Isoprenol
 | |
 | |
| Names | |
|---|---|
| IUPAC name
3-Methylbut-3-en-1-ol | |
| Other names
3-Methyl-3-buten-1-ol | |
| Identifiers | |
| 763-32-6 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 12448 |
| ECHA InfoCard | 100.011.009 |
| EC Number | 212-110-8 |
| |
| |
| Properties[1] | |
| C5H10O | |
| Molar mass | 86.132 g/mol |
| Density | 0.853 g/cm3 |
| Boiling point | 130 to 132 °C (266 to 270 °F; 403 to 405 K) |
| Refractive index (nD) |
1.433 |
| Hazards[2] | |
| GHS pictograms |   |
| GHS signal word | WARNING |
| H226, H319 | |
| P210, P233, P240, P241, P242, P243, P264, P280, P303+361+353, P305+351+338, P337+313, P370+378, P403+235, P501 | |
| Flash point | 36 °C (97 °F; 309 K)[note 1] |
| Related compounds | |
| Related compounds |
Prenol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Isoprenol, also known as 3-methylbut-3-en-1-ol, is a hemiterpene alcohol. It is produced industrially as an intermediate to 3-methylbut-2-en-1-ol (prenol): global production in 2001 can be estimated as 6–13 thousand tons.[3]
Isoprenol is produced by the reaction between isobutene (2-methylpropene) and formaldehyde.
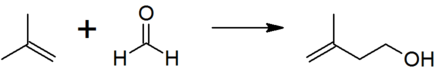
The thermodynamically preferred isomer with the more substituted double bond cannot be directly formed in this reaction, but isomerization yields the desired product:

The reaction is catalyzed by any species which can form an allyl complex without excessive hydrogenation of the substrate, for example poisoned palladium catalysts.[4]
Notes
- ↑ Sigma-Aldrich Co. gives a value for the flash point of isoprenol of 42 °C (108 °F). The difference in the two values does not alter the safety classification of isoprenol as a category 3 flammable liquid under the GHS; but the lower value quoted here (from the New Zealand Environmental Risk Management Authority) would make it a class IC flammable liquid instead of a class II combustible liquid under the U.S. OSHA classification (29 C.F.R § 1910.106), and F3 rather than F2 under the NFPA 704 standard.
References
- ↑ Sigma-Aldrich Co., 3-Methyl-3-buten-1-ol. Retrieved on 2009-08-31..
- ↑ HSNO Chemical Classification Information Database, New Zealand Environmental Risk Management Authority, retrieved 2009-08-31.
- ↑ 3-Methyl-2-buten-1-ol (PDF), SIDS Initial Assessment Report, Geneva: United Nations Environment Programme, May 2005. Major produce in a world is BASF(Germany) and Kuraray(Japan).
- ↑ See, e.g., Kogan, S. B.; Kaliya, M.; Froumin, N. (2006), "Liquid phase isomerization of isoprenol into prenol in hydrogen environment", Appl. Catal. A: Gen., 297 (2): 231–36, doi:10.1016/j.apcata.2005.09.010.
This article is issued from Wikipedia - version of the 6/21/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.