Isotopes of darmstadtium
Darmstadtium (Ds) is a synthetic element, and thus a standard atomic mass cannot be given. Like all synthetic elements, it has no stable isotopes. The first isotope to be synthesized was 269Ds in 1994. There are 7 or 8 known radioisotopes from 267Ds (or 269Ds) to 281Ds (with many gaps) and 2 or 3 known isomers. The longest-lived isotope is 281Ds with a half-life of 9.6 seconds.
Table
| nuclide symbol |
Z(p) | N(n) | isotopic mass (u) |
half-life | decay mode(s)[n 1] |
daughter isotope(s) |
nuclear spin |
|---|---|---|---|---|---|---|---|
| excitation energy | |||||||
| 267Ds[n 2] | 110 | 157 | 267.14377(15)# | 3(+6−2) µs | 9/2+# | ||
| 269Ds | 110 | 159 | 269.14475(3) | 230(110) µs [179(+245−66) µs] |
α | 265Hs | 3/2+# |
| 270Ds | 110 | 160 | 270.14458(5) | 160(100) µs [0.10(+14−4) ms] |
α | 266Hs | 0+ |
| 270mDs | 1140(70) keV | 10(6) ms [6.0(+82−22) ms] |
α | 266Hs | (10)(−#) | ||
| 271Ds | 110 | 161 | 271.14595(10)# | 210(170) ms | α | 267Hs | 11/2−# |
| 271mDs | 29(29) keV | 1.3(5) ms | α | 267Hs | 9/2+# | ||
| 273Ds | 110 | 163 | 273.14856(14)# | 0.17(+17−6) ms | α | 269Hs | 13/2−# |
| 277Ds[n 3] | 110 | 167 | 277.15591(41)# | 4.1 ms[1] | α | 273Hs | 11/2+# |
| 279Ds[n 4] | 110 | 169 | 279.16010(64)# | 0.18(+5−3) s | SF (90%) | (various) | |
| α (10%) | 275Hs | ||||||
| 280Ds[n 5] | 110 | 170 | 280.16131(89)# | << 1 s[2] | 0+ | SF | (various) |
| 281Ds[n 6] | 110 | 171 | 281.16451(59)# | 9.6 s | SF (94%) | (various) | 3/2+# |
| α (6%) | 277Hs | ||||||
| 281mDs[n 7][n 6] | 3.7 min | α | 277mHs | ||||
- ↑ Abbreviations:
SF: Spontaneous fission - ↑ Unconfirmed isotope
- ↑ Not directly synthesized, occurs in decay chain of 285Fl
- ↑ Not directly synthesized, occurs as decay product of 283Cn
- ↑ Not directly synthesized, occurs in decay chain of 288Mc
- 1 2 Not directly synthesized, occurs in decay chain of 289Fl
- ↑ This isomer is unconfirmed
Notes
- Values marked # are not purely derived from experimental data, but at least partly from systematic trends. Spins with weak assignment arguments are enclosed in parentheses.
- Uncertainties are given in concise form in parentheses after the corresponding last digits. Uncertainty values denote one standard deviation, except isotopic composition and standard atomic mass from IUPAC, which use expanded uncertainties.
