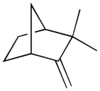
Camphene
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2,2-dimethyl-3-methylene-bicyclo[2.2.1]heptane | |||
| Identifiers | |||
| 79-92-5 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:3830 | ||
| ChemSpider | 6364 | ||
| ECHA InfoCard | 100.001.123 | ||
| EC Number | 201-234-8 | ||
| KEGG | C06076 | ||
| PubChem | 6616 | ||
| UNII | G3VG94Z26E | ||
| |||
| |||
| Properties | |||
| C10H16 | |||
| Molar mass | 136.24 g·mol−1 | ||
| Appearance | Crystalline solid[3] | ||
| Density | 0.842 g/cm3[3] | ||
| Melting point | 51 to 52 °C (124 to 126 °F; 324 to 325 K)[3] | ||
| Boiling point | 159 °C (318 °F; 432 K)[3] | ||
| Practically insoluble[3] | |||
| Hazards | |||
| R-phrases | R11 R36/37/38 R50/53 | ||
| S-phrases | S16 S26 S29 S37/39 S57 | ||
| Flash point | 40 °C (104 °F; 313 K) | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Camphene is a bicyclic monoterpene. It is nearly insoluble in water, but very soluble in common organic solvents. It volatilizes readily at room temperature and has a pungent smell. It is a minor constituent of many essential oils such as turpentine, cypress oil, camphor oil, citronella oil, neroli, ginger oil, and valerian. It is produced industrially by catalytic isomerization of the more common alpha-pinene. Camphene is used in the preparation of fragrances and as a food additive for flavoring. Its mid-19th century use as a fuel for lamps was limited by its explosiveness.
References
- ↑ IUCLID Datasheet
- ↑ Fisher Scientific MSDS
- 1 2 3 4 5 Merck Index, 11th Edition, 1736
This article is issued from Wikipedia - version of the 6/23/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.