Orthoester
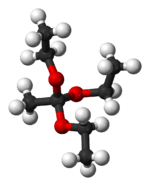
In organic chemistry, an orthoester is a functional group containing three alkoxy groups attached to one carbon atom, i.e. with the general formula RC(OR′)3. Orthoesters may be considered as products of exhaustive alkylation of unstable orthocarboxylic acids and it is from these that the name 'ortho ester' is derived. An example is ethyl orthoacetate, CH3C(OCH2CH3)3, more correctly known as 1,1,1-triethoxyethane. Orthoesters are used in organic synthesis as protecting groups for esters.
Synthesis
Orthoesters can be prepared by the Pinner reaction, in which nitriles react with alcohols under acid catalysis:
- RCN + 3 R′OH → RC(OR′)3 + NH3
Reactions
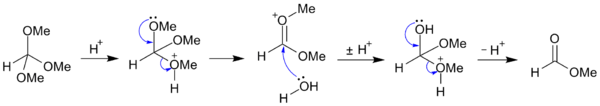
Hydrolysis
Orthoesters are readily hydrolyzed in mild aqueous acid to form esters:
- RC(OR′)3 + H2O → RCO2R′ + 2 R′OH
For example, trimethyl orthoformate CH(OCH3)3 may be hydrolyzed (under acidic conditions) to methyl formate and methanol;[1] and may be further hydrolyzed (under alkaline conditions) to salts of formic acid and methanol.[2]
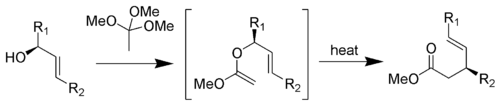
Johnson–Claisen rearrangement
The Johnson–Claisen rearrangement is the reaction of an allylic alcohol with an orthoester containing a deprotonatable alpha carbon (e.g. triethyl orthoacetate) to give a γ,δ-unsaturated ester.[3]
Bodroux–Chichibabin aldehyde synthesis
In the Bodroux–Chichibabin aldehyde synthesis an orthoester reacts with a Grignard reagent to form an aldehyde; this is an example of a formylation reaction.
As a protecting group

Both trimethylorthoacetate and triethylorthoacetate are commonly used reagents in organic chemistry. Another example is the bicyclic OBO protecting group (4-methyl-2,6,7-trioxa-bicyclo[2.2.2]octan-1-yl) which is formed by the action of (3-methyloxetan-3-yl)methanol on activated carboxylic acids in the presence of Lewis acids and was developed by Elias James Corey. The group is base stable and can be cleaved in two steps under mild conditions, mildly acidic hydrolysis yields the ester of tris(hydroxymethyl)ethane which is then cleaved using e.g. an aqueous carbonate solution.[4]
See Also
- Acetal - C(OR)2R2
- Orthocarbonate - C(OR)4.
References
- ↑ Clayden, Jonathan; Greeves, Nick; Warren, Stuart; Wothers, Peter (2001). Organic Chemistry (1st ed.). Oxford University Press. p. 345. ISBN 978-0-19-850346-0.
- ↑ United States Patent Application 20070049501, Saini; Rajesh K.; and Savery; Karen, March 1, 2007
- ↑ Johnson, William Summer.; Werthemann, Lucius.; Bartlett, William R.; Brocksom, Timothy J.; Li, Tsung-Tee.; Faulkner, D. John.; Petersen, Michael R. (February 1970). "Simple stereoselective version of the Claisen rearrangement leading to trans-trisubstituted olefinic bonds. Synthesis of squalene". Journal of the American Chemical Society. 92 (3): 741–743. doi:10.1021/ja00706a074.
- ↑ Kocieński, Philip J. (2005). Protecting groups (3. ed.). Stuttgart: Thieme. ISBN 978-3-13-135603-1.