Gamma ray

Gamma ray (also called gamma radiation), denoted by the lower-case Greek letter gamma (γ or ), is penetrating electromagnetic radiation of a kind arising from the radioactive decay of atomic nuclei. It consists of photons in the highest observed range of photon energy. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900 while studying radiation emitted by radium. In 1903, Ernest Rutherford named this radiation gamma rays. Rutherford had previously discovered two other types of radioactive decay, which he named alpha and beta rays.
Gamma rays are able to ionize other atoms (ionizing radiation), and are thus biologically hazardous. The decay of an atomic nucleus from a high energy state to a lower energy state, a process called gamma decay, produces gamma radiation.
Natural sources of gamma rays on Earth are observed in the gamma decay of radionuclides and secondary radiation from atmospheric interactions with cosmic ray particles. There are rare terrestrial natural sources, such as lightning strikes and terrestrial gamma-ray flashes, that produce gamma rays not of a nuclear origin. Additionally, gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced, that in turn cause secondary gamma rays via bremsstrahlung, inverse Compton scattering, and synchrotron radiation. However, a large fraction of such astronomical gamma rays are screened by Earth's atmosphere and can only be detected by spacecraft. Gamma rays are produced by nuclear fusion in stars including the Sun (such as the CNO cycle), but are absorbed or inelastically scattered by the stellar material, reducing their energy, before escaping and are not observable from Earth as gamma rays.
Gamma rays typically have frequencies above 10 exahertz (or >1019 Hz), and therefore have energies above 100 keV and wavelengths less than 10 picometers (10−11 m), which is less than the diameter of an atom. However, this is not a strict definition, but rather only a rule-of-thumb description for natural processes. Electromagnetic radiation from radioactive decay of atomic nuclei is referred to as "gamma rays" no matter its energy, so that there is no lower limit to gamma energy derived from radioactive decay. This radiation commonly has energy of a few hundred keV, and almost always less than 10 MeV. In astronomy, gamma rays are defined by their energy, and no production process needs to be specified. The energies of gamma rays from astronomical sources range to over 10 TeV, an energy far too large to result from radioactive decay.[1] A notable example is the extremely powerful bursts of high-energy radiation referred to as long duration gamma-ray bursts, of energies higher than can be produced by radioactive decay. These bursts of gamma rays, thought to be due to the collapse of stars called hypernovae, are the most powerful events so far discovered in the cosmos.
History of discovery
The first gamma ray source to be discovered historically was the radioactive decay process called gamma decay. In this type of decay, an excited nucleus emits a gamma ray almost immediately upon formation (it is now understood that a nuclear isomeric transition, however, can produce inhibited gamma decay with a measurable and much longer half-life). Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900, while studying radiation emitted from radium. Villard knew that his described radiation was more powerful than previously described types of rays from radium, which included beta rays, first noted as "radioactivity" by Henri Becquerel in 1896, and alpha rays, discovered as a less penetrating form of radiation by Rutherford, in 1899. However, Villard did not consider naming them as a different fundamental type.[2][3] Villard's radiation was recognized as being of a type fundamentally different from previously named rays, by Ernest Rutherford, who in 1903 named Villard's rays "gamma rays" by analogy with the beta and alpha rays that Rutherford had differentiated in 1899.[4] The "rays" emitted by radioactive elements were named in order of their power to penetrate various materials, using the first three letters of the Greek alphabet: alpha rays as the least penetrating, followed by beta rays, followed by gamma rays as the most penetrating. Rutherford also noted that gamma rays were not deflected (or at least, not easily deflected) by a magnetic field, another property making them unlike alpha and beta rays.
Gamma rays were first thought to be particles with mass, like alpha and beta rays. Rutherford initially believed that they might be extremely fast beta particles, but their failure to be deflected by a magnetic field indicated that they had no charge.[5] In 1914, gamma rays were observed to be reflected from crystal surfaces, proving that they were electromagnetic radiation.[5] Rutherford and his coworker Edward Andrade measured the wavelengths of gamma rays from radium, and found that they were similar to X-rays, but with shorter wavelengths and (thus) higher frequency. This was eventually recognized as giving them also more energy per photon, as soon as the latter term became generally accepted. A gamma decay was then understood to usually emit a single gamma photon.
