DNA oxidation
DNA oxidation is the process of oxidative damage on Deoxyribonucleic Acid. It occurs most readily at guanine residues due to the high oxidation potential of this base relative to cytosine, thymine, and adenine. It is widely believed to be linked to certain disease and cancers.
Oxidized bases in DNA
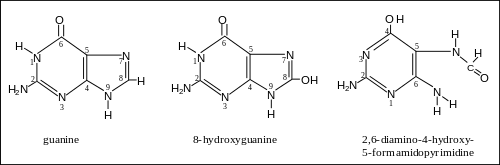
More than 20 oxidatively damaged DNA base lesions were identified in 2003 by Cooke et al.[1] and these overlap the 12 oxidized bases reported in 1992 by Dizdaroglu.[2] Two of the most frequently oxidized bases found by Dizdaroglu after ionizing radiation (causing oxidative stress) were the two oxidation products of guanine shown in the Figure. One of these products was 8-OH-Gua (8-hydroxyguanine). (The article 8-Oxo-2'-deoxyguanosine refers to the same damaged base since the keto form 8-oxo-Gua described there may undergo a tautomeric shift to the enol form 8-OH-Gua shown here.) The other product was FapyGua (2,6-diamino-4-hydroxy-5-formamidopyrimidine). Another frequent oxidation product was 5-OH-Hyd (5-hydroxyhydantoin) derived from cytosine.
Removal of oxidized bases
Most oxidized bases are removed from DNA by enzymes operating within the base excision repair pathway.[1] Removal of oxidized bases in DNA is fairly rapid. For example, 8-oxo-dG was increased 10-fold in the livers of mice subjected to ionizing radiation, but the excess 8-oxo-dG was removed with a half-life of 11 minutes.[3]
Steady-state levels of DNA damages
Steady-state levels of endogenous DNA damages represent the balance between formation and repair. Swenberg et al.[4] measured average amounts of steady state endogenous DNA damages in mammalian cells. The seven most common damages they found are shown in Table 1. Only one directly oxidized base, 8-hydroxyguanine, at about 2,400 8-OH-G per cell, was among the most frequent DNA damages present in the steady-state.
| Endogenous lesions | Number per cell |
|---|---|
| Abasic sites | 30,000 |
| N7-(2-hydroxethyl)guanine (7HEG) | 3,000 |
| 8-hydroxyguanine | 2,400 |
| 7-(2-oxoethyl)guanine | 1,500 |
| Formaldehyde adducts | 960 |
| Acrolein-deoxyguanine | 120 |
| Malondialdehyde-deoxyguanine | 60 |
Increased 8-oxo-dG in carcinogenesis and disease
_and_with_tumorigenesis_(B)._Brown_shows_8-oxo-dG.jpg)
As reviewed by Valavanidis et al.[6] increased levels of 8-oxo-dG in a tissue can serve as a biomarker of oxidative stress. They also noted that increased levels of 8-oxo-dG are frequently found associated with carcinogenesis and disease.
In the figure shown in this section, the colonic epithelium from a mouse on a normal diet has a low level of 8-oxo-dG in its colonic crypts (panel A). However, a mouse likely undergoing colonic tumorigenesis (due to deoxycholate added to its diet[5]) has a high level of 8-oxo-dG in its colonic epithelium (panel B). Deoxycholate increases intracellular production of reactive oxygen resulting in increased oxidative stress,[7][8] and this may contribute to tumorigenesis and carcinogenesis. Of 22 mice fed the diet supplemented with deoxycholate, 20 (91%) developed colonic tumors after 10 months on the diet, and the tumors in 10 of these mice (45% of mice) included an adenocarcinoma (cancer).[5] Cooke et al.[1] point out that a number of diseases, such as Alzheimer's disease and systemic lupus erythematosus, have elevated 8-oxo-dG but no increased carcinogenesis.
Indirect role of oxidative damage in carcinogenesis
Valavanidis et al.[6] pointed out that oxidative DNA damage, such as 8-oxo-dG, may contribute to carcinogenesis by two mechanisms. The first mechanism involves modulation of gene expression, whereas the second is through the induction of mutations.
