Cold urticaria
| Cold urticaria | |
|---|---|
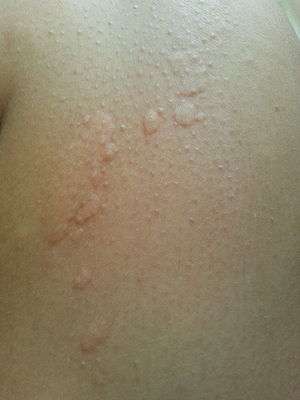 | |
| Allergic urticaria on leg in the form of hives induced by cold. | |
| Classification and external resources | |
| Specialty | dermatology |
| ICD-10 | L50.2 (ILDS L50.210) |
| ICD-9-CM | 708.2 |
| OMIM | 120100 |
| DiseasesDB | 4688 |
Cold urticaria (essentially meaning "cold hives") is a disorder where hives (urticaria) or large red welts form on the skin after exposure to a cold stimulus.[1] The welts are usually itchy and often the hands and feet will become itchy and swollen as well. Hives vary in size from about 7mm in diameter to as big as about 27mm diameter or larger. The disease is classified as chronic when hives appear for longer than 6 weeks; they can last for life, though their course is often unpredictable. This disorder, or perhaps two disorders with the same clinical manifestations, can be inherited (familial cold urticaria) or acquired[2] (primary acquired cold urticaria). The acquired form is most likely to occur between ages 18–25, although it can occur as early as 5 years old in some cases.
Types
Cold urticaria may be divided into the following types:[3]
Primary cold contact urticaria
Primary cold contact urticaria is a cutaneous condition characterized by weals, and occurs in rainy, windy weather, and after swimming in cold water and after contact with cold objects, including ice cubes.[3]
Secondary cold contact urticaria
Secondary cold contact urticaria is a cutaneous condition characterized by weals, due to serum abnormalities such as cryoglobulinemia or cryofibrinogenemia are extremely rare, and are then associated with other manifestations such as Raynaud's phenomenon or purpura.[3]
Reflex cold urticaria
Reflex cold urticaria is a cutaneous condition in which generalized cooling of the body induces widespread wealing.[3]
Familial cold urticaria
| Familial cold urticaria | |
|---|---|
| Classification and external resources | |
| Specialty | dermatology |
| ICD-10 | L50.2 (ILDS L50.220) |
| OMIM | 120100 |
| MeSH | D056587 |
Familial cold urticaria (also properly known as familial cold autoinflammatory syndrome, FCAS) is an autosomal dominant condition characterized by rash, conjunctivitis, fever/chills and arthralgias[4] elicited by exposure to cold - sometimes temperatures below 22 °C (72 °F).[3][5]
It has been mapped to CIAS1[6] and is a slightly milder member of the disease family including Muckle–Wells syndrome and NOMID. It is rare and is estimated as having a prevalence of 1 per million people and mainly affects Americans and Europeans.[7]
FCAS is one of the cryopyrin-associated periodic syndromes (CAPS) caused by mutations in the CIAS1/NALP3 (aka NLRP3) gene at location 1q44.[8][9][10] The disease was described in The Lancet Volume 364[11] by Hoffman H.M.[12] et al.
The effect of FCAS on the quality of life of patients is far reaching. A survey of patients in the United States in 2008 found, "To cope with their underlying disease and to try to avoid symptomatic, painful, flares patients reported limiting their work, school, family, and social activities. Seventy-eight percent of survey participants described an impact of the disease on their work, including absenteeism and impaired job advancement; frequently, they quit their job as a consequence of their disease".[13]
Treatment using anakinra (Kineret) has been shown effective for FCAS, although this does mean daily injections of the immunosuppressant into an area such as the lower abdomen.[14][15] The monoclonal antibody canakinumab (Ilaris) is also used.[16]
Symptoms

When the body is exposed to the cold in individuals afflicted by the condition, hives appear and the skin in the affected area typically becomes itchy. Hives result from dilation of capillaries which allow fluid to flow out into the surrounding tissue which is the epidermis.They resolve when the body absorbs this fluid.[17] The border of a hive is described as polycyclic, or made up of many circles, and changes as fluid leaks out and then is absorbed.[18] Pressing on a hive causes the skin to blanch distinguishing it from a bruise or papule. Hives can last for a few minutes or a few days, and vary from person to person. Also a burning sensation occurs. During a severe reaction, hypotension, which can be life-threatening, can occur. A serious reaction is most likely to occur if the hives occur with less than 3 minutes of exposure (during a cold test).[19]
Cause
The hives are a histamine reaction in response to cold stimuli, including a drastic drop in temperature, cold air, and cold water. There are many causes for cold hives, most are idiopathic (meaning they have no known cause). Some rare conditions can cause cold hives, and it can be useful to test for these conditions if the cold hives are in any way unusual.
Scientists from the USA National Institutes of Health have identified a genetic mutation in three unrelated families that causes a rare immune disorder characterized by excessive and impaired immune function: immune deficiency, autoimmunity, inflammatory skin disorders and cold-induced hives (cold urticaria).
