Silicate
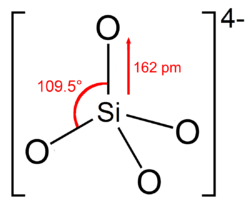
4) structure
A silicate is a compound containing an anionic silicon compound. The great majority of the silicates are oxides, but hexafluorosilicate ([SiF6]2−) and other anions are also included.
"Orthosilicate" is the anion SiO4−
4 or its compounds. Related to orthosilicate are families of anions (and their compounds) with the formula [SiO2+n]2n−. Important members are the cyclic and single chain silicates {[SiO3]2−}n and the sheet-forming silicates {[SiO2.5]−}n.[1]
Silicates constitute the majority of Earth's crust, as well as the other terrestrial planets, rocky moons, and asteroids. Sand, Portland cement, and thousands of minerals are examples of silicates. Silicate compounds, including the minerals, consist of silicate anions whose charge is balanced by various cations. Myriad silicate anions can exist, and each can form compounds with many different cations. Hence this class of compounds is very large. Both minerals and synthetic materials fit in this class.
Structural principles
In the vast majority of silicates, including silicate minerals, the Si occupies a tetrahedral environment, being surrounded by 4 oxygen centres. In these structures, the chemical bonds to silicon conform to the octet rule. These tetrahedra sometimes occur as isolated SiO4−
4 centres, but most commonly, the tetrahedra are joined together in various ways, such as pairs (Si
2O6−
7) and rings (Si
6O12−
18). Commonly the silicate anions are chains, double chains, sheets, and three-dimensional frameworks. All these such species have negligible solubility in water at normal conditions.
Occurrence in solution
Silicates are well characterized as solids, but are less commonly observed in solution. The anion SiO4−
4 is the conjugate base of silicic acid, Si(OH)4, and both are elusive as are all of the intermediate species. Instead, solutions of silicates are usually observed as mixtures of condensed and partially protonated silicate clusters. The nature of soluble silicates is relevant to understanding biomineralization and the synthesis of aluminosilicates, such as the industrially important catalysts called zeolites.[2]
Silicates with non-tetrahedral silicon
Although the tetrahedron is the common coordination geometry for silicon compounds, silicon is well known to also adopt higher coordination numbers. A well-known example of such a high coordination number is hexafluorosilicate (SiF2−
6). Octahedral coordination by 6 oxygen centres is observed. At very high pressure, even SiO2 adopts this geometry in the mineral stishovite, a dense polymorph of silica found in the lower mantle of the Earth. This structure is also formed by shock during meteorite impacts. Octahedral Si in the form of hexahydroxysilicate ([Si(OH)6]2−) is observed in thaumasite a mineral occurring rarely in nature but sometimes observed amongst other calcium silicate hydrate artificially formed in cement and concrete submitted to a severe sulfate attack.
Silicate rock and minerals
In geology and astronomy, the term silicate is used to denote types of rock that consist predominantly of silicate minerals. On Earth, a wide variety of silicate minerals occur in an even wider range of combinations as a result of the processes that form and re-work the crust. These processes include partial melting, crystallization, fractionation, metamorphism, weathering and diagenesis. Living things also contribute to the silicate cycle near the Earth's surface. A type of plankton known as diatoms construct their exoskeletons, known as tests, from silica. The tests of dead diatoms are a major constituent of deep ocean sediment.
Silica, or silicon dioxide, SiO2, is sometimes considered a silicate, although it is the special case with no negative charge and no need for counter-ions. Silica is found in nature as the mineral quartz, and its polymorphs.
Portland cement
Portland cement is produced by hydration of clinker, which consists of various calcium silicates, including tricalcium silicate (Ca3SiO5, also written CaO·Ca2SiO4) and dicalcium silicate (Ca2SiO4). These components are often generated in situ by heating various clays and limestone.[3]
Mineralogy
Mineralogically, silicate minerals are divided according to structure of their silicate anion into the following groups:[4][5]
- Nesosilicates (lone tetrahedron) – [SiO4]4−, e.g. olivine.
- Sorosilicates (double tetrahedra) – [Si2O7]6−, e.g. epidote, melilite group.
- Cyclosilicates (rings) – [SinO3n]2n−, e.g. tourmaline group.
- Inosilicates (single chain) – [SinO3n]2n−, e.g. pyroxene group.
- Inosilicates (double chain) – [Si4nO11n]6n−, e.g. amphibole group.
- Phyllosilicates (sheets) – [Si2nO5n]2n−, e.g. micas and clays.
- Tectosilicates (3D framework) – [AlxSiyO2x+2y)]x−, e.g. quartz, feldspars, zeolites.
Note that tectosilicates can only have additional cations if some of the silicon is replaced by a lower-charge cation such as aluminium. Al for Si substitution is common.
References
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ Knight, Christopher T. G.; Balec, Raymond J.; Kinrade, Stephen D. (2007). "The Structure of Silicate Anions in Aqueous Alkaline Solutions". Angewandte Chemie International Edition. 46: 8148–8152. doi:10.1002/anie.200702986.
- ↑ Siegbert Sprung "Cement" in Ullmann's Encyclopedia of Industrial Chemistry, 2012 Wiley-VCH, Weinheim. doi:10.1002/14356007.a05_489.pub2
- ↑ Deer, W.A.; Howie, R.A., & Zussman, J. (1992). An introduction to the rock forming minerals (2nd edition ed.). London: Longman ISBN 0-582-30094-0
- ↑ Hurlbut, Cornelius S.; Klein, Cornelis (1985). Manual of Mineralogy, Wiley, (20th edition ed.). ISBN 0-471-80580-7