Epigenetic alterations
Epigenetic alteration, for instance by methylation of CpG islands in a promoter region of a gene, can repress expression of the gene (see DNA methylation in cancer). In general, epigenetic alteration can modulate gene expression. As reviewed by Bernstein and Bernstein,[9] the repair of various types of DNA damages can, with low frequency, leave remnants of the different repair processes and thereby cause epigenetic alterations. 8-oxo-dG is primarily repaired by base excision repair (BER).[10] Li et al.[11] reviewed studies indicating that one or more BER proteins also participate(s) in epigenetic alterations involving DNA methylation, demethylation or reactions coupled to histone modification. Nishida et al.[12] examined 8-oxo-dG levels and also evaluated promoter methylation of 11 tumor suppressor genes (TSGs) in 128 liver biopsy samples. These biopsies were taken from patients with chronic hepatitis C, a condition causing oxidative damages in the liver. Among 5 factors evaluated, only increased levels of 8-oxo-dG was highly correlated with promoter methylation of TSGs (p<0.0001). This promoter methylation could have reduced expression of these tumor suppressor genes and contributed to carcinogenesis.
Mutagenesis
Yasui et al.[13] examined the fate of 8-oxo-dG when this oxidized derivative of deoxyguanosine was inserted into the thymidine kinase gene in a chromosome within human lymphoblastoid cells in culture. They inserted 8-oxo-dG into about 800 cells, and could detect the products that occurred after the insertion of this altered base, as determined from the clones produced after growth of the cells. 8-oxo-dG was restored to G in 86% of the clones, probably reflecting accurate base excision repair or translesion synthesis without mutation. G:C to T:A transversions occurred in 5.9% of the clones, single base deletions in 2.1% and G:C to C:G transversions in 1.2%. Together, these more common mutations totaled 9.2% of the 14% of mutations generated at the site of the 8-oxo-dG insertion. Among the other mutations in the 800 clones analyzed, there were also 3 larger deletions, of sizes 6, 33 and 135 base pairs. Thus 8-oxo-dG, if not repaired, can directly cause frequent mutations, some of which may contribute to carcinogenesis.
RNA Oxidation
RNAs in native milieu are exposed to various insults. Among these threats, oxidative stress is one of the major causes of damage to RNAs. The level of oxidative stress that a cell endures is reflected by the quantity of Reactive oxygen species (ROS). ROS are generated from normal oxygen metabolism in cells and are recognized as a list of active molecules, such as O2•−, 1O2, H2O2 and, •OH .[14] A nucleic acid can be oxidized by ROS through a Fenton reaction.[15] To date, around 20 oxidative lesions have been discovered in DNA.[16] RNAs are likely to be more sensitive to ROS for the following reasons: i) the basically single-stranded structure exposes more sites to ROS; ii) compared with nuclear DNA, RNAs are less compartmentalized; iii) RNAs distribute broadly in cells not only in the nucleus as DNAs do, but also in large portions in the cytoplasm.[17][18] This theory has been supported by a series of discoveries from rat livers, human leukocytes, etc. Actually, monitoring a system by applying the isotopical label [18O]-H2O2 shows greater oxidation in cellular RNA than in DNA. Oxidation randomly damages RNAs, and each attack bring problems to the normal cellular metabolism. Although alteration of genetic information on mRNA is relatively rare, oxidation on mRNAs in vitro and in vivo results in low translation efficiency and aberrant protein products.[19] Though the oxidation strikes the nucleic strands randomly, particular residues are more susceptible to ROS, such hotspot sites being hit by ROS at a high rate. Among all the lesions discovered thus far, one of the most abundant in DNA and RNA is the 8-hydroxyguanine.[20] Moreover, 8-hydroxyguanine is the only one measurable among all the RNA lesions. Besides its abundance, 8-hydroxydeoxyguanosine (8-oxodG) and 8-hydroxyguanosine (8-oxoG) are identified as the most detrimental oxidation lesions for their mutagenic effect,[21] in which this non-canonical counterpart can faultily pair with both adenine and cytosine at the same efficiency.[22][23] This mis-pairing brings about the alteration of genetic information through the synthesis of DNA and RNA. In RNA, oxidation levels are mainly estimated through 8-oxoG-based assays. So far, approaches developed to directly measure 8-oxoG level include HPLC-based analysis and assays employing monoclonal anti-8-oxoG antibody. The HPLC-based method measures 8-oxoG with an electrochemical detector (ECD) and total G with a UV detector.[24] The ratio that results from comparing the two numbers provides the extent that the total G is oxidized. Monoclonal anti-8-oxoG mouse antibody is broadly applied to directly detect this residue on either tissue sections or membrane, offering a more visual way to study its distribution in tissues and in discrete subsets of DNA or RNA. The established indirect techniques are mainly grounded on this lesion’s mutagenic aftermath, such as the lacZ assay.[25] This method was first set up and described by Taddei and was a potentially powerful tool to understand the oxidation situation at both the RNA sequence level and single nucleotide level. Another source of oxidized RNAs is mis-incorporation of oxidized counterpart of single nucleotides. Indeed, the RNA precursor pool size is hundreds of sizes bigger than DNA’s.
