Hydroxy group
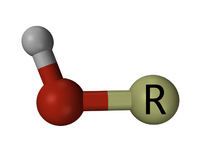
A hydroxyl or hydroxy group is a chemical functional group containing one oxygen atom connected by a covalent bond to one hydrogen atom (−OH). It is sometimes called the alcohol functional group because when bonded to carbon in a molecule that otherwise contains only hydrogen and carbon the hydroxy (not hydroxyl) group defines the molecule as an alcohol, resulting in a name ending in -ol. A hydroxyl group bonded covalently to the carbon of a carbonyl group (C=O) produces a carboxyl group (C(O)OH) that is the defining group of a carboxylic acid. When the −OH group participates in an ionic bond, the [OH−] anion is called the hydroxide ion. As a free radical, it is the hydroxyl radical.
Hydroxy group
According to IUPAC rules the term hydroxyl refers to the radical OH only, while the functional group −OH is called hydroxy group.[1]
The oxygen atom in the hydroxy group is much more electronegative than the hydrogen, so that the electrons are closer to the oxygen atom. Similar to water, compounds containing the hydroxy group are capable of forming hydrogen bonds. Primary alcohols are miscible with water. In complex compounds, the portion of the molecule containing the hydroxy group is hydrophilic.
The hydroxy group appears throughout organic chemical structure and biochemical structure. It appears covalently bonded to carbon in sugars. The hydroxy group appears on the second carbon atom of the hydroxy amino acids serine and threonine. It appears on the phenol side chain of tyrosine. The capacity of the hydroxy group to form hydrogen bonds imparts water solubility and internal structural stability to proteins that include these amino acids.
Hydroxy groups participate in the dehydration reactions that link simple biological molecules into long chains. The joining of a fatty acid to glycerol to form a triacylglycerol removes the −OH from the carboxy end of the fatty acid. The joining of two aldehyde sugars to form a disaccharide removes the −OH from the carboxy group at the aldehyde end of one sugar. The creation of a peptide bond to link two amino acids to make a protein removes the −OH from the carboxy group of one amino acid.
Hydroxyl radical
Hydroxyl radicals are highly reactive and undergo chemical reactions that make them short-lived. When biological systems are exposed to hydroxyl radicals, they can cause damage to cells, including those in humans, where they react with DNA, lipids, and proteins.
Lunar and other extraterrestrial observations
In 2009, India's Chandrayaan-1 satellite, NASA's Cassini spacecraft and the Deep Impact probe have each detected the presence of water by evidence of hydroxyl fragments on the Moon. As reported by Richard Kerr, "A spectrometer [the Moon Mineralogy Mapper, a.k.a. "M3"] detected an infrared absorption at a wavelength of 3.0 micrometers that only water or hydroxyl—a hydrogen and an oxygen bound together—could have created."[2] NASA also reported in 2009 that the LCROSS probe revealed an ultraviolet emission spectrum consistent with hydroxyl presence.[3] The Venus Express orbiter has been continuously sending back Venus science data since April 11, 2006. Results from Venus Express include the detection of hydroxyl in the atmosphere.
See also
| Look up hydroxy group in Wiktionary, the free dictionary. |
| Wikimedia Commons has media related to Hydroxyl group. |
References
- ↑ "Alcohols". IUPAC. Retrieved 23 March 2015.
- ↑ Richard A. Kerr (24 September 2009). "A Whiff of Water Found on the Moon". Science Now. Retrieved 2016-06-01.
- ↑ Jonas Dino (13 November 2009). "LCROSS Impact Data Indicates Water on Moon". NASA. Retrieved 2009-11-14.
External links
- Reece, Jane; Urry, Lisa; Cain, Michael; Wasserman, Steven; Minorsky, Peter; Jackson, Robert (2011). "Unit 1, Chapter 4 &5." In Campbell Biology (9th ed.). Berge, Susan; Golden, Brandy; Triglia, Logan (eds.). San Francisco: Pearson Benjamin Cummings. ISBN 978-0-321-55823-7