Gene–environment interaction
Gene–environment interaction (or genotype–environment interaction or G×E) is when two different genotypes respond to environmental variation in different ways. A norm of reaction is a graph that shows the relationship between genes and environmental factors when phenotypic differences are continuous.[1] They can help illustrate GxE interactions. When the norm of reaction is not parallel, as shown in the figure below, there is a gene by environment interaction. This indicates that each genotype responds to environmental variation in a different way.
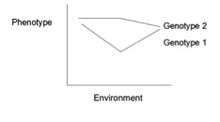
Gene–environment interactions are studied to gain a better understanding of various phenomena. In genetic epidemiology, gene-environment interactions are useful for understanding some diseases. Sometimes, sensitivity to environmental risk factors for a disease are inherited rather than the disease itself being inherited. Individuals with different genotypes are affected differently by exposure to the same environmental factors, and thus gene-environment interactions can result in different disease phenotypes. For example, sunlight exposure has a stronger influence on skin cancer risk in fair-skinned humans than in individuals with darker skin.[2]
Nature versus nurture debates assume that variation in a trait is primarily due to either genetic differences or environmental differences. However, the current scientific opinion holds that neither genetic differences nor environmental differences are solely responsible for producing phenotypic variation, and that virtually all traits are influenced by both genetic and environmental differences.[3][4][5] Statistical analysis of the genetic and environmental differences contributing to the phenotype would have to be used to confirm these as gene-environment interactions.
Definitions
There are two different conceptions of gene–environment interaction. Tabery[6] has labeled them biometric and developmental interaction, while Sesardic[7] uses the terms statistical and commonsense interaction.
The biometric (or statistical) conception has its origins in research programs that seek to measure the relative proportions of genetic and environmental contributions to phenotypic variation within populations. Biometric gene–environment interaction has particular currency in population genetics and behavioral genetics.[6] Any interaction results in the breakdown of the additivity of the main effects of heredity and environment, but whether such interaction is present in particular settings is an empirical question. Biometric interaction is relevant in the context of research on individual differences rather than in the context of the development of a particular organism.[8]
Developmental gene–environment interaction is a concept more commonly used by developmental geneticists and developmental psychobiologists. Developmental interaction is not seen merely as a statistical phenomenon. Whether statistical interaction is present or not, developmental interaction is in any case manifested in the causal interaction of genes and environments in producing an individual's phenotype.[8]
Examples
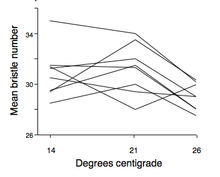
- In Drosophila: A classic example of gene–environment interaction was performed on drosophila by Gupta and Lewontin in 1981. In their experiment they demonstrated that the mean bristle number on drosophila could vary with changing temperatures. As seen in the graph to the right, different genotypes reacted differently to the changing environment. Each line represents a given genotype, and the slope of the line reflects the changing phenotype (bristle number) with changing temperature. Some individuals had an increase in bristle number with increasing temperature while others had a sharp decrease in bristle number with increasing temperature. This showed that the norms of reaction were not parallel for these flies, proving that gene-environment interactions exist.[9]
- In plants: Seven genetically distinct yarrow plants were collected and three cuttings taken from each plant. One cutting of each genotype was planted at low, medium, and high elevations, respectively. When the plants matured, no one genotype grew best at all altitudes, and at each altitude the seven genotypes fared differently. For example, one genotype grew the tallest at the medium elevation but attained only middling height at the other two elevations. The best growers at low and high elevation grew poorly at medium elevation. The medium altitude produced the worst overall results, but still yielded one tall and two medium-tall samples. Altitude had an effect on each genotype, but not to the same degree nor in the same way.[10]
- Phenylketonuria (PKU) is a human genetic condition caused by mutations to a gene coding for a particular liver enzyme. In the absence of this enzyme, an amino acid known as phenylalanine does not get converted into the next amino acid in a biochemical pathway, and therefore too much phenylalanine passes into the blood and other tissues. This disturbs brain development leading to mental retardation and other problems. PKU affects approximately 1 out of every 15,000 infants in the U.S. However, most affected infants do not grow up impaired because of a standard screening program used in the U.S. and other industrialized societies. Newborns found to have high levels of phenylalanine in their blood can be put on a special, phenylalanine-free diet. If they are put on this diet right away and stay on it, these children avoid the severe effects of PKU.[11] This example shows that a change in environment (lowering Phenylalanine consumption) can affect the phenotype of a particular trait, demonstrating a gene-environment interaction.
