Crofelemer
 | |
| Clinical data | |
|---|---|
| Trade names | Mytesi (Fulyzaq) |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral (tablets) |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Little or no absorption from the gut |
| Identifiers | |
| Synonyms | SP-303 |
| CAS Number | 148465-45-6 |
| PubChem (SID) | 17397714 |
| ChemSpider | none |
| UNII | PY79D6C8RX |
| KEGG | D03605 |
| Chemical and physical data | |
| Formula | (C15O6,7H12)n |
| Molar mass | 860–9100 g/mol |
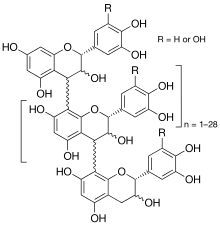
Crofelemer (USAN, trade name Mytesi) is a botanical drug for the treatment of diarrhoea associated with anti-HIV drugs such as nucleoside analog reverse transcriptase inhibitors and protease inhibitors. Other possible uses include diarrhoea in children, acute infectious diarrhoea, and diarrhoea in patients with irritable bowel syndrome.[1] It is a purified oligomeric proanthocyanidin from "dragon's blood", the sap of the South American tree Croton lechleri.[2]
Crofelemer treats the symptoms of disease, but it is not used to treat infectious diarrhoea (diarrhoea caused by infection of the digestive system by a bacterium, virus or parasite). It was initially developed by Napo Pharmaceuticals, which licensed it to Glenmark Pharmaceuticals in 140 emerging markets and to Salix Pharmaceuticals in the US, EU and some other markets. A Phase III clinical trial for diarrhoea in HIV patients was completed in 2012, and the drug was approved by the US Food and Drug Administration (FDA) on 31 December 2012.[3][4][5]
Mechanism of action
The drug is taken by mouth and works by voltage-independently blocking two structurally unrelated chloride channels in the gut, namely the cystic fibrosis transmembrane conductance regulator (CFTR) with an in vitro maximum inhibition of about 60%, and the calcium-activated chloride channel anoctamin 1, with a maximum inhibition of over 90%.[2] This is a hitherto undescribed mechanism of action. As a result of the channel inhibition, fewer chloride ions are excreted into the gut, which also decreases the excretion of sodium ions and water, improving stool consistency and reducing duration of the diarrhoea.[3][6] The mechanism seems to be selective as other channels involved in intestinal fluid secretion, namely sodium and potassium channels, are not affected by crofelemer, nor is cAMP or calcium signalling.[2]
The substance is hardly, if at all, absorbed from the gut into the bloodstream, and is consequently excreted with the stool.[6]
Adverse effects and interactions
Crofelemer seems to be well tolerated; the only adverse effects found in clinical studies were mild gastrointestinal effects at the same level as under placebo.[6] Studies regarding interactions with other drugs are still to be conducted.
Origin and chemistry

The substance is a purified oligomeric proanthocyanidin from the sap, or more correctly the latex, of the South American tree Croton lechleri (locally called Sangre de Grado or Sangre de Drago). This is one of several plants producing bright red latex or resin called "dragon's blood".[2] Crofelemer is a complex mixture of procyanidins and prodelphinidins with up to 30 (epi)catechin or (epi)gallocatechin units per molecule, resulting in a molecular mass of up to 9 kDa.[2]
History
The latex of C. lechleri is traditionally used in South American medicine for the treatment of diarrhoea, wounds, inflammations, tumours, insect bites, and other conditions.[2][7] A number of chemicals were isolated in the late 1980s and 1990s and tested in cellular and animal models, for example identifying taspine as a cicatrizant (wound healing promoter).[8] Immunomodulatory, antioxidative, antiproliferative and mutagenic effects of dragon's blood and its components also received some attention from the scientific community. The purified proanthocyanidin fraction was first described in 1994 under the name SP-303 as an antiviral substance,[9] but a study testing it for the treatment of herpes simplex was not successful.[10] In 1999 the drug was reported to improve the symptoms of cholera toxin induced diarrhoea in mice.[11]
SP-303 was eventually named crofelemer and patented by Napo Pharmaceuticals, which licensed it to Glenmark Pharmaceuticals in 2005, for exclusive development and marketing rights in 140 emerging markets including India,[12] and to Salix Pharmaceuticals for exclusive development and marketing rights in North America, the European Union and Japan, in 2008.[13] Subsequently, Napo sued Salix and terminated the agreements with Salix and Glenmark in 2011, alleging that they were stalling the drug's development.[14] As of October 2012, crofelemer has completed a Phase III trial and was approved in December 2012 by the FDA for the indication "symptomatic relief of non-infectious diarrhoea in patients with HIV/AIDS on anti-retroviral therapy".[1][5]
References
- 1 2 Drugs.com: Crofelemer Approval Status
- 1 2 3 4 5 6 Tradtrantip, L.; Namkung, W.; Verkman, A. S. (2009). "Crofelemer, an Antisecretory Antidiarrheal Proanthocyanidin Oligomer Extracted from Croton lechleri, Targets Two Distinct Intestinal Chloride Channels". Molecular Pharmacology. 77 (1): 69–78. doi:10.1124/mol.109.061051. PMC 2802429
 . PMID 19808995.
