Angiomyolipoma
| Angiomyolipoma | |
|---|---|
|
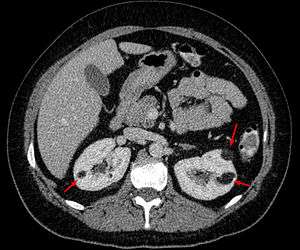
Angiomyolipoma in both kidneys (arrows) in computer tomography. The tumours are hypodense (dark) due to fat content | |
| Classification and external resources | |
| Specialty | oncology |
| ICD-10 | D30.0 |
| ICD-9-CM | 223.0 |
| ICD-O | M8860/0 |
| DiseasesDB | 29496 |
| MeSH | D018207 |
Angiomyolipomas are the most common benign tumour of the kidney and are composed of blood vessels, smooth muscle cells and fat cells. Angiomyolipomas are strongly associated with the genetic disease tuberous sclerosis, in which most individuals will have several angiomyolipomas affecting both kidneys. They are also commonly found in women with the rare lung disease lymphangioleiomyomatosis. Angiomyolipomas are less commonly found in the liver and rarely in other organs. Whether associated with these diseases or sporadic, angiomyolipomas are caused by mutations in either the TSC1 or TSC2 genes, which govern cell growth and proliferation.
Although regarded as benign, angiomyolipomas may grow such that kidney function is impaired or the blood vessels may dilate and burst leading to haemorrhage. Large angiomyolipoma can be treated with embolisation. Drug therapy for angiomyolipoma is at the research stage.
The Tuberous Sclerosis Alliance has recently published Guidelines that provide useful information for the diagnosis, surveillance and management of angiomyolipomas.[1]
Classification
Angiomyolipomas are tumours consisting of perivascular epithelioid cells (cells which are found surrounding blood vessels and which resemble epithelial cells). A tumour of this kind is known as a PEComa, from the initials of perivascular epithelioid cell. Older literature may classify them as hamartomas (benign tumours consisting of cells in their correct location but forming a disorganised mass) or choristoma (benign tumours consisting of normal cells in the wrong location). PEComas are themselves a kind of mesenchymal tumour which involves cells that form the connective tissue, cardiovascular and lymphatic systems.[2]
An angiomyolipoma is composed of varying proportions of vascular cells, immature smooth muscle cells and fat cells.[2] These three components respectively give rise to the components of the name: angio-, myo- and lip-. The -oma suffix indicates a tumour.
Angiomyolipomas are typically found in the kidney but have also been commonly found in the liver and less commonly the ovary, fallopian tube, spermatic cord, palate and colon.[2]
Symptoms
If the dilated blood vessels in an angiomyolipoma rupture, the resulting retroperitoneal haemorrhage causes sudden pain, accompanied with nausea and vomiting. When the patient presents in the emergency department, up to 20% will be in shock.[2]
Pathophysiology
Since all three components of an angiomyolipoma (vascular cells, immature smooth muscle cells and fat cells) contain a "second hit" mutation, they are believed to have derived from a common progenitor cell that suffered the common second hit mutation.[2]
Diagnosis
There are three methods of scanning that detect angiomyolipoma: ultrasound, CT and MRI. Ultrasound is standard and is particularly sensitive to the fat in angiomyolipoma but less so to the solid components. However it is hard to make accurate measurements with ultrasound. Computed tomography (CT) is very detailed and fast and allows accurate measurement. However, it exposes the patient to radiation and the dangers that a contrast dye used to aid the scanning may itself harm the kidneys. Magnetic resonance imaging (MRI) is safer than CT but many patients (particularly those with the learning difficulties or behavioural problems found in tuberous sclerosis) require sedation or general anaesthesia and the scan cannot be performed quickly.[2] Some other kidney tumours contain fat, so the presence of fat isn't diagnostic. It can be difficult to distinguish a fat-poor angiomyolipoma from a renal cell carcinoma[3] and a lesion growing at greater than 5 mm per year may warrant a biopsy in order to distinguish it from this form of cancer.[2]
Incidental discovery of angiomyolipomas should trigger consideration of tuberous sclerosis complex (TSC) and lymphangioleiomyomatosis, especially if they are large, bilateral and/or multiple. Screening for TSC includes a detailed physical exam, including dermatologic and ophthalmologic evaluations, by TSC expert clinicians and a CT or MRI of the brain. Screening for LAM includes a high resolution CT of the lung and pulmonary function testing.