"The mutation discovered occurs in a gene for phospholipase C-gamma2 (PLCG2), an enzyme involved in the activation of immune cells. The investigators have named the condition PLCG2-associated antibody deficiency and immune dysregulation, or PLAID."[20]
Diagnosis
Diagnosis is typically obtained by an allergist or other licensed practitioner performing a cold test. During the cold test, a piece of ice is held against the forearm, typically for 3–4 minutes. A positive result is a specific looking mark of raised red hives. The hives may be the shape of the ice, or it may radiate from the contact area of the ice.[21]
" However, while these techniques assist in diagnosis, they do not provide information about temperature and stimulation time thresholds at which patients will start to develop symptoms."[21]
which is essential because it can establish disease severity and monitor the effectiveness of treatment.[21]
Treatment
The first-line therapy in ColdU, as recommended by EAACI/GA2 LEN/EDF/WAO guidelines, is symptomatic relief with second-generation H1- antihistamines. if standard doses are ineffective increasing up to 4-fold is recommended to control symptoms.[22]
The second-generation H1-antihistamine, rupatadine, was found to significantly reduced the development of chronic cold urticaria symptom without an increase in adverse effects using 20 and 40 mg.[21]
Allergy medications containing antihistamines such as diphenhydramine (Benadryl), cetirizine (Zyrtec), loratidine (Claritin), cyproheptadine (Periactin), and fexofenadine (Allegra) may be taken orally to prevent and relieve some of the hives (depending on the severity of the allergy).[23] For those who have severe anaphylactic reactions, a prescribed medicine such as doxepin, which is taken daily, should help to prevent and/or lessen the likelihood of a reaction and thus, anaphylaxis. There are also topical antihistamine creams which are used to help relieve hives in other conditions, but there is not any documentation stating it will relieve hives induced by cold temperature.
Cold hives can result in a potentially serious, or even fatal, systemic reaction (anaphylactic shock). People with cold hives may have to carry an injectable form of epinephrine (like Epi-pen or Twinject) for use in the event of a serious reaction. The best treatment for this allergy is avoiding exposure to cold temperature.[24]
Ebastine has been proposed as an approach to prevent acquired cold urticaria.[25]
Situations when reactions may occur
- Cold weather/Snow: Exposure to cold or cool air for more than a few minutes can trigger a reaction.
- Sweat: A reaction may even occur on a warm day when there is sweat on the skin. If there is a breeze it will rapidly cool the skin and create hives.
- Cold/Cool IV Lines: While in the hospital, inserting IV lines that have been refrigerated or are cool into individuals who are allergic to cold temperature can/may result in pain within the limb the IV is placed and a line of hives may appear within a radius of 6-8 inches from the IV site in the pattern of a line going up the limb.
- Swimming: Swimming can be especially dangerous as the rapid heart rate combined with the onset of hives can lead to hypotension.
- Air Conditioning: Entering a cool building during a summer day can result in an allergic reaction to exposed areas of skin, congestion from breathing the cool air and possibly feeling fatigued.
- Cold Foods/Drinks: Eating or drinking cold or cool substances such as ice cream or even iced tea may result in the individuals tongue and surrounding tissues swelling.
- Cool/Cold Surfaces: Sitting on sidewalks which are cool, leaning on or grabbing a cold pole will result in hives forming on the area which had contact with the cool surfaces.
- Restriction of Blood Flow: Typing or other activities which tense muscles and reduce blood flow can cool the body parts enough to cause itching and hives.
- Blood Flow: While putting hands or feet in hot water nails turn purple/white until the body gets used to the temperature.
See also
- Amyloidosis
- Cholinergic urticaria, a similar hives reaction in response to heat
- Diascopy
- Erythema
- Goose bumps, an unrelated, normal response to cold temperatures
- List of cutaneous conditions
- Skin lesion
References
- ↑ Feinberg, Jeff H., and Charles B. Toner. "Successful Treatment of Disabling Cholinergic Urticaria." Military Medicine 173.2 (2008): 217-20. Web
- ↑ Siebenhaar F, Weller K, : clinical picture and update on diagnosis and treatment (May 2007). "Acquired cold urticaria: Clinical picture and update on diagnosis and treatment". Clin. Exp. Dermatol. 32 (3): 241–5. doi:10.1111/j.1365-2230.2007.02376.x. PMID 17355280.
- 1 2 3 4 5 Rapini, Ronald P.; Bolognia, Jean L.; Jorizzo, Joseph L. (2007). Dermatology: 2-Volume Set. St. Louis: Mosby. pp. 267–8. ISBN 1-4160-2999-0.
- ↑ Tunca, Ozdogan, Mehmet, Huri. "Molecular and Genetic Characteristics of Hereditary Autoinflammatory" (PDF). Molecular and Genetic Characteristics of Hereditary Autoinflammatory. Betham Science. Retrieved 12 April 2011.
