Dicobalt octacarbonyl
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Octacarbonyldicobalt(Co—Co) | |
| Other names
Cobalt carbonyl, di-mu-Carbonylhexacarbonyldicobalt, Cobalt octacarbonyl, Cobalt tetracarbonyl dimer, Dicobalt carbonyl, Octacarbonyldicobalt | |
| Identifiers | |
| 10210-68-1 | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 2007057 |
| ECHA InfoCard | 100.030.454 |
| PubChem | 25049 |
| RTECS number | GG0300000 |
| UNII | MDH533S43Q |
| UN number | 3281 |
| |
| |
| Properties | |
| Co2(CO)8 | |
| Molar mass | 341.95 g/mol |
| Appearance | red-orange crystals white crystalline solid when pure[1] |
| Density | 1.87 g/cm3 |
| Melting point | 51 to 52 °C (124 to 126 °F; 324 to 325 K) |
| Boiling point | 52 °C (126 °F; 325 K) c.a. decomposes |
| insoluble | |
| Vapor pressure | 0.7 mmHg (20 °C)[1] |
| Structure | |
| 1.33 D (C2v isomer) 0 D (D3d isomer) | |
| Hazards | |
| Main hazards | Toxic, evolves CO gas on decomposition[1] |
| Safety data sheet | External MSDS |
| Flash point | nonflammable [1] |
| US health exposure limits (NIOSH): | |
| PEL (Permissible) |
none[1] |
| REL (Recommended) |
TWA 0.1 mg/m3[1] |
| IDLH (Immediate danger) |
N.D.[1] |
| Related compounds | |
| Related metal carbonyls |
Iron pentacarbonyl Diiron nonacarbonyl Nickel tetracarbonyl |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
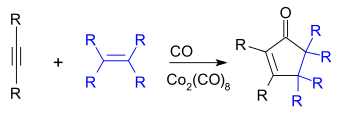
Dicobalt octacarbonyl is the inorganic compound Co2(CO)8. This metal carbonyl is a reagent and catalyst in organometallic chemistry and organic synthesis.[2] It is used as a catalyst for hydroformylation, the conversion of alkenes into aldehydes.[3] It is highly reactive towards alkynes, and is sometimes used as an alkyne protecting group. As the cobalt-alkyne complex, it plays a role in promoting both the Nicholas reaction and the Pauson–Khand reaction.
Synthesis, structure, properties
It is synthesised by the high pressure carbonylation of cobalt(II) salts, often in the presence of cyanide. It is an orange-colored, pyrophoric solid that is thermally unstable. It exists as two isomers in solution[4] that rapidly interconvert.
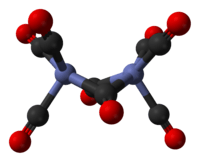
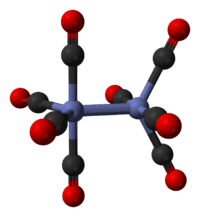
The major isomer contains two bridging CO ligands linking the Co atoms and six terminal CO ligands, three on each Co atom. It can be described by the formula (CO)3Co(μ-CO)2Co(CO)3 and has C2v symmetry. This structure resembles Fe2(CO)9, but with one fewer bridging CO. The Co---Co distance is 2.52 Å, and the Co–COterminal and Co–CObridge distances are 1.80 and 1.90 Å, respectively.[5] Analysis of the bonding suggests the absence of a direct Co–Co bond.[6]
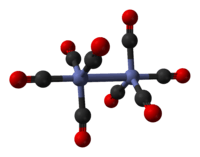
The minor isomer has no bridging CO ligands, but instead has a direct Co–Co bond and eight terminal CO ligands, four on each Co atom. It can be described by the formula (CO)4Co-Co(CO)4 and has D3d symmetry. It features an unbridged Co-Co bond that is 2.70 Å in length.[7] The minor isomer has been crystallized together with C60.
Reactions
The most characteristic reaction of Co2(CO)8 is its hydrogenation to tetracarbonylhydrocobalt, [HCo(CO)4]:
- Co2(CO)8 + H2 → 2 HCo(CO)4
This hydride is the active agent for hydroformylation reactions. It adds to alkenes to give an alkyl-Co(CO)4 product that then proceeds to insert CO and undergo hydrogenolysis to produce the aldehyde. Reduction of Co2(CO)8 gives the conjugate base of HCo(CO)4:
- Co2(CO)8 + 2 Na → 2 NaCo(CO)4
The CO ligands can be replaced with tertiary phosphine ligands to give Co2(CO)8-x(PR3)x. These bulky derivatives are more selective catalysts for hydroformylation reactions. "Hard" Lewis bases, e.g. pyridine, cause disproportionation:
- 6 C5H5N + 1.5 Co2(CO)8 → [Co(C5H5N)6][Co(CO)4]2 + 4 CO
The Pauson–Khand reaction, in which an alkyne, an alkene, and carbon monoxide cyclize to give a cyclopentenone, can be catalyzed by Co2(CO)8:
Co2(CO)8 reacts with alkynes to form a stable covalent complex, which is useful as a protective group for the alkyne. This complex itself can also be used in the Pauson–Khand reaction.
Heating causes decarbonylation and formation of the tetrahedral cluster:
- 2 Co2(CO)8 → Co4(CO)12 + 4 CO
Safety
Co2(CO)8 a volatile source of cobalt(0), is pyrophoric and releases carbon monoxide upon decomposition.[8] The National Institute for Occupational Safety and Health has recommended that workers should not be exposed to concentrations greater than 0.1 mg/m3 over an eight hour time-weighted average, without the proper respiratory gear.[9]
See also
References
- 1 2 3 4 5 6 7 "NIOSH Pocket Guide to Chemical Hazards #0147". National Institute for Occupational Safety and Health (NIOSH).
- ↑ Pauson, P. L. “Octacarbonyldicobalt” in Encyclopedia of Reagents for Organic Synthesis (Ed: L. Paquette) 2004, J. Wiley & Sons, New York. doi:10.1002/047084289X.ro001.pub3
- ↑ Elschenbroich, C.; Salzer, A. ”Organometallics : A Concise Introduction” (2nd Ed) (1992) Wiley-VCH: Weinheim. ISBN 3-527-28165-7
- ↑ Ray L. Sweany and Theodore L. Brown "Infrared spectra of matrix-isolated dicobalt octacarbonyl. Evidence for the third isomer" Inorganic Chemistry 1977, 16, pp 415 - 421. doi:10.1021/ic50168a037
- ↑ G. G. Sumner, H. P. Klug, L. E. Alexander "The crystal structure of dicobalt octacarbonyl" Acta Crystallographica, 1964 Volume 17 Part 6 Pages 732-742. doi:10.1107/S0365110X64001803
- ↑ Jennifer C. Green, Malcolm L. H. Green, Gerard Parkin "The occurrence and representation of three-centre two-electron bonds in covalent inorganic compounds" Chem. Commun. 2012, 11481-11503. doi:10.1039/c2cc35304k
- ↑ Thelma Y. Garcia, James C. Fettinger, Marilyn M. Olmstead, Alan L. Balch, "Splendid symmetry: crystallization of an unbridged isomer of Co2(CO)8 in Co2(CO)8·C60" Chem. Commun., 2009, p. 7143-7145. doi:10.1039/b915083h.
- ↑ Cole Parmer MSDS
- ↑ CDC - NIOSH Pocket Guide to Chemical Hazards
8NoCo-Co.png)