Treatment
Everolimus is FDA approved for the treatment of angiomyolipomas. Treatment should be considered for asymptomatic, growing AML measuring larger than 3 cm in diameter.[1]
Angiomyolipoma do not normally require surgery unless there is life-threatening bleeding.[3] Some centres may perform preventative selective embolisation of the angiomyolipoma if it is more than 4 cm in diameter, due to the risk of haemorrhage.[4]
People with tuberous sclerosis are advised to have yearly renal scans, though it is possible that patients with very stable lesions could be monitored less frequently. The research in this area is lacking. Even if no angiomyolipoma is found, one can develop at any life stage. The angiomyolipoma can grow rapidly.[2]
In tuberous sclerosis, typically many angiomyolipomas affecting each kidney. It is not uncommon for more than one intervention to be required during lifetime. Since kidney function may already be impaired (up to half the kidney may be lost before function loss is detectable), it is vital to preserve as much kidney as possible when removing any lesion. Large angiomyolipomas are treated by embolisation which reduces the risk of haemorrhage and can also shrink the lesion. A side effect of this treatment is postembolisation syndrome: severe pain and fever however this is easily managed and lasts only a few days.[2]
A ruptured aneurysm in an angiomyolipoma leads to blood loss that must be stopped (though embolisation) and compensated for (through intravenous fluid replacement). Therefore, removal of the affected kidney (nephrectomy) is strongly discouraged though may occur if the emergency department is not knowledgeable about tuberous sclerosis.[2]
Embolisation involves inserting a catheter along the blood vessels to the tumour. The blood vessels are then blocked, typically by injecting ethanol or inert particles. The procedure can be very painful, so analgesics are used. The destroyed kidney tissue often causes post-embolisation syndrome, which manifests as nausea, vomiting, fever and abdominal pain, and lasts a few days. Embolisation (in general) has an 8% rate of morbidity and a 2.5% rate of mortality, so is not considered lightly.[4]
Patients with kidney loss should be monitored for hypertension (and treated for it if discovered) and avoid nephrotoxic drugs such as certain pain relievers and IV contrast agents. Such patients who are unable to communicate effectively (due to age or intellectual disability) are at risk of dehydration. Where multiple or large angiomyolipomas have caused chronic kidney disease, dialysis is required.[2]
Robotic assisted partial nephrectomy has been proposed as a surgical treatment of a ruptured angiomyolipoma combining the advantages both of a kidney preservation procedure and the benefits of a minimal invasive procedure without compromising the safety of the patient.[5]
Prognosis
Small angiomyolipomas and those without dilated blood vessels (aneurysms) cause few problems, but angiomyolipomas have been known to grow as rapidly as 4 cm in one year. An angiomyolipoma larger than 5 cm and those containing an aneurysm pose a significant risk of rupture, which is a medical emergency as it is potentially life-threatening. One population study found the cumulative risk of haemorrhage to be 10% in males and 20% in females.[2]
A second problem occurs when the renal angiomyolipomas take over so much kidney that the function is impaired leading to chronic kidney disease. This may be severe enough to require dialysis. A population survey of patients with TSC and normal intelligence found 1% were on dialysis.[2]
Epidemiology
Angiomyolipomas are the most common benign tumour of the kidney and are found either in patients with tuberous sclerosis or sporadically. About 80–90% of cases are sporadic and these are most commonly found in middle-aged women.[3]
In patients with TSC, a longitudinal study found 80% will have some form of renal lesion by around 10 years of age. Of these, 75% are angiomyolipomas and 17% cysts. The angiomyolipomas increased in size in around 60% of these children. An autopsy study and TSC clinic survey found a prevalence of 67% and 85% respectively for patients with TSC. Both genders are affected equally.[2]
Other Resources
The Tuberous Sclerosis Alliance and The LAM Foundation provide education and support for families affected by tuberous sclerosis complex, lymphangioleiomyomatosis and angiomyolipomas. Both organizations support networks of clinics that are expert in the diagnosis and management of angiomyolipomas.
References
- 1 2 Northrup, H; Krueger, DA (2013). "International Tuberous Sclerosis Complex Consensus Group. Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference". Pediatr Neurol. 49 (4): 243–254. doi:10.1016/j.pediatrneurol.2013.08.001. PMC 4080684
 . PMID 24053982.
. PMID 24053982. - 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Bissler JJ, Henske EP. Renal Manifestations of Tuberous Sclerosis Complex. In: Kwiatkowski DJ, Wiittlemore DJ, Thiele EA, editors. Tuberous Sclerosis Complex: Genes, Clinical Features and Therapeutics. Wiley-VCH Verlag GmbH; 2010. p. 321–325. ISBN 3-527-32201-9.
- 1 2 3 Shin, NY; Kim, MJ; Chung, JJ; Chung, YE; Choi, JY; Park, YN (May–Jun 2010). "The differential imaging features of fat-containing tumors in the peritoneal cavity and retroperitoneum: the radiologic-pathologic correlation." (PDF). Korean Journal of Radiology. 11 (3): 333–45. doi:10.3348/kjr.2010.11.3.333. PMC 2864861
 . PMID 20461188.
. PMID 20461188. - 1 2 Loffroy, R; Rao, P; Kwak, BK; Ota, S; De Lin, M; Liapi, E; Geschwind, JF (May–Jun 2010). "Transcatheter arterial embolization in patients with kidney diseases: an overview of the technical aspects and clinical indications." (PDF). Korean Journal of Radiology. 11 (3): 257–68. doi:10.3348/kjr.2010.11.3.257. PMC 2864852
 . PMID 20461179.
. PMID 20461179. - ↑ Ploumidis, A; Katafigiotis, I; Thanou, M; Bodozoglou, N; Athanasiou, L; Ploumidis, A (2013). "Spontaneous Retroperitoneal Hemorrhage (Wunderlich Syndrome) due to Large Upper Pole Renal Angiomyolipoma: Does Robotic-Assisted Laparoscopic Partial Nephrectomy Have a Role in Primary Treatment?". Case reports in urology. 2013: 498694. doi:10.1155/2013/498694. PMC 3784227
 . PMID 24106637.
. PMID 24106637.