it is not commercially available but is prepared upon demand.
Synthesis
This compound is prepared by diazotization of aniline.[2] The conversion involves in situ production of nitrous acid (HNO2), which reacts with the aniline:
- C6H5NH2 + HNO2 + HCl → [C6H5N2]Cl + 2 H2O
The reactions are conducted at low temperature to minimize decomposition of the diazonium salt.
Diazonium chloride can also be prepared by treating nitrite esters with aniline in presence of HCl. Nitrite esters are formed from alcohol and nitrous acid.[3]
- C5H11ONO + HCl + C6H5NH2 → [C6H5N2]Cl + C5H11OH + H2O
Chemical properties
The diazo group (N2) can be replaced by many other groups, usually anions, giving a variety of substituted phenyl derivatives:
- C6H5N2+ + Nu− → C6H5Nu + N2
These transformations are associated with many named reactions including the Schiemann reaction, Sandmeyer reaction, and Gomberg-Bachmann reaction. A wide range of groups that can be used to replace N2 including halide, SH−, CO2H−, OH−. Of considerable practical value in the dye industry are the diazo coupling reactions.
Safety
The compound is explosive.[4]
References
- ↑ March, J. (1992). Advanced Organic Chemistry (4th ed.). New York: J. Wiley and Sons. ISBN 0-471-60180-2.
- ↑ Flood, D. T. (1933). "Fluorobenzene". Org. Synth. 13: 46.; Coll. Vol., 2, p. 295 The procedure described the tetrafluoroborate salt of phenyldiazonium.
- ↑ Jain, S. K. (2009). Conceptual Chemistry for class XII. New Delhi: S. Chand & Company. pp. 1179–1183. ISBN 81-219-1623-2.
- ↑ Nesmajanow, A. N. (1932). "β-Naphthylmercuric chloride". Org. Synth. 12: 54.; Coll. Vol., 2, p. 432