Isotopes and nuclear properties
Nucleosynthesis
Super-heavy elements such as darmstadtium are produced by bombarding lighter elements in particle accelerators that induce fusion reactions. Whereas most of the isotopes of darmstadtium can be synthesized directly this way, some heavier ones have only been observed as decay products of elements with higher atomic numbers.[3]
Depending on the energies involved, the former are separated into "hot" and "cold". In hot fusion reactions, very light, high-energy projectiles are accelerated toward very heavy targets (actinides), giving rise to compound nuclei at high excitation energy (~40–50 MeV) that may either fission or evaporate several (3 to 5) neutrons.[4] In cold fusion reactions, the produced fused nuclei have a relatively low excitation energy (~10–20 MeV), which decreases the probability that these products will undergo fission reactions. As the fused nuclei cool to the ground state, they require emission of only one or two neutrons, and thus, allows for the generation of more neutron-rich products.[3] The latter is a distinct concept from that of where nuclear fusion claimed to be achieved at room temperature conditions (see cold fusion).[5]
Cold fusion
Before the first successful synthesis of darmstadtium in 1994 by the GSI team, scientists at GSI also tried to synthesize darmstadtium by bombarding lead-208 with nickel-64 in 1986. No darmstadtium atoms were identified. After an upgrade of their facilities, the team at GSI successfully detected 9 atoms of 271Ds in two runs of their discovery experiment in 1994.[6] This reaction was successfully repeated in 2000 by GSI (4 atoms), in 2000[7][8] and 2004[9] by the Lawrence Berkeley National Laboratory (LBNL) (9 atoms in total) and in 2002 by RIKEN (14 atoms).[10] The GSI team studied the analogous reaction with nickel-62 instead of nickel-64 in 1994 as part of their discovery experiment. Three atoms of 269Ds were detected.[6] A fourth decay chain was measured but was subsequently retracted.[11]
In addition to the official discovery reactions, in October–November 2000, the team at GSI also studied the analogous reaction using a lead-207 target in order to synthesize the new isotope 270Ds. They succeeded in synthesising 8 atoms of 270Ds, relating to a ground state isomer, 270Ds, and a high-spin metastable state, 270mDs.[12]
In 1986, a team at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, studied the reaction:
- 209
83Bi + 59
27Co → 267
110Ds + 1
0n
They were unable to detect any darmstadtium atoms. In 1995, the team at LBNL reported that they had succeeded in detecting a single atom of 267Ds using this reaction. However, several decays were not measured and further research is required to confirm this discovery.[13]
Hot fusion
In September 1994, the team at Dubna detected a single atom of 273Ds by bombarding a plutonium-244 target with accelerated sulfur-34 nuclei.[14]
Experiments have been performed in 2004 at the Flerov Laboratory of Nuclear Reactions in Dubna studying the fission characteristics of the compound nucleus 280Ds, produced through the nuclear reaction:
- 232
90Th + 48
20Ca → 280
110Ds* → fission
The result revealed how compound nuclei such as this fission predominantly by expelling magic and doubly magic nuclei such as 132Sn (Z=50, N=82). No darmstadtium atoms were obtained.[15] A compound nucleus is a loose combination of nucleons that have not arranged themselves into nuclear shells yet. It has no internal structure and is held together only by the collision forces between the target and projectile nuclei. It is estimated that it requires around 10−14 s for the nucleons to arrange themselves into nuclear shells, at which point the compound nucleus becomes a nuclide, and this number is used by IUPAC as the minimum half-life a claimed isotope must have to potentially be recognised as being discovered. Thus, the isotope 280Ds currently remains unknown.[16][17]
As decay product
| Evaporation residue | Observed darmstadtium isotope |
|---|---|
| 277Cn | 273Ds[18] |
| 285Fl, 281Cn | 277Ds[19] |
| 291Lv, 287Fl, 283Cn | 279Ds[20] |
| 293Lv, 289Fl, 285Cn | 281Ds[21] |
Darmstadtium has been observed as decay products of copernicium. Copernicium currently has six known isotopes, four of which have been shown to undergo alpha decays to become darmstadtium nuclei, with mass numbers between 273 and 281. Darmstadtium isotopes with mass numbers 277, 279 and 281 to date have only been produced by copernicium nuclei decay. Parent copernicium nuclei can be themselves decay products of flerovium or livermorium. To date, no other elements have been known to decay to darmstadtium.[17] For example, in 2004, the Dubna team (JINR) identified darmstadtium-281 as a product in the decay of livermorium via an alpha decay sequence:[21]
- 293
116Lv
→ 289
114Fl
+ 4
2He - 289
114Fl
→ 285
112Cn
+ 4
2He - 285
112Cn
→ 281
110Ds
+ 4
2He
Retracted isotopes
- 280Ds
The first synthesis of element 114 resulted in two atoms assigned to 288Fl, decaying to the 280Ds, which underwent spontaneous fission. The assignment was later changed to 289Fl and the darmstadtium isotope to 281Ds. Hence, 280Ds is currently unknown.
