Sodium perchlorate
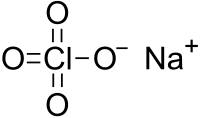 | |
 | |
| Names | |
|---|---|
| Other names
Sodium chlorate(VII) Sodium hyperchlorate Perchloric acid, sodium salt | |
| Identifiers | |
| 7601-89-0 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:132103 |
| ChEMBL | ChEMBL1644700 |
| ChemSpider | 22668 |
| ECHA InfoCard | 100.028.647 |
| EC Number | 231-511-9 |
| PubChem | 522606 |
| RTECS number | SC9800000 |
| UN number | 1502 |
| |
| |
| Properties | |
| NaClO4 NaClO4.H2O (monohydrate) | |
| Molar mass | 122.44 g/mol |
| Appearance | White crystalline solid |
| Density | 2.4994 g/cm3 2.02 g/cm3 (monohydrate) |
| Melting point | 468 °C (874 °F; 741 K) (decomposes, anhydrous) 130 °C (monohydrate) |
| Boiling point | 482 °C (900 °F; 755 K) (decomposes, monohydrate) |
| 209.6 g/100 mL (25 °C, anhydrous) 209 g/100 mL (15 °C, monohydrate) | |
| Refractive index (nD) |
1.4617 |
| Structure | |
| orthorhombic | |
| Hazards | |
| Safety data sheet | ICSC 0715 |
| EU classification (DSD) |
Oxidant (O) Harmful (Xn) |
| R-phrases | R9, R22 |
| S-phrases | (S2), S13, S22, S27 |
| NFPA 704 | |
| Flash point | 400 °C (752 °F; 673 K) |
| Related compounds | |
| Other anions |
Sodium chloride Sodium hypochlorite Sodium chlorite Sodium chlorate |
| Other cations |
Lithium perchlorate Potassium perchlorate Ammonium perchlorate Caesium perchlorate |
| Related compounds |
Perchloric acid |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Sodium perchlorate is the inorganic compound with the chemical formula NaClO4. It is the most soluble of the common perchlorate salts. It is a white crystalline, hygroscopic solid that is highly soluble in water and in alcohol. It usually comes as the monohydrate, which has a rhombic crystal system.[1]
Its heat of formation is −382.75 kJ mol−1.[2]
Uses
Sodium perchlorate is the precursor to many other perchlorate salts, often taking advantage of their low solubility relative to NaClO4 (209 g/100 mL at 25 °C). Perchloric acid is made by treating NaClO4 with HCl.
NaClO4 finds only minimal use in pyrotechnics because it is hygroscopic; ammonium and potassium perchlorates are preferred. These salts are prepared by double decomposition from a solution of sodium perchlorate and potassium or ammonium chlorides.
Laboratory applications
NaClO4 has a variety of uses in the laboratory, often as an unreactive electrolyte. For example, it is used in standard DNA extraction and hybridization reactions in molecular biology.
In medicine
Sodium perchlorate can be used to block iodine uptake before administration of iodinated contrast agents in patients with subclinical hyperthyroidism (suppressed TSH).[3]
Production
Sodium perchlorate is produced by anodic oxidation of sodium chlorate at an inert electrode, such as platinum.[4]
- ClO3−(aq) + H2O(l) → ClO4−(aq) + H2(g)
See also
References
- ↑ Eagleson, Mary (1994). Concise Encyclopedia Chemistry. revised, illustrated. Walter de Gruyter. p. 1000. ISBN 9783110114515. Retrieved March 7, 2013.
- ↑ WebBook page for NaClO4
- ↑ Becker C. [Prophylaxis and treatment of side effects due to iodinated contrast media relevant to radiological practice]. Radiologe. 2007 Sep;47(9):768-73.
- ↑ Helmut Vogt, Jan Balej, John E. Bennett, Peter Wintzer, Saeed Akbar Sheikh, Patrizio Gallone "Chlorine Oxides and Chlorine Oxygen Acids" in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH. doi:10.1002/14356007.a06_483
External links
| Salts and the ester of the perchlorate ion | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HClO4 | He | ||||||||||||||||||
| LiClO4 | Be(ClO4)2 | B(ClO4)4− | ROClO3 | N(ClO4)3 NH4ClO4 |
O | FClO4 | Ne | ||||||||||||
| NaClO4 | Mg(ClO4)2 | Al(ClO4)3 | Si | P | S | ClO4− ClOClO3 Cl2O7 |
Ar | ||||||||||||
| KClO4 | Ca(ClO4)2 | Sc(ClO4)3 | Ti(ClO4)4 | VO(ClO4)3 | Cr(ClO4)3 | Mn(ClO4)2 | Fe(ClO4)3 | Co(ClO4)2, Co(ClO4)3 |
Ni(ClO4)2 | Cu(ClO4)2 | Zn(ClO4)2 | Ga(ClO4)3 | Ge | As | Se | Br | Kr | ||
| RbClO4 | Sr(ClO4)2 | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd(ClO4)2 | AgClO4 | Cd(ClO4)2 | In | Sn | Sb | Te | I | Xe | ||
| CsClO4 | Ba(ClO4)2 | Hf | Ta | W | Re | Os | Ir | Pt | Au | Hg2(ClO4)2, Hg(ClO4)2 |
Tl(ClO4)3 | Pb(ClO4)2 | Bi(ClO4)3 | Po | At | Rn | |||
| Fr | Ra | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||
| ↓ | |||||||||||||||||||
| La | Ce(ClO4)x | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | |||||
| Ac | Th | Pa | UO2(ClO4)2 | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||||