- A single nucleotide polymorphism rs1800566 in NAD(P)H Quinone Dehydrogenase 1 (NQO1) alters the risk of asthma and general lung injury upon interaction with NOx pollutants, in individuals with this mutation. [12] [13]
- A functional polymorphism in the monoamine oxidase A (MAOA) gene promoter can moderate the association between early life trauma and increased risk for violence and antisocial behavior. Low MAOA activity is a significant risk factor for aggressive and antisocial behavior in adults who report victimization as children. Persons who were abused as children but have a genotype conferring high levels of MAOA expression are less likely to develop symptoms of antisocial behavior.[14] These findings must be interpreted with caution, however, because gene association studies on complex traits are notorious for being very difficult to confirm.[15]
- In Drosophila Eggs: Contrary to the aforementioned examples, length of egg development in drosophila as a function of temperature demonstrates the lack of gene-environment interactions. The attached graph shows parallel reaction norms for a variety of individual drosophila flies, showing that there is not a gene-environment interaction present between the two variables. In other words, each genotype responds similarly to the changing environment producing similar phenotypes. For all individual genotypes, average egg development time decreases with increasing temperature. The environment is influencing each of the genotypes in the same predictable manner.[9]
 Egg Development Time by Temperature
Egg Development Time by Temperature
Medical significance
- Doctors are interested in knowing whether disease can be prevented by reducing exposure to environmental risks. Some people carry genetic factors that confer susceptibility or resistance to a certain disorder in a particular environment. The interaction between the genetic factors and environmental stimulus is what results in the disease phenotype.[16] There may be significant public health benefits in using gene by environment interactions to prevent or cure disease.[17]
- An individual’s response to a drug can result from various gene by environment interactions.[16] Therefore, the clinical importance of pharmacogenetics and gene by environment interactions comes from the possibility that genomic, along with environmental information, will allow more accurate predictions of an individual’s drug response. This would allow doctors to more precisely select a certain drug and dosage to achieve therapeutic response in a patient while minimizing side effects and adverse drug reactions.[18] This information could also help to prevent the health care costs associated with adverse drug reactions and inconveniently prescribing drugs to patients who likely won’t respond to them.[16]
- In a similar manner, an individual can respond to other environmental stimuli, factors or challenges differently according to specific genetic differences or alleles. These other factors include the diet and specific nutrients within the diet, physical activity, alcohol and tobacco use, sleep (bed time, duration), and any of a number of exposures (or exposome), including toxins, pollutants, sunlight (latitude north/south of the equator), among any number of others. The diet, for example, is modifiable and has significant impact on a host of cardiometabolic diseases, including cardiovascular disease, coronary artery disease, coronary heart disease, type 2 diabetes, hypertension, stroke, myocardial infarction, and non-alcoholic fatty liver disease. In the clinic, typically assessed risks of these conditions include blood lipids (triglyceride, and HDL, LDL and total cholesterol), glycemic traits (plasma glucose and insulin, HOMA-IR, beta cell function as HOMA-BC), obesity anthropometrics (BMI/obesity, adiposity, body weight, waist circumference, waist-to-hip ratio), vascular measures (diastolic and systolic blood pressure), and biomarkers of inflammation. Gene-Environment interactions can modulate the adverse effects of an allele that confers increased risk of disease, or can exacerbate the genotype-phenotype relationship and increase risk, in a manner often referred to as nutrigenetics.[19] A catalog of genetic variants that associate with these and related cardiometabolic phenotypes and modified by common environmental factors is available.[20]
See also
- Biopsychosocial model
- Diathesis–stress model
- Differential susceptibility
- Envirome
- Epidemiology
- Epigenetics
- Evolutionary developmental psychology
- Exposome
- Gene-environment correlation
- Genetic epidemiology
- Genomics
- Molecular epidemiology
- Molecular pathological epidemiology
- Molecular pathology
References
- ↑ Krebs, J.R. (April 2, 2012). An Introduction to Behavioural Ecology. Oxford: Wiley-Blackwell. ISBN 1405114169.