- 277Ds
In the claimed synthesis of 293Og in 1999, the isotope 277Ds was identified as decaying by 10.18 MeV alpha emission with a half-life of 3.0 ms. This claim was retracted in 2001. This isotope was finally created in 2010 and its decay data supported the fabrication of previous data.[22]
- 273mDs
In the synthesis of 277Cn in 1996 by GSI (see copernicium), one decay chain proceeded via 273Ds, which decayed by emission of a 9.73 MeV alpha particle with a lifetime of 170 ms. This would have been assigned to an isomeric level. This data could not be confirmed and thus this isotope is currently unknown or unconfirmed.
- 272Ds
In the first attempt to synthesize darmstadtium, a 10 ms SF activity was assigned to 272Ds in the reaction 232Th(44Ca,4n). Given current understanding regarding stability, this isotope has been retracted from the table of isotopes.
Nuclear isomerism
- 281Ds
The production of 281Ds by the decay of 289Fl or 293Lv has produced two very different decay modes. The most common and readily confirmed mode is spontaneous fission with a half-life of 11 s. A much rarer and as yet unconfirmed mode is alpha decay by emission of an alpha particle with energy 8.77 MeV with an observed half-life of around 3.7 min. This decay is associated with a unique decay pathway from the parent nuclides and must be assigned to an isomeric level. The half-life suggests that it must be assigned to an isomeric state but further research is required to confirm these reports.[21]
- 271Ds
Decay data from the direct synthesis of 271Ds clearly indicates the presence of two nuclear isomers. The first emits alpha particles with energies 10.74 and 10.69 MeV and has a half-life of 1.63 ms. The other only emits alpha particles with an energy of 10.71 MeV and has a half-life of 69 ms. The first has been assigned to the ground state and the latter to an isomeric level. It has been suggested that the closeness of the alpha decay energies indicates that the isomeric level may decay primarily by delayed isomeric transition to the ground state, resulting in an identical measured alpha energy and a combined half-life for the two processes.[23]
- 270Ds
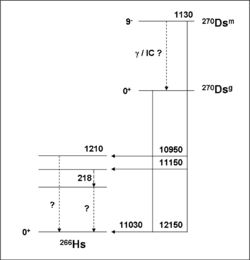
The direct production of 270Ds has clearly identified two nuclear isomers. The ground state decays by alpha emission into the ground state of 266Hs by emitting an alpha particle with energy 11.03 MeV and has a half-life of 0.10 ms. The metastable state decays by alpha emission, emitting alpha particles with energies of 12.15, 11.15, and 10.95 MeV, and has a half-life of 6 ms. When the metastable state emits an alpha particle of energy 12.15 MeV, it decays into the ground state of 266Hs, indicating that it has 1.12 MeV of excess energy.[12]
Chemical yields of isotopes
Cold fusion
The table below provides cross-sections and excitation energies for cold fusion reactions producing darmstadtium isotopes directly. Data in bold represent maxima derived from excitation function measurements. + represents an observed exit channel.
| Projectile | Target | CN | 1n | 2n | 3n |
|---|---|---|---|---|---|
| 62Ni | 208Pb | 270Ds | 3.5 pb | ||
| 64Ni | 208Pb | 272Ds | 15 pb, 9.9 MeV |
Fission of compound nuclei with Z=110
Experiments have been performed in 2004 at the Flerov Laboratory of Nuclear Reactions in Dubna studying the fission characteristics of the compound nucleus280Ds. The nuclear reaction used is 232Th+48Ca. The result revealed how nuclei such as this fission predominantly by expelling closed shell nuclei such as 132Sn (Z=50, N=82).[24]
Theoretical calculations
Decay characteristics
Theoretical calculation in a quantum tunneling model reproduces the experimental alpha decay half live data.[25][26] It also predicts that the isotope 294110 would have alpha decay half-life of the order of 311 years.[27][28]
Evaporation residue cross sections
The below table contains various targets-projectile combinations for which calculations have provided estimates for cross section yields from various neutron evaporation channels. The channel with the highest expected yield is given.
