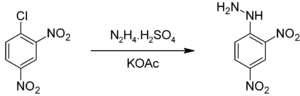2,4-Dinitrophenylhydrazine
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
(2,4-dinitrophenyl)hydrazine | |||
| Other names
2,4-DNPH; 2,4-DNP; Brady's reagent | |||
| Identifiers | |||
| 119-26-6 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:66932 | ||
| ChEMBL | ChEMBL352799 | ||
| ChemSpider | 3001507 | ||
| ECHA InfoCard | 100.003.918 | ||
| KEGG | C11283 | ||
| |||
| |||
| Properties | |||
| C6H6N4O4 | |||
| Molar mass | 198.14 g/mol | ||
| Appearance | Red or orange powder | ||
| Melting point | 198 to 202 °C (388 to 396 °F; 471 to 475 K) dec. | ||
| Slight | |||
| Hazards | |||
| Main hazards | Flammable, possibly carcinogenic | ||
| Safety data sheet | MSDS | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
2,4-Dinitrophenylhydrazine (DNPH, Brady's reagent) is the chemical compound C6H3(NO2)2NHNH2. Dinitrophenylhydrazine is a red to orange solid. It is a substituted hydrazine, and is often used to qualitatively test for carbonyl groups associated with aldehydes and ketones. The hydrazone derivatives can also be used as evidence toward the identity of the original compound. The melting point of the derivative is often used, with reference to a database of values, to determine the identity of a specific carbonyl compound. It is relatively sensitive to shock and friction; it is a shock explosive so care must be taken with its use. To reduce its explosive hazard, it is usually supplied wet.
Synthesis
2,4-Dinitrophenylhydrazine is commercially available usually as a wet powder. It can be prepared by the reaction of hydrazine sulfate with 2,4-dinitrochlorobenzene:[1]
Brady’s reagent is prepared by dissolving 2,4-dinitrophenylhydrazine in a solution containing methanol and some concentrated sulfuric acid.
Brady's test
2,4-Dinitrophenylhydrazine can be used to qualitatively detect the carbonyl functionality of a ketone or aldehyde functional group. A positive test is signaled by a yellow, orange or red precipitate (known as a dinitrophenylhydrazone.) If the carbonyl compound is aromatic, then the precipitate will be red; if aliphatic, then the precipitate will have a more yellow color.[2] The reaction between 2,4-Dinitrophenylhydrazine and a ketone is shown below:
- RR'C=O + C6H3(NO2)2NHNH2 → C6H3(NO2)2NHNCRR' + H2O
This reaction can be described as a condensation reaction, with two molecules joining together with loss of water. It is also considered an addition-elimination reaction: nucleophilic addition of the -NH2 group to the C=O carbonyl group, followed by the removal of a H2O molecule.
The mechanism for the reaction between 2,4-dinitrophenylhydrazine and an aldehyde or ketone is shown below:[3]
Crystals of different hydrazones have characteristic melting and boiling points, allowing the identity of a substance to be determined in a method known as derivatization. In particular, the use of 2,4-dinitrophenylhydrazine was developed by Brady and Elsmie.[4] Modern spectroscopic and spectrometric techniques have since superseded these techniques.

Dinitrophenylhydrazine does not react with other carbonyl-containing functional groups such as carboxylic acids, amides, and esters. For carboxylic acids, amides and esters, there is resonance associated stability as a lone-pair of electrons interacts with the p-orbital of the carbonyl carbon resulting in increased delocalization in the molecule. This stability would be lost by addition of a reagent to the carbonyl group. Hence, these compounds are more resistant to addition reactions. Also with carboxylic acids there is the effect of the compound acting as a base, leaving the resulting carboxylate negatively charged hence unable to be attacked by this nucleophile.
See also
References
- ↑ Allen, C. F. H. (1943). "2,4-Dinitrophenylhydrazine". Org. Synth.; Coll. Vol., 2, p. 228
- ↑ http://wiki.colby.edu/download/attachments/110920618/Experiment+%232.pdf?version=1&modificationDate=1265312071267
- ↑ Adapted from Chemistry in Context, 4th Edition, 2000, Graham Hill and John Holman
- ↑ Brady, O. L.; Elsmie, G. V. (1926). "The use of 2:4-dinitrophenylhydrazine as a reagent for aldehydes and ketones". Analyst. 51 (599): 77–78. Bibcode:1926Ana....51...77B. doi:10.1039/AN9265100077.