Sources of gamma rays
Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes such as potassium-40, and also as a secondary radiation from various atmospheric interactions with cosmic ray particles. Some rare terrestrial natural sources that produce gamma rays that are not of a nuclear origin, are lightning strikes and terrestrial gamma-ray flashes, which produce high energy emissions from natural high-energy voltages. Gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced. Such electrons produce secondary gamma rays by the mechanisms of bremsstrahlung, inverse Compton scattering and synchrotron radiation. A large fraction of such astronomical gamma rays are screened by Earth's atmosphere and must be detected by spacecraft. Notable artificial sources of gamma rays include fission such as occurs in nuclear reactors, and high energy physics experiments, such as neutral pion decay and nuclear fusion.
General characteristics
| Nuclear physics |
|---|
 |
| Nucleus · Nucleons (p, n) · Nuclear force · Nuclear structure · Nuclear reaction |
|
Nuclear models and stability |
|
Nucleosynthesis topics Nuclear fusion Processes: Stellar · Big Bang · Supernova Nuclides: Primordial · Cosmogenic · Artificial |
|
Scientists Alvarez · Becquerel · Bethe · A.Bohr · N.Bohr · Chadwick · Cockcroft · Ir.Curie · Fr.Curie · Pi.Curie · Skłodowska-Curie · Davisson · Fermi · Hahn · Jensen · Lawrence · Mayer · Meitner · Oliphant · Oppenheimer · Proca · Purcell · Rabi · Rutherford · Soddy · Strassmann · Szilárd · Teller · Thomson · Walton · Wigner |
The distinction between X-rays and gamma rays has changed in recent decades. Originally, the electromagnetic radiation emitted by X-ray tubes almost invariably had a longer wavelength than the radiation (gamma rays) emitted by radioactive nuclei.[6] Older literature distinguished between X- and gamma radiation on the basis of wavelength, with radiation shorter than some arbitrary wavelength, such as 10−11 m, defined as gamma rays.[7] However, with artificial sources now able to duplicate any electromagnetic radiation that originates in the nucleus, as well as far higher energies, the wavelengths characteristic of radioactive gamma ray sources vs. other types, now completely overlap. Thus, gamma rays are now usually distinguished by their origin: X-rays are emitted by definition by electrons outside the nucleus, while gamma rays are emitted by the nucleus.[6][8][9][10] Exceptions to this convention occur in astronomy, where gamma decay is seen in the afterglow of certain supernovas, but radiation from high energy processes known to involve other radiation sources than radioactive decay is still classed as gamma radiation.
Naming conventions and overlap in terminology

In the past, the distinction between X-rays and gamma rays was based on energy, with gamma rays being considered a higher-energy version of electromagnetic radiation. However, modern high-energy X-rays produced by linear accelerators for megavoltage treatment in cancer often have higher energy (4 to 25 MeV) than do most classical gamma rays produced by nuclear gamma decay. One of the most common gamma ray emitting isotopes used in diagnostic nuclear medicine, technetium-99m, produces gamma radiation of the same energy (140 keV) as that produced by diagnostic X-ray machines, but of significantly lower energy than therapeutic photons from linear particle accelerators. In the medical community today, the convention that radiation produced by nuclear decay is the only type referred to as "gamma" radiation is still respected.
Because of this broad overlap in energy ranges, in physics the two types of electromagnetic radiation are now often defined by their origin: X-rays are emitted by electrons (either in orbitals outside of the nucleus, or while being accelerated to produce bremsstrahlung-type radiation),[12] while gamma rays are emitted by the nucleus or by means of other particle decays or annihilation events. There is no lower limit to the energy of photons produced by nuclear reactions, and thus ultraviolet or lower energy photons produced by these processes would