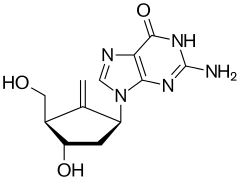
Entecavir
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɛnˈtɛkəvɪər/ en-TEK-a-vir or en-TE-ka-veer |
| Trade names | Baraclude |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605028 |
| License data | |
| Pregnancy category | |
| Routes of administration | Oral |
| ATC code | J05AF10 (WHO) |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | n/a (≥70)[1] |
| Protein binding | 13% (in vitro) |
| Metabolism | negligible/nil |
| Biological half-life | 128–149 hours |
| Excretion | Renal 62–73% |
| Identifiers | |
| |
| CAS Number |
209216-23-9 |
| PubChem (CID) | 153941 |
| DrugBank |
DB00442 |
| ChemSpider |
135679 |
| UNII |
5968Y6H45M |
| KEGG |
D04008 |
| ChEBI |
CHEBI:59902 |
| ChEMBL |
CHEMBL713 |
| Chemical and physical data | |
| Formula | C12H15N5O3 |
| Molar mass | 277.279 g/mol |
| 3D model (Jmol) | Interactive image |
| Melting point | 220 °C (428 °F) value applies to entecavir monohydrate and is a minimum value[2] |
| |
| |
| | |
Entecavir (ETV) is an antiviral medication used in the treatment of hepatitis B virus (HBV) infection. Other uses include liver transplants and HIV/AIDS. Entecavir is used for people living with both HIV and HBV due to its activity against both infections. However, use of entecavir in HIV/AIDS should only be along with other anti-HIV medication to prevent drug resistance. Entecavir is take by mouth as a tablet or solution. Doses are based on a person's weight.[3]
It prevents the hepatitis B virus from multiplying and reduces the amount of virus in the body.[1] Entecavir acts as a guanosine nucleoside analogue reverse transcriptase inhibitor. Other nucleoside and nucleotide analogues include lamivudine, telbivudine, adefovir dipivoxil, and tenofovir.
Medical uses
Entecavir is mainly used to treat chronic hepatitis B infection in adults and children 2 years and older with active viral replication and evidence of active disease with elevations in liver enzymes.[1] It is also used to prevent HBV reinfection after liver transplant[4] and to treat HIV patients infected with HBV. Entecavir is weakly active against HIV, but is not recommended for use in HIV-HBV co-infected patients without a fully suppressive anti-HIV regimen[5] as it may select for resistance to lamivudine and emtricitabine in HIV.[6]
The efficacy of entecavir has been studied in several randomized, double-blind, multicentre trials. Entecavir by mouth is effective and generally well tolerated treatment.[7]
Pregnancy and breastfeeding
It is considered pregnancy category C in the United States, and currently no adequate and well-controlled studies exist in pregnant women.[8]
Side effects
The majority of people who use entecavir have little to no side effects.[9] The most common side effects include headache, fatigue, dizziness, and nausea.[1] Less common effects include trouble sleeping and gastrointestinal symptoms such as sour stomach, diarrhea, and vomiting.[10]
Serious side effects from entecavir include lactic acidosis, liver problems, liver enlargement, and fat in the liver.[3]
Laboratory tests may show an increase in alanine transaminase (ALT), hematuria, glycosuria, and an increase in lipase.[11] Periodic monitoring of hepatic function and hematology are recommended.[1]
Mechanism of action
Entecavir is a nucleoside analog,[12] or more specifically, a deoxyguanosine analogue that belongs to a class of carbocyclic nucleosides and inhibits reverse transcription, DNA replication and transcription in the viral replication process.