DNS = Di-nuclear system; σ = cross section
| Target | Projectile | CN | Channel (product) | σmax | Model | Ref |
|---|---|---|---|---|---|---|
| 208Pb | 64Ni | 272Ds | 1n (271Ds) | 10 pb | DNS | [29] |
| 232Th | 48Ca | 280Ds | 4n (276Ds) | 0.2 pb | DNS | [30] |
| 230Th | 48Ca | 278Ds | 4n (274Ds) | 1 pb | DNS | [30] |
| 238U | 40Ar | 278Ds | 4n (274Ds) | 2 pb | DNS | [30] |
References
- ↑ V. K. Utyonkov (March 31 – April 2, 2015). "Synthesis of superheavy nuclei at limits of stability: 239,240Pu + 48Ca and 249-251Cf + 48Ca reactions" (PDF). Super Heavy Nuclei International Symposium, Texas A & M University, College Station TX, USA.
- ↑ http://xxx.lanl.gov/pdf/1502.03030.pdf
- 1 2 Armbruster, Peter & Munzenberg, Gottfried (1989). "Creating superheavy elements". Scientific American. 34: 36–42.
- ↑ Barber, Robert C.; Gäggeler, Heinz W.; Karol, Paul J.; Nakahara, Hiromichi; Vardaci, Emanuele; Vogt, Erich (2009). "Discovery of the element with atomic number 112 (IUPAC Technical Report)". Pure and Applied Chemistry. 81 (7): 1331. doi:10.1351/PAC-REP-08-03-05.
- ↑ Fleischmann, Martin; Pons, Stanley (1989). "Electrochemically induced nuclear fusion of deuterium". Journal of Electroanalytical Chemistry and Interfacial Electrochemistry. Elsevier. 261 (2): 301–308. doi:10.1016/0022-0728(89)80006-3. Retrieved 15 October 2012.
- 1 2 Hofmann, S.; Ninov, V.; Heßberger, F. P.; Armbruster, P.; Folger, H.; Münzenberg, G.; Schött, H. J.; Popeko, A. G.; Yeremin, A. V.; Andreyev, A. N.; Saro, S.; Janik, R.; Leino, M. (1995). "Production and decay of269110". Zeitschrift für Physik A. 350 (4): 277–280. Bibcode:1995ZPhyA.350..277H. doi:10.1007/BF01291181.
- ↑ Ginter, T. N.; Gregorich, K.; Loveland, W.; Lee, D.; Kirbach, U.; Sudowe, R.; Folden, C.; Patin, J.; Seward, N.; Wilk, P.; Zielinski, P.; Aleklett, K.; Eichler, R.; Nitsche, H.; Hoffman, D. (2003). "Confirmation of production of element 110 by the 208Pb(64Ni,n) reaction". Physical Review C. 67 (6): 064609. Bibcode:2003PhRvC..67f4609G. doi:10.1103/PhysRevC.67.064609.
- ↑ Ginter, T. N.; Gregorich, K.; Loveland, W.; Lee, D.; Kirbach, U.; Sudowe, R.; Folden, C.; Patin, J.; Seward, N. (8 December 2002). "Confirmation of production of element 110 by the 208Pb(64Ni,n) reaction". LBNL repositories. Retrieved 2008-03-02. (preprint)
- ↑ Folden, C. M.; Gregorich, KE; Düllmann, ChE; Mahmud, H; Pang, GK; Schwantes, JM; Sudowe, R; Zielinski, PM; Nitsche, H; Hoffman, D. (2004). "Development of an Odd-Z-Projectile Reaction for Heavy Element Synthesis: 208Pb(64Ni,n)271Ds and 208Pb(65Cu,n)272111". Physical Review Letters. 93 (21): 212702. Bibcode:2004PhRvL..93u2702F. doi:10.1103/PhysRevLett.93.212702. PMID 15601003.