. PMID 19808995. - 1 2 H. Spreitzer (10 September 2012). "Neue Wirkstoffe – Crofelemer". Österreichische Apothekerzeitung (in German) (19/2012): 22.
- ↑ Clinical trial number NCT00547898 for "Safety and Effectiveness of 3 Doses of Crofelemer Compared to Placebo in the Treatment of HIV Associated Diarrhea (ADVENT)" at ClinicalTrials.gov
- 1 2 "FDA approves first anti-diarrheal drug for HIV/AIDS patients". FDA. Retrieved 31 December 2012.
- 1 2 3 Cottreau, J.; Tucker, A.; Crutchley, R.; Garey, K. W. (2012). "Crofelemer for the treatment of secretory diarrhea". Expert Review of Gastroenterology & Hepatology. 6 (1): 17–23. doi:10.1586/egh.11.87. PMID 22149578.
- ↑ Jones, K. (2003). "Review of Sangre de Drago (Croton lechleri) – A South American Tree Sap in the Treatment of Diarrhea, Inflammation, Insect Bites, Viral Infections, and Wounds: Traditional Uses to Clinical Research". The Journal of Alternative and Complementary Medicine. 9 (6): 877–896. doi:10.1089/107555303771952235. PMID 14736360.
- ↑ Vaisberg, A.; Milla, M.; Planas, M.; Cordova, J.; De Agusti, E.; Ferreyra, R.; Mustiga, M.; Carlin, L.; Hammond, G. (2007). "Taspine is the Cicatrizant Principle in Sangre de Grado Extracted from Croton lechleri". Planta Medica. 55 (2): 140–143. doi:10.1055/s-2006-961907. PMID 2748730.
- ↑ Ubillas, R.; Jolad, S.D.; Bruening, R.C.; Kernan, M.R.; King, S.R.; Sesin, D.F.; Barrett, M.; Stoddart, C.A.; Flaster, T.; et al. (1994). "SP-303, an antiviral oligomeric proanthocyanidin from the latex of Croton lechleri (sangre de drago)". Phytomedicine. 1 (2): 77–106. doi:10.1016/S0944-7113(11)80026-7. PMID 23195881.
- ↑ Safrin, S.; McKinley, G.; McKeough, M.; Robinson, D.; Spruance, S. L. (1994). "Treatment of acyclovir-unresponsive cutaneous herpes simplex virus infection with topically applied SP-303". Antiviral Research. 25 (3–4): 185–192. doi:10.1016/0166-3542(94)90002-7. PMID 7710268.
- ↑ Gabriel, S. E.; Davenport, S. E.; Steagall, R. J.; Vimal, V.; Carlson, T.; Rozhon, E. J. (1999). "A novel plant-derived inhibitor of cAMP-mediated fluid and chloride secretion". The American journal of physiology. 276 (1 Pt 1): G58–G63. PMID 9886979.
- ↑ "Glenmark gets an arbitration upshot on anti-diarrheal compound Crofelemer". BusinessLine.
- ↑ "Napo Terminates Salix for Breach of Agreement for Failure to Commercially Develop Crofelemer". BusinessWire. 10 November 2011.
- ↑ "What's Next In Line For Salix?". RTTNews. 27 August 2012.