Potential factors for RNA quality control
There have been furious debates on whether the issue of RNA quality control does exist. However, with the concern of various length of half life of diverse RNA species ranging from several minutes to hours, degradation of defective RNA can not easily be attributed to its transient character anymore. Indeed, reaction with ROS takes only few minutes, which is even shorter than average life-span of the most unstable RNAs.[17] Adding the fact that stable RNA take the lion’s share of total RNA, RNA error deleting become hypercritical and should not be neglected anymore .This theory is upheld by the fact that level of oxidized RNA decreases after removal the oxidative challenge .[26][27] Some potential facors include ribonucleases, which are suspected to selectively degrade damaged RNAs under stresses. Also enzymes working at RNA precursor pool level,are known to control quality of RNA sequence by changing error precursor to the form that can't be included directly into nascent strand.
References
- 1 2 3 Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003). "Oxidative DNA damage: mechanisms, mutation, and disease". FASEB J. 17 (10): 1195–214. doi:10.1096/fj.02-0752rev. PMID 12832285.
- ↑ Dizdaroglu M (1992). "Oxidative damage to DNA in mammalian chromatin". Mutat. Res. 275 (3-6): 331–42. doi:10.1016/0921-8734(92)90036-o. PMID 1383774.
- ↑ Hamilton ML, Guo Z, Fuller CD, Van Remmen H, Ward WF, Austad SN, Troyer DA, Thompson I, Richardson A (2001). "A reliable assessment of 8-oxo-2-deoxyguanosine levels in nuclear and mitochondrial DNA using the sodium iodide method to isolate DNA". Nucleic Acids Res. 29 (10): 2117–26. doi:10.1093/nar/29.10.2117. PMC 55450
 . PMID 11353081.
. PMID 11353081. - ↑ Swenberg JA, Lu K, Moeller BC, Gao L, Upton PB, Nakamura J, Starr TB. (2011) Endogenous versus exogenous DNA adducts: their role in carcinogenesis, epidemiology, and risk assessment. Toxicol Sci. 120(Suppl 1):S130-45. PMID 21163908
- 1 2 3 Prasad AR, Prasad S, Nguyen H, Facista A, Lewis C, Zaitlin B, Bernstein H, Bernstein C (2014). "Novel diet-related mouse model of colon cancer parallels human colon cancer". World J Gastrointest Oncol. 6 (7): 225–43. doi:10.4251/wjgo.v6.i7.225. PMC 4092339
 . PMID 25024814.
. PMID 25024814. - 1 2 Valavanidis A, Vlachogianni T, Fiotakis K, Loridas S (2013). "Pulmonary oxidative stress, inflammation and cancer: respirable particulate matter, fibrous dusts and ozone as major causes of lung carcinogenesis through reactive oxygen species mechanisms". Int J Environ Res Public Health. 10 (9): 3886–907. doi:10.3390/ijerph10093886. PMC 3799517
 . PMID 23985773.
. PMID 23985773. - ↑ Tsuei J, Chau T, Mills D, Wan YJ. Bile acid dysregulation, gut dysbiosis, and gastrointestinal cancer. Exp Biol Med (Maywood). 2014 Nov;239(11):1489-504. doi: 10.1177/1535370214538743. PMID 24951470
- ↑ Ajouz H, Mukherji D, Shamseddine A. Secondary bile acids: an underrecognized cause of colon cancer. World J Surg Oncol. 2014 May 24;12:164. doi: 10.1186/1477-7819-12-164. Review. PMID 24884764
- ↑ Bernstein C, Bernstein H (2015). "Epigenetic reduction of DNA repair in progression to gastrointestinal cancer". World J Gastrointest Oncol. 7 (5): 30–46. doi:10.4251/wjgo.v7.i5.30. PMC 4434036
 . PMID 25987950.
. PMID 25987950. - ↑ Scott TL, Rangaswamy S, Wicker CA, Izumi T (2014). "Repair of oxidative DNA damage and cancer: recent progress in DNA base excision repair". Antioxid. Redox Signal. 20 (4): 708–26. doi:10.1089/ars.2013.5529. PMC 3960848
 . PMID 23901781.