Entecavir reduces the amount of HBV in the blood by reducing its ability to multiply and infect new cells.[13]
Administration
Entecavir is take by mouth as a tablet or solution. Doses are based on a person's weight.[3] The solution is recommended for children greater than 2 years old that weigh up to 30kg. Entecavir is recommended on an empty stomach at least 2 hours before or after a meal, generally at the same time everyday. It is not used in children younger than 2 years old. Dose adjustments are also recommended for people with decreased kidney function.[3]
History
- 1992: SQ-34676 at Squibb as part of anti-herpes virus program[14]
- 1997: BMS 200475 developed at BMS pharmaceutical research institute as antiviral nucleoside analogue à Activity demonstrated against HBV, HSV-1, HCMV, VZV in cell lines & no or little activity against HIV or influenza[15]
- Superior activity observed against HBV pushed research towards BMS 200475, its base analogues and its enantiomer against HBV in HepG2.2.15 cell line[15]
- Comparison to other NAs, proven more selective potent inhibitor of HBV by virtue of being Guanine NA[16]
- 1998: Inhibition of hepadnaviral polymerases was demonstrated in vitro in comparison to a number of NAs-TP[17]
- Metabolic studies showed more efficient phosphorylation to triphosphate active form[18]
- 3-year treatment of woodchuck model of CHB à sustained antiviral efficacy and prolonged life spans without detectable emergence of resistance[19]
- Efficacy # LVD resistant HBV replication in vitro[20]
- Superior activity compared to LVD in vivo for both HBeAg+ & HBeAg− patients[21][22]
- Efficacy in LVD refractory CHB patients[23]
- Entecavir was approved by the U.S. FDA in March 2005.
Patent information
Bristol-Myers Squibb was the original patent holder for Baraclude®, the brand name of entecavir in the US and Canada. The drug patent expiration for Baraclude® was in 2015.[24][25] On August 26, 2014, Teva Pharmaceuticals USA gained FDA approval for generic equivalents of Baraclude® 0.5 mg and 1 mg tablets;[26] Hetero Labs received such approval on August 21, 2015;[27] and Aurobindo Pharma on August 26, 2015.[28]
References
- 1 2 3 4 5 "Baraclude (entecavir) Tablets for Oral Use & Oral Solution. U.S. Full Prescribing Information." Bristol-Myers Squibb Company, 2005. Revised December 2013.
- ↑ The Merck Index (14th ed.). 2006. p. 613. ISBN 978-0-911910-00-1.
- 1 2 3 4 "DailyMed - BARACLUDE- entecavir tablet, film coated BARACLUDE- entecavir solution". dailymed.nlm.nih.gov. Retrieved 2016-11-09.
- ↑ Fung, J; Cheung, C; Chan, SC; et al. (2011). "Entecavir Monotherapy is Effective in Suppressing Hepatitis B Virus After Liver Transplantation". Gastroenterology. 141 (4): 1212–9. doi:10.1053/j.gastro.2011.06.083.
- ↑ "Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents" (PDF). Panel on Antiretroviral Guidelines for Adults and Adolescents. Retrieved 15 March 2015.
- ↑ McMahon, Moira (21 June 2007). "The Anti-Hepatitis B Drug Entecavir Inhibits HIV-1 Replication and Can Select HIV-1 Variants Resistant to Antiretroviral Drugs". N Engl J Med. 356 (25): 2614–2621. doi:10.1056/NEJMoa067710. PMID 17582071. Retrieved 15 March 2015.
- ↑ Scott, LJ; Keating, GM (2009). "Entecavir". Drugs. 69 (8): 1003–1033. doi:10.2165/00003495-200969080-00005.
- ↑ "Entecavir (Baraclude) Use During Pregnancy". www.drugs.com. Retrieved 2016-11-07.
- ↑ "Entecavir: Indications, Side Effects, Warnings - Drugs.com". www.drugs.com. Retrieved 2016-11-10.
- ↑ "Entecavir Side Effects in Detail - Drugs.com". www.drugs.com. Retrieved 2016-11-10.
- ↑ "DailyMed - BARACLUDE- entecavir tablet, film coated BARACLUDE- entecavir solution". dailymed.nlm.nih.gov. Retrieved 2016-11-10.
- ↑ Sims KA, Woodland AM (December 2006). "Entecavir: a new nucleoside analog for the treatment of chronic hepatitis B infection". Pharmacotherapy. 26 (12): 1745–57. doi:10.1592/phco.26.12.1745. PMID 17125436.

- ↑ "Entecavir: Indications, Side Effects, Warnings - Drugs.com". www.drugs.com. Retrieved 2016-11-07.