- ↑ James, William; Berger, Timothy; Elston, Dirk (2005). Andrews' Diseases of the Skin: Clinical Dermatology. (10th ed.). Saunders. ISBN 0-7216-2921-0.
- ↑ Hoffman HM, Mueller JL, Broide DH, Wanderer AA, Kolodner RD (November 2001). "Mutation of a new gene encoding a putative pyrin-like protein causes familial cold autoinflammatory syndrome and Muckle-Wells syndrome". Nat. Genet. 29 (3): 301–5. doi:10.1038/ng756. PMID 11687797.
- ↑ Home Reference, Genetics. "Familial cold autoinflammatory syndrome". Familial cold autoinflammatory syndrome. U.S. National Library of Medicine. Retrieved 12 April 2011.
- ↑ CAPS, Community (2008-01-01). "Familial Cold Auto-inflammatory Syndrome (FCAS): Fact Sheet". Regeneron Pharmaceuticals. Retrieved 2010-02-01.
- ↑ HGNC, HUGO. "Gene Name Database". Gene Name Database. Wellcome Foundation.
- ↑ Protein Data Bank (PDB), RSCB. "1q44". Crystal Structure of an Arabidopsis Thaliana Putative Steroid Sulphotransferase. RCSB. Retrieved 12 April 2011.
- ↑ Hoffman H.M., Vol 364; et al. "Prevention of cold-associated acute inflammation in familial cold autoinflammatory syndrome by interleukin-1 receptor antagonist". Lancet Vol 364. The Lancet.
- ↑ Hoffman, Harold. "Associate Professor of Pediatrics and Medicine, UC San Diego". The prevention of cold-associated acute inflammation in cold auto-inflammatory syndrome by interleukin-1 receptor antagonist. The Lancet vol 364.
- ↑ Stych, B; Dobrovolny, D (2008). "Familial cold auto-inflammatory syndrome (FCAS): characterization of symptomatology and impact on patients' lives.". Curr Med Res Opin (Tarrytown, NY, USA : 2008). 24 (6): 1577–82. doi:10.1185/03007990802081543. PMID 18423104.
- ↑ Ross JB; Finlayson, LA; Klotz, PJ; Langley, RG; Gaudet, R; Thompson, K; Churchman, SM; McDermott, MF; Hawkins, PN; et al. (2010-02-01). "Use of anakinra (Kineret) in the treatment of familial cold autoinflammatory syndrome with a 16-month follow-up". Journal of cutaneous medicine and surgery. 12 (1): 8–16. PMID 18258152.
- ↑ Samy K Metyas, Hal M Hoffman (2008-02-01). "Anakinra prevents symptoms of familial cold autoinflammatory syndrome and Raynaud's disease.". Journal of Rheumatology. Retrieved 2010-02-01.
- ↑ Walsh, GM (2009). "Canakinumab for the treatment of cryopyrin-associated periodic syndromes". Drugs of today (Barcelona, Spain : 1998). 45 (10): 731–5. doi:10.1358/dot.2009.45.10.1436882. PMID 20069137.
- ↑ citation needed
- ↑ Bailey, Evan, and Marcus Shaker. "An Update on Childhood Urticaria and Angioedema." Current Opinion in Pediatrics 20.4 (2008): 425-30. Web.
- ↑ Soter, Nicholas A., Stephen I. Wasserman, and K. Frank Austen. "Cold Urticaria: Release into the Circulation of Histamine and Eosinophil Chemotactic Factor of Anaphylaxis during Cold Challenge." New England Journal of Medicine N Engl J Med 294.13 (1976): 687-90. Web.
- ↑ News published on the National Institute of Allergy and Infectious Diseases (NIAID) website (Jan. 11, 2012)
- 1 2 3 4 Abajian, M., L. Curto-Barredo, K. Krause, E. Santamaria, I. Izquierdo, M. Church, M. Maurer, and A. Giménez-Arnau. "Rupatadine 20 Mg and 40 Mg Are Effective in Reducing the Symptoms of Chronic Cold Urticaria." Acta Dermato Venereologica Acta Derm Venerol 96.1 (2016): 56-59. Web.
- ↑ Zuberbier T, Aberer W, Asero R, Bindslev-Jensen C, Brzoza Z, Canonica GW, et al. The EAACI/GA(2) LEN/EDF/WAO Guideline for the definition, classification, diagnosis, and management of urticaria: the 2013 revision and update. Allergy 2014; 69: 868–887.
- ↑ Mahmoudi M (May 2001). "Cold-induced urticaria". J Am Osteopath Assoc. 101 (5 Suppl): S1–4. PMID 11409259.
- ↑ "Cold urticaria - Mayo Clinic". www.mayoclinic.org. Retrieved 2016-05-04.
- ↑ Magerl M, Schmolke J, Siebenhaar F, Zuberbier T, Metz M, Maurer M (December 2007). "Acquired cold urticaria symptoms can be safely prevented by ebastine". Allergy. 62 (12): 1465–8. doi:10.1111/j.1398-9995.2007.01500.x. PMID 17900265.