. PMID 23901781. - ↑ Li J, Braganza A, Sobol RW (2013). "Base excision repair facilitates a functional relationship between Guanine oxidation and histone demethylation". Antioxid. Redox Signal. 18 (18): 2429–43. doi:10.1089/ars.2012.5107. PMC 3671628
 . PMID 23311711.
. PMID 23311711. - ↑ Nishida N, Arizumi T, Takita M, Kitai S, Yada N, Hagiwara S, Inoue T, Minami Y, Ueshima K, Sakurai T, Kudo M (2013). "Reactive oxygen species induce epigenetic instability through the formation of 8-hydroxydeoxyguanosine in human hepatocarcinogenesis". Dig Dis. 31 (5-6): 459–66. doi:10.1159/000355245. PMID 24281021.
- ↑ Yasui M, Kanemaru Y, Kamoshita N, Suzuki T, Arakawa T, Honma M (2014). "Tracing the fates of site-specifically introduced DNA adducts in the human genome". DNA Repair (Amst.). 15: 11–20. doi:10.1016/j.dnarep.2014.01.003. PMID 24559511.
- ↑ Buechter, DD. (1988) Free radicals and oxygen toxicity.Pharm Res. 5:253-60.
- ↑ Wardman, P. and Candeias, L.P. (1996). Fenton chemistry: an introduction. Radiat. Res. 145, 523–531.
- ↑ Cooke, M. S., Evans, M. D., Dizdaroglu, M., Lunec, J. (2003) Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 17,195–1214
- 1 2 Li, Z., Wu, J. and Deleo, C.J. (2006) RNA Damage and Surveillance under Oxidative Stress. IUBMB Life, 58(10): 581-588
- ↑ Hofer, T., Seo, A. Y., Prudencio, M., and Leeuwenburgh, C. (2006) A method to determine RNA and DNA oxidation simultaneously by HPLC-ECD: greater RNA than DNA oxidation in rat liver after doxorubicin administration. Biol. Chem. 387, 103 – 111
- ↑ Dukan,S., Farwell, A., Ballesteros, M., Taddei, F., Radman,M. and Nystrom,T. (2000) Protein oxidation in response to increased transcriptional and translational errors. Proc. Natl. Acad. Sci. USA, 97 No.11 5746-5749
- ↑ Gajewski, E., Rao, G., Nackerdien, Z., and Dizdaroglu, M. (1990) Modification of DNA bases in mammalian chromatin by radiationgenerated free radicals. Biochemistry 29, 7876 – 7882.
- ↑ Ames, B. N., and Gold, L. S. (1991) Endogenous mutagens and the causes of aging and cancer. Mutat. Res. 250, 3 – 16.
- ↑ Shibutani, S., Takeshita, M., and Grollman, A. P. (1991) Insertion of specific bases during DNA synthesis past the oxidation-damaged base 8-oxodG. Nature 349, 431–434.
- ↑ Taddei, F., Hayakawa, H., Bouton, M., Cirinesi, A., Matic, I., Sekiguchi, M., and Radman, M. (1997) Counteraction by MutT protein of transcriptional errors caused by oxidative damage. Science 278, 128 – 130.
- ↑ Weimann, A., Belling, D., and Poulsen, H. E. (2002) Quantification of 8-oxoGuanine and guanine as the nucleobase, nucleoside and deoxynucleoside forms in human urine by high-performance liquid chromatography-electrospray tandem mass spectrometry. Nucleic Acids Res. 30, E7.
- ↑ Park, E. M., Shigenaga, M. K., Degan, P., Korn, T. S., Kitzler, J. W., Wehr, C. M., Kolachana, P., and Ames, B. N. (1992) Assay of excised oxidative DNA lesions: isolation of 8-oxoguanine and its nucleoside derivatives from biological fluids with a monoclonal antibody column. Proc. Natl. Acad. Sci. USA 89, 3375 – 3379.
- ↑ Shen, Z., Wu, W., and Hazen, S. L. (2000) Activated leukocytes oxidatively damage DNA, RNA, and the nucleotide pool through halide-dependent formation of hydroxyl radical. Biochemistry 39, 5474 – 5482.
- ↑ Kajitani, K., Yamaguchi, H., Dan Y., Furuichi, M., Kang D., and Nakabeppu, Y. (2006) MTH1, and oxidized purine nucleoside triphosphatase, suppresses the accumulation of oxidative damage of nucleic acids in the hippocampalmicroglia during kainite-induced excitotoxicity. J.Neurosci. 26, 1688-1689.