- ↑ Slusarchyk, W. A., A. K. Field, J. A. Greytok, P. Taunk, A. V. Tooumari, M. G. Young, and R. Zahler. 4-Hydroxy-3-(hydroxymethyl)-2-methylcyclopentyl purines and pyrimidines, a novel class of anti-herpesvirus agents. Abstract from the Fifth International Conference on Antiviral Research. Antivir Res 1992.17(Suppl. 1):98
- 1 2 Bisacchi, G. S., S. T. Chao, C. Bachard, J. P. Daris, S. F. Innaimo, J. A. Jacobs, O. Kocy, P. Lapointe, A. Martel, Z. Merchant, W. A. Slusarchyk, J. E. Sundeen, M. G. Young, R. Colonno, and R. Zahler. BMS-200475, a novel carbocyclic 29-deoxyguanosine analog with potent and selective antihepatitis B virus activity in vitro. Bioorg. Med. Chem. Lett. 1997. 7:127–132
- ↑ Innaimo S F, Seifer M, Bisacchi G S, Standring D N, Zahler R, Colonno R J. Identification of BMS-200475 as a Potent and Selective Inhibitor of Hepatitis B Virus. Antimicrob. Agents Chemother. 1997. 41(7): 1444–1448
- ↑ Seifer, M., R. K. Hamatake, R. J. Colonno, and D. N. Standring. In vitro inhibition of hepadnavirus polymerases by the triphosphates of BMS-200475 and lobucavir. Antimicrob. Agents Chemother. 1998. 42:3200–3208
- ↑ Yamanaka, G., T. Wilson, S. Innaimo, G. S. Bisacchi, P. Egli, J. K. Rinehart, R. Zahler, and R. J. Colonno. Metabolic studies on BMS-200475, a new antiviral compound active against hepatitis B virus. Antimicrob. Agents Chemother. 1999. 43:190–193
- ↑ Colonno, R. J., E. V. Genovesi, I. Medina, L. Lamb, S. K. Durham, M. L. Huang, L. Corey, M. Littlejohn, S. Locarnini, B. C. Tennant, B. Rose, and J. M. Clark. Long-term entecavir treatment results in sustained antiviral efficacy and prolonged life span in the woodchuck model of chronic hepatitis infection. J. Infect. Dis. 2001.184:1236–1245
- ↑ Levine, S., D. Hernandez, G. Yamanaka, S. Zhang, R. Rose, S. Weinheimer, and R. J. Colonno. Efficacies of entecavir against lamivudine-resistant hepatitis B virus replication and recombinant polymerases in vitro. Antimicrob. Agents Chemother. 2002.46:2525–2532
- ↑ Chang, T. T. A comparison of entecavir and lamivudine for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 2006. 354:1001–1010
- ↑ Lai CL, Shouval D, Lok AS, Chang TT, Cheinquer H, Goodman Z, DeHertogh D, Wilber R, Zink RC, Cross A, Colonno R, Fernandes L (9 March 2006). "Entecavir versus Lamivudine for Patients with HBeAg-Negative Chronic Hepatitis B". The New England Journal of Medicine. 354 (10): 1011–20. doi:10.1056/NEJMoa051287. PMID 16525138.
- ↑ Sherman, M., C. Yurdaydin, J. Sollano, M. Silva, Y. F. Liaw, J. Cianciara, A. Boron-Kaczmarska, P. Martin, Z. Goodman, R. J. Colonno, A. Cross, G. Denisky, B. Kreter, R. Hindes, and the AI463026 Behold Study Group. Entecavir for the treatment of lamivudine-refractory, HBeAg-positive chronic hepatitis B. Gastroenterology 2006. 130:2039–2049.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Archived from the original on 4 March 2016. Retrieved 2015-08-29.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". Orange Book. Patent and Exclusivity for: N021798. Retrieved 14 November 2016.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Search results from the "OB_Rx" table for query on "202122.". Archived from the original on 22 December 2015. Retrieved 2015-08-29.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Search results from the "OB_Rx" table for query on "205740.". Archived from the original on 4 March 2016. Retrieved 2015-08-29.
- ↑ "Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations". www.accessdata.fda.gov. Search results from the "OB_Rx" table for query on "206217.". Archived from the original on 4 March 2016. Retrieved 2015-08-29.