- ↑ Green A; Trichopoulos D (2002). Skin cancer. In Textbook of Cancer Epidemiology (eds Adami, H., Hunter, D. & Trichopoulos, D.) pp. 281–300. Oxford: Oxford University Press.
- ↑ Ridley, M. (2003) Nature via Nurture: Genes, Experience, & What Makes Us Human. Harper Collins. ISBN 0-00-200663-4
- ↑ Rutter, Michael. (2006) Genes and Behavior: Nature-Nurture Interplay Explained Oxford, UK: Blackwell Publishers
- ↑ Cuhna, Flavio and James J. Heckman Investing in Our Young People, in A. J. Reynolds, A. Rolnick, M. M. Englund, & J. Temple, eds., Cost-effective Early Childhood Programs in the First Decade: A Human Capital Integration, Chapter 18, pp. 381-414, 2010, New York: Cambridge University Press
- 1 2 Tabery, J (2007). "Biometric and developmental gene-environment interactions: Looking back, moving forward". Development and Psychopathology. 19: 961–976. doi:10.1017/s0954579407000478.
- ↑ Sesardic, N. (2005). Making sense of heritability. Cambridge: Cambridge University Press, p. 48.
- 1 2 Tabery, James and Griffiths, Paul E. (2010). Historical and Philosophical Perspectives on Behavioral Genetics and Developmental Science", in Hood, Halpern, Greenberg, and Lerner (Eds.), Handbook of Developmental Science, Behavior, and Genetics. Wiley-Blackwell, pp. 41-60.
- 1 2 Anand P. Gupta; R. C. Lewontin (1982). "A Study of Reaction Norms in Natural Populations of Drosophila pseudoobscura". Evolution. 36 (5): 934–948. doi:10.2307/2408073.
- ↑ Clausen J, Keck D, Hiesey WM (1948). "Experimental studies on the nature of species. III. Environmental responses of climatic races of Achillea, Carnegie Inst Washington Publ 581": 1–129.
- ↑ AAAS publication on Behavioral Genetics
- ↑ Castro-Giner, F., Künzli, N., Jacquemin, B., Forsberg, B., de Cid, R., Sunyer, J., Jarvis, D., Briggs, D., Vienneau, D., Norback, D., González, J. R. (2009). Traffic-related air pollution, oxidative stress genes, and asthma (ECHRS). Environmental health perspectives, 1919-1924.
- ↑ Basharat, Z., Messaoudi, A., Ruba, S., Yasmin, A. (2016). NQO1 rs1800566 polymorph is more prone to NOx induced lung injury: Endorsing deleterious functionality through informatics approach. Gene.591(1):14-20.
- ↑ Caspi A, et al. (2002). "Role of genotype in the cycle of violence in maltreated children". Science. 297 (5582): 851–854. doi:10.1126/science.1072290. PMID 12161658.
- ↑ Munafò M, et al. (2009). "Gene x Environment Interactions at the Serotonin Transporter Locus". Biol Psychiatry. 65: 211–219. doi:10.1016/j.biopsych.2008.06.009. PMID 18691701.
- 1 2 3 Haga, Susanne; Burke, Wylie (2004). "Using Pharmacogenetics to Improve Drug Safety and Efficacy.". JAMA. 291 (23): 2869–2871. doi:10.1001/jama.291.23.2869.
- ↑ Khoury MJ, Davis R, Gwinn M, Lindegren ML, Yoon P (2005). "Do we need genomic research for the prevention of common diseases with environmental causes?". Am J Epidemiol. 161 (9): 799–805. doi:10.1093/aje/kwi113. PMID 15840611.
- ↑ Eichelbaum, Michel; Ingelman-Sundberg, Magnus; Evans, William (2006). "Pharmacogenomics and Individualized Drug Therapy". Annual Review of Medicine. 57: 119–137. doi:10.1146/annurev.med.56.082103.104724.
- ↑ Ordovas, JM (2008). "Genotype-phenotype associations: modulation by diet and obesity". Obesity. 16: S40–S46. doi:10.1038/oby.2008.515. PMID 19037211.
- ↑ Parnell, LD; Blokker, BA; Dashti, HS; et al. (2014). "CardioGxE, a catalog of gene-environment interactions for cardiometabolic traits". BioData Mining. 7: 21. doi:10.1186/1756-0381-7-21.