- ↑ Morita, K.; Morimoto, K.; Kaji, D.; Haba, H.; Ideguchi, E.; Kanungo, R.; Katori, K.; Koura, H.; Kudo, H.; Ohnishi, T.; Ozawa, A.; Suda, T.; Sueki, K.; Tanihata, I.; Xu, H.; Yeremin, A. V.; Yoneda, A.; Yoshida, A.; Zhao, Y.-L.; Zheng, T. (2004). "Production and decay of the isotope 271Ds (Z = 110)". The European Physical Journal A. 21 (2): 257–263. Bibcode:2004EPJA...21..257M. doi:10.1140/epja/i2003-10205-1.
- ↑ George Johnson (15 October 2002). "At Lawrence Berkeley, Physicists Say a Colleague Took Them for a Ride". The New York Times.
- 1 2 3 Hofmann; Heßberger, F. P.; Ackermann, D.; Antalic, S.; Cagarda, P.; Ćwiok, S.; Kindler, B.; Kojouharova, J.; Lommel, B.; Mann, R.; Münzenberg, G.; Popeko, A. G.; Saro, S.; Schött, H. J.; Yeremin, A. V. (2001). "The new isotope 270110 and its decay products 266Hs and 262Sg" (PDF). Eur. Phys. J. A. 10: 5–10. Bibcode:2001EPJA...10....5H. doi:10.1007/s100500170137.
- ↑ Ghiorso, A.; Lee, D.; Somerville, L.; Loveland, W.; Nitschke, J.; Ghiorso, W.; Seaborg, G.; Wilmarth, P.; Leres, R.; Wydler, A.; Nurmia, M.; Gregorich, K.; Czerwinski, K.; Gaylord, R.; Hamilton, T.; Hannink, N. J.; Hoffman, D. C.; Jarzynski, C.; Kacher, C.; Kadkhodayan, B.; Kreek, S.; Lane, M.; Lyon, A.; McMahan, M. A.; Neu, M.; Sikkeland, T.; Swiatecki, W. J.; Türler, A.; Walton, J. T.; Yashita, S. (1995). "Evidence for the possible synthesis of element 110 produced by the 59Co+209Bi reaction". Physical Review C. 51 (5): R2293. Bibcode:1995PhRvC..51.2293G. doi:10.1103/PhysRevC.51.R2293.
- ↑ Lazarev, Yu. A.; Lobanov, Yu.; Oganessian, Yu.; Utyonkov, V.; Abdullin, F.; Polyakov, A.; Rigol, J.; Shirokovsky, I.; Tsyganov, Yu.; Iliev, S.; Subbotin, V. G.; Sukhov, A. M.; Buklanov, G. V.; Gikal, B. N.; Kutner, V. B.; Mezentsev, A. N.; Subotic, K.; Wild, J. F.; Lougheed, R. W.; Moody, K. J. (1996). "α decay of 273110: Shell closure at N=162". Physical Review C. 54 (2): 620–625. Bibcode:1996PhRvC..54..620L. doi:10.1103/PhysRevC.54.620.
- ↑ Flerov lab annual report 2004
- ↑ Emsley, John (2011). Nature's Building Blocks: An A–Z Guide to the Elements (New ed.). New York, NY: Oxford University Press. p. 590. ISBN 978-0-19-960563-7.
- 1 2 Sonzogni, Alejandro. "Interactive Chart of Nuclides". National Nuclear Data Center: Brookhaven National Laboratory. Retrieved 2008-06-06.
- ↑ Hofmann, S.; Ninov, V.; Heßberger, F. P.; Armbruster, P.; Folger, H.; Münzenberg, G.; Schött, H. J.; Popeko, A. G.; Yeremin, A. V.; Saro, S.; Janik, R.; Leino, M. (1996). "The new element 112". Zeitschrift für Physik A. 354 (1): 229–230. doi:10.1007/BF02769517.
- ↑ Public Affairs Department (26 October 2010). "Six New Isotopes of the Superheavy Elements Discovered: Moving Closer to Understanding the Island of Stability". Berkeley Lab. Retrieved 2011-04-25.
