1,4-Naphthoquinone
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Naphthalene-1,4-dione | |
| Other names
1,4-Naphthoquinone Naphthoquinone α-Naphthoquinone | |
| Identifiers | |
| 130-15-4 | |
| 3D model (Jmol) | Interactive image |
| ChEMBL | ChEMBL55934 |
| ChemSpider | 8215 |
| ECHA InfoCard | 100.004.526 |
| PubChem | 8530 |
| |
| |
| Properties | |
| C10H6O2 | |
| Molar mass | 158.15 g/mol |
| Density | 1.422 g/cm3 |
| Melting point | 126 °C (259 °F; 399 K) |
| Boiling point | Begins to sublime at 100 °C |
| 0.09 g/L | |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
1,4-Naphthoquinone or para-naphthoquinone is an organic compound derived from naphthalene. Several isomeric naphthoquinones are known, notably 1,2-naphthoquinone. 1,4-Naphthoquinone forms volatile yellow triclinic crystals and has a sharp odor similar to benzoquinone. It is almost insoluble in cold water, slightly soluble in petroleum ether, and more soluble in polar organic solvents. In alkaline solutions it produces a reddish-brown color. Vitamin K is a derivative of 1,4-naphthoquinone. It is a planar molecule with one aromatic ring fused to a quinone subunit.[2]
Preparation
The industrial synthesis involves aerobic oxidation of naphthalene over a vanadium oxide catalyst:[3]
- C10H8 + 3/2 O2 → C10H6O2 + H2O
In the laboratory, naphthoquinone can be produced by the oxidation of a variety of naphthalene compounds. An inexpensive route involves oxidation of naphthalene with chromium trioxide.[4]
Reactions
1,4-Naphthoquinone acts as strong dienophile in Diels-Alder reaction. Its adduct with 1,3-butadiene can be prepared by two methods: 1) long (45 days) exposure of naphthoquinone in neat liquid butadiene taken in huge excess at room temperature in a thick-wall glass tube or 2) fast catalyzed cycloaddition at low temperature in the presence of 1 equivalent of tin(IV) chloride:[5]
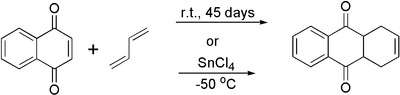
Uses
1,4-Naphthoquinone is mainly used as a precursor to anthroquinone by reaction with butadiene followed by oxidation. Nitration gives 5-nitro-1,4-naphthalenedione, precursor to an aminoanthroquinone that is used as a dye precursor.[3]
Derivatives
Naphthoquinone forms the central chemical structure of many natural compounds, most notably the K vitamins. 2-Methylnaphthoquinone is a more effective coagulant than vitamin K.
Other natural naphtoquinones include juglone, plumbagin, droserone.
Naphthoquinone derivatives have significant pharmacological properties. They are cytotoxic, they have significant antibacterial, antifungal, antiviral, insecticidal, anti-inflammatory, and antipyretic properties. Plants with naphthoquinone content are widely used in China and the countries of South America, where they are used to treat malignant and parasitic diseases.[6]
Naphthoquinone functions as a ligand (through the electrophilic C=C).[7]
See also
References
- ↑ Merck Index, 11th Edition, 6315.
- ↑ Gaultier, J.; Hauw, C. (1965). "Structure de l'α-Naphtoquinone". Acta Crystallographica. 18 (2): 179–183. doi:10.1107/S0365110X65000439.
- 1 2 Grolig, J.; Wagner, R. (2005), "Naphthoquinones", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a17_067
- ↑ Braude, E. A.; Fawcett, J. S. (1953). "1,4-Naphthoquinone" (PDF). Org. Synth. 33: 50.; Coll. Vol., 4, p. 698
- ↑ M.A. Filatov; S. Baluschev; I.Z. Ilieva; V. Enkelmann; T. Miteva; K. Landfester; S.E. Aleshchenkov; A.V. Cheprakov (2012). "Tetraaryltetraanthra[2,3]porphyrins: Synthesis, Structure, and Optical Properties". J. Org. Chem. 77 (24): 11119–11131. doi:10.1021/jo302135q.
- ↑ Babula, P.; Adam, V.; Havel, L.; Kizek, R. (2007). "Naphthoquinones and their Pharmacological Properties". Ceská a Slovenská Farmacie (in Czech). 56 (3): 114–120. PMID 17867522.
- ↑ Kündig, E. P.; Lomberget, T.; Bragg, R.; Poulard, C.; Bernardinelli, G. (2004). "Desymmetrization of a meso-Diol Complex Derived from [Cr(CO)3(η6-5,8-Naphthoquinone)]: Use of New Diamine Acylation Catalysts". Chemical Communications. 2004 (13): 1548–1549. doi:10.1039/b404006f.