Bischler–Möhlau indole synthesis
| Bischler-Möhlau indole synthesis | |
|---|---|
| Named after | August Bischler Richard Möhlau |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000523 |
The Bischler–Möhlau indole synthesis is a chemical reaction that forms a 2-aryl-indole from an α-bromo-acetophenone and excess aniline; it is named after August Bischler and Richard Möhlau [1][2][3][4][5]
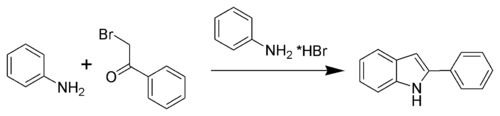
In spite of its long history, this classical reaction has received relatively little attention in comparison with other methods for indole synthesis, perhaps owing to the harsh reaction conditions that it requires. Recently, milder methods have been developed, including the use of lithium bromide as a catalyst and an improved procedure involving the use of microwave irradiation.[6][7][8]
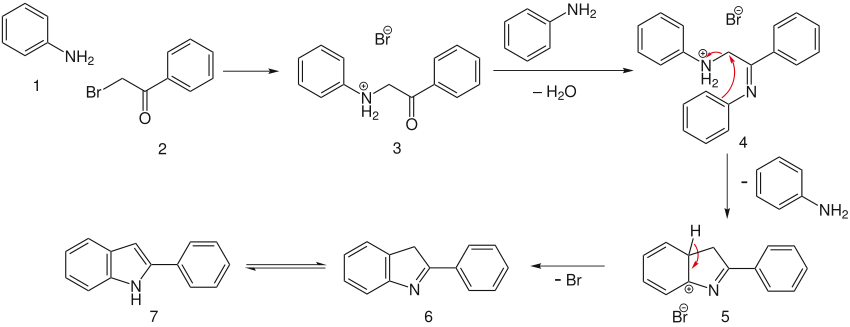
Reaction mechanism
The first two step involve the reaction of the α-bromo-acetophenone with molecules of aniline to form intermediate 4. The charged aniline forms a decent enough leaving group for an electrophilic cyclization to form intermediate 5, which quickly aromatizes and tautomerizes to give the desired indole 7.
See also
References
- ↑ Bischler, Aug. (1892). "Ueber die Entstehung einiger substituirter Indole". Berichte der deutschen chemischen Gesellschaft. 25 (2): 2860–2879. doi:10.1002/cber.189202502123. ISSN 0365-9496.
- ↑ Bischler, Aug.; Fireman, P. (1893). "Zur Kenntniss einiger α-β- Diphenylindole". Berichte der deutschen chemischen Gesellschaft. 26 (2): 1336–1349. doi:10.1002/cber.18930260232. ISSN 0365-9496.
- ↑ Möhlau, R. (1881). "Ueber die Einwirkung primärer aromatischer Aminbasen auf Acetophenonbromid". Chemische Berichte. 14: 171. doi:10.1002/cber.18810140146.
- ↑ Möhlau, R. (1882). "Ueber Diphenyldiisoindol". Chemische Berichte. 15 (2): 2480. doi:10.1002/cber.188201502204.
- ↑ Fischer, Emil Hermann; Schmitt, T. (1888). "Ueber Pr-2-Phenylindol". Chemische Berichte. 21: 1071. doi:10.1002/cber.188802101200.
- ↑ Pchalek, K.; Jones, A. W.; Wekking, M. M. T.; Black, D. S. C. (2005). "Synthesis of activated 3-substituted indoles: An optimised one-pot procedure". Tetrahedron. 61: 77. doi:10.1016/j.tet.2004.10.060.
- ↑ Sridharan, V.; Perumal, S.; Avendaño, C.; Menéndez, J. C. (2006). "Microwave-Assisted, Solvent-Free Bischler Indole Synthesis". Synlett: 91. doi:10.1055/s-2005-922760.
- ↑ Vara, Yosu; Aldaba, Eneko; Arrieta, Ana; Pizarro, José L.; Arriortua, María I.; Cossío, Fernando P. (2008). "Regiochemistry of the microwave-assisted reaction between aromatic amines and α-bromoketones to yield substituted 1H-indoles". Organic & Biomolecular Chemistry. 6 (10): 1763. doi:10.1039/B719641E.