- ↑ Yeremin, A. V.; et al. (1999). "Synthesis of nuclei of the superheavy element 114 in reactions induced by 48Ca". Nature. 400 (6741): 242–245. Bibcode:1999Natur.400..242O. doi:10.1038/22281.
- 1 2 3 Oganessian, Y. T.; Utyonkov, V.; Lobanov, Y.; Abdullin, F.; Polyakov, A.; Shirokovsky, I.; Tsyganov, Y.; Gulbekian, G.; Bogomolov, S.; Gikal, B.; et al. (2004). "Measurements of cross sections for the fusion-evaporation reactions 244Pu(48Ca,xn)292−x114 and 245Cm(48Ca,xn)293−x116". Physical Review C. 69 (5): 054607. Bibcode:2004PhRvC..69e4607O. doi:10.1103/PhysRevC.69.054607.
- ↑ see oganesson
- ↑ Hofmann, S (1998). "New elements - approaching". Reports on Progress in Physics. 61 (6): 639–689. Bibcode:1998RPPh...61..639H. doi:10.1088/0034-4885/61/6/002.
- ↑ see Flerov lab annual report 2004
- ↑ P. Roy Chowdhury; C. Samanta; D. N. Basu (2006). "α decay half-lives of new superheavy elements". Phys. Rev. C. 73: 014612. arXiv:nucl-th/0507054
 . Bibcode:2006PhRvC..73a4612C. doi:10.1103/PhysRevC.73.014612.
. Bibcode:2006PhRvC..73a4612C. doi:10.1103/PhysRevC.73.014612. - ↑ C. Samanta; P. Roy Chowdhury; D. N. Basu (2007). "Predictions of alpha decay half lives of heavy and superheavy elements". Nucl. Phys. A. 789: 142–154. arXiv:nucl-th/0703086
 . Bibcode:2007NuPhA.789..142S. doi:10.1016/j.nuclphysa.2007.04.001.
. Bibcode:2007NuPhA.789..142S. doi:10.1016/j.nuclphysa.2007.04.001. - ↑ P. Roy Chowdhury; C. Samanta; D. N. Basu (2008). "Search for long lived heaviest nuclei beyond the valley of stability". Phys. Rev. C. 77 (4): 044603. arXiv:0802.3837
 . Bibcode:2008PhRvC..77d4603C. doi:10.1103/PhysRevC.77.044603.
. Bibcode:2008PhRvC..77d4603C. doi:10.1103/PhysRevC.77.044603. - ↑ P. Roy Chowdhury,; C. Samanta; D. N. Basu (2008). "Nuclear half-lives for α -radioactivity of elements with 100 ≤ Z ≤ 130". Atomic Data and Nuclear Data Tables. 94 (6): 781–806. arXiv:0802.4161
 . Bibcode:2008ADNDT..94..781C. doi:10.1016/j.adt.2008.01.003.
. Bibcode:2008ADNDT..94..781C. doi:10.1016/j.adt.2008.01.003. - ↑ Feng, Zhao-Qing; Jin, Gen-Ming; Li, Jun-Qing; Scheid, Werner (2007). "Formation of superheavy nuclei in cold fusion reactions". Physical Review C. 76 (4): 044606. arXiv:0707.2588
 . Bibcode:2007PhRvC..76d4606F. doi:10.1103/PhysRevC.76.044606.
. Bibcode:2007PhRvC..76d4606F. doi:10.1103/PhysRevC.76.044606. - 1 2 3 Feng, Z; Jin, G; Li, J; Scheid, W (2009). "Production of heavy and superheavy nuclei in massive fusion reactions". Nuclear Physics A. 816: 33–51. arXiv:0803.1117
 . Bibcode:2009NuPhA.816...33F. doi:10.1016/j.nuclphysa.2008.11.003.
. Bibcode:2009NuPhA.816...33F. doi:10.1016/j.nuclphysa.2008.11.003.
- Isotope masses from:
- M. Wang; G. Audi; A. H. Wapstra; F. G. Kondev; M. MacCormick; X. Xu; et al. (2012). "The AME2012 atomic mass evaluation (II). Tables, graphs and references." (PDF). Chinese Physics C. 36 (12): 1603–2014. Bibcode:2012ChPhC..36....3M. doi:10.1088/1674-1137/36/12/003.
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillon (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- Isotopic compositions and standard atomic masses from:
- J. R. de Laeter; J. K. Böhlke; P. De Bièvre; H. Hidaka; H. S. Peiser; K. J. R. Rosman; P. D. P. Taylor (2003). "Atomic weights of the elements. Review 2000 (IUPAC Technical Report)". Pure and Applied Chemistry. 75 (6): 683–800. doi:10.1351/pac200375060683.
- M. E. Wieser (2006). "Atomic weights of the elements 2005 (IUPAC Technical Report)". Pure and Applied Chemistry. 78 (11): 2051–2066. doi:10.1351/pac200678112051. Lay summary.
- Half-life, spin, and isomer data selected from the following sources. See editing notes on this article's talk page.
- G. Audi; A. H. Wapstra; C. Thibault; J. Blachot; O. Bersillonn (2003). "The NUBASE evaluation of nuclear and decay properties" (PDF). Nuclear Physics A. 729: 3–128. Bibcode:2003NuPhA.729....3A. doi:10.1016/j.nuclphysa.2003.11.001.
- National Nuclear Data Center. "NuDat 2.1 database". Brookhaven National Laboratory. Retrieved September 2005. Check date values in:
|access-date=(help) - N. E. Holden (2004). "Table of the Isotopes". In D. R. Lide. CRC Handbook of Chemistry and Physics (85th ed.). CRC Press. Section 11. ISBN 978-0-8493-0485-9.
| Isotopes of meitnerium | Isotopes of darmstadtium | Isotopes of roentgenium |
| Table of nuclides | ||
| Isotopes of the chemical elements | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 H |
2 He | ||||||||||||||||
| 3 Li |
4 Be |
5 B |
6 C |
7 N |
8 O |
9 F |
10 Ne | ||||||||||
| 11 Na |
12 Mg |
13 Al |
14 Si |
15 P |
16 S |
17 Cl |
18 Ar | ||||||||||
| 19 K |
20 Ca |
21 Sc |
22 Ti |
23 V |
24 Cr |
25 Mn |
26 Fe |
27 Co |
28 Ni |
29 Cu |
30 Zn |
31 Ga |
32 Ge |
33 As |
34 Se |
35 Br |
36 Kr |
| 37 Rb |
38 Sr |
39 Y |
40 Zr |
41 Nb |
42 Mo |
43 Tc |
44 Ru |
45 Rh |
46 Pd |
47 Ag |
48 Cd |
49 In |
50 Sn |
51 Sb |
52 Te |
53 I |
54 Xe |
| 55 Cs |
56 Ba |
|
72 Hf |
73 Ta |
74 W |
75 Re |
76 Os |
77 Ir |
78 Pt |
79 Au |
80 Hg |
81 Tl |
82 Pb |
83 Bi |
84 Po |
85 At |
86 Rn |
| 87 Fr |
88 Ra |
|
104 Rf |
105 Db |
106 Sg |
107 Bh |
108 Hs |
109 Mt |
110 Ds |
111 Rg |
112 Cn |
113 Nh |
114 Fl |
115 Mc |
116 Lv |
117 Ts |
118 Og |
| |
57 La |
58 Ce |
59 Pr |
60 Nd |
61 Pm |
62 Sm |
63 Eu |
64 Gd |
65 Tb |
66 Dy |
67 Ho |
68 Er |
69 Tm |
70 Yb |
71 Lu | ||
| |
89 Ac |
90 Th |
91 Pa |
92 U |
93 Np |
94 Pu |
95 Am |
96 Cm |
97 Bk |
98 Cf |
99 Es |
100 Fm |
101 Md |
102 No |
103 Lr | ||
| |||||||||||||||||