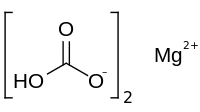
Magnesium bicarbonate
 | |
| Names | |
|---|---|
| IUPAC name
Magnesium hydrogen carbonate | |
| Other names
Magnesium bicarbonate | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 92335 |
| ECHA InfoCard | 100.016.582 |
| PubChem | 102204 |
| |
| Properties | |
| Mg(HCO3)2 | |
| Molar mass | 146.34 g/mol |
| 0.077 g / (100 mL) | |
| Related compounds | |
| Other cations |
Calcium bicarbonate |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Magnesium bicarbonate or magnesium hydrogen carbonate, Mg(HCO3)2, is the bicarbonate salt of magnesium. It can be formed through the reaction of dilute solutions of carbonic acid (such as seltzer water) and magnesium hydroxide (milk of magnesia).
Magnesium bicarbonate exists only in aqueous solution. To produce it, a suspension of magnesium hydroxide is treated with pressurized carbon dioxide, producing a solution of magnesium bicarbonate:[1]
- Mg(OH)2 + 2 CO2 → Mg(HCO3)2
Drying the resulting solution causes the magnesium bicarbonate to decompose, yielding magnesium carbonate, carbon dioxide, and water:
- Mg2+ + 2 HCO3− → MgCO3 + CO2 + H2O
References
- ↑ Margarete Seeger; Walter Otto; Wilhelm Flick; Friedrich Bickelhaupt; Otto S. Akkerman (2005), "Magnesium Compounds", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a15_595.pub2
This article is issued from Wikipedia - version of the 11/16/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.