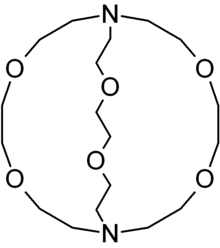
Cryptand

Cryptands are a family of synthetic bi- and polycyclic multidentate ligands for a variety of cations.[2] The Nobel Prize for Chemistry in 1987 was given to Donald J. Cram, Jean-Marie Lehn, and Charles J. Pedersen for their efforts in discovering and determining uses of cryptands and crown ethers, thus launching the now flourishing field of supramolecular chemistry.[3] The term cryptand implies that this ligand binds substrates in a crypt, interring the guest as in a burial. These molecules are three-dimensional analogues of crown ethers but are more selective and strong as complexes for the guest ions. The resulting complexes are lipophilic.
Structure
The most common and most important cryptand is N[CH2CH2OCH2CH2OCH2CH2]3N; the formal IUPAC name for this compound is 1,10-diaza-4,7,13,16,21,24-hexaoxabicyclo[8.8.8]hexacosane. This compound is termed [2.2.2]cryptand where the numbers indicate the number of ether oxygen atoms (and hence binding sites) in each of the three bridges between the amine nitrogen "caps". Many cryptands are commercially available under the tradename "Kryptofix."[4] All-amine cryptands exhibit particularly high affinity for alkali metal cations, which has allowed the isolation of salts of K−.[5]
Properties
The 3-dimensional interior cavity of a cryptand provides a binding site – or host – for "guest" ions. The complex between the cationic guest and the cryptand is called a cryptate. Cryptands form complexes with many "hard cations" including NH+
4, lanthanoids, alkali metals, and alkaline earth metals. In contrast to crown ethers, cryptands bind the guest ions using both nitrogen and oxygen donors. This three-dimensional encapsulation mode confers some size-selectivity, enabling discrimination among alkali metal cations (e.g. Na+ vs. K+).
Uses
Cryptands are more expensive and difficult to prepare, but offer much better selectivity and strength of binding[6] than other complexants for alkali metals, such as crown ethers. They are able to bind otherwise insoluble salts into organic solvents. They can also be used as phase transfer catalysts by transferring ions from one phase to another.[7] Cryptands enabled the synthesis of the alkalides and electrides. They have also been used in the crystallization of Zintl ions such as Sn4−
9.
See also
References
- ↑ Alberto, R.; Ortner, K.; Wheatley, N.; Schibli, R.; Schubiger, A. P. (2001). "Synthesis and properties of boranocarbonate: a convenient in situ CO source for the aqueous preparation of [99mTc(OH2)3(CO)3]+". J. Am. Chem. Soc. 121 (13): 3135–3136. doi:10.1021/ja003932b.
- ↑ Von Zelewsky, A. (1995). Stereochemistry of Coordination Compounds. Chichester: John Wiley. ISBN 0-471-95057-2.
- ↑ Lehn, J. M. (1995). Supramolecular Chemistry: Concepts and Perspectives. Weinheim: VCH.
- ↑ 23978-09-8
- ↑ Kim, J.; Ichimura, A. S.; Huang, R. H.; Redko, M.; Phillips, R. C.; Jackson, J. E.; Dye, J. L. (1999). "Crystalline Salts of Na− and K− (Alkalides) that Are Stable at Room Temperature". J. Am. Chem. Soc. 121 (45): 10666–10667. doi:10.1021/ja992667v.
- ↑ Dietrich, B. (1996). "Cryptands". In Gokel, G. W. Comprehensive Supramolecular Chemistry. 1. Oxford: Elsevier. p. 153–211. ISBN 0-08-040610-6.
- ↑ Landini, D.; Maia, A.; Montanari, F.; Tundo, P. (1979). "Lipophilic [2.2.2]cryptands as phase-transfer catalysts. Activation and nucleophilicity of anions in aqueous-organic thirteen-phase systems and in organic solvents of low polarity". J. Am. Chem. Soc. 101 (10): 2526–2530. doi:10.1021/ja00504a004.
General reading
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "cryptand".
- Lee, J.D. (1991). Concise Inorganic Chemistry (4th ed.). New York: Chapman & Hall. pp. 306–308 & 353. ISBN 0-412-40290-4.
- Krakowiak, K. E.; Bradshaw, J. S.; An, H.-Y.; Izatt, R. M. (1993). "Simple methods for the preparation of cryptands". Pure Appl. Chem. 65 (3): 511–514. doi:10.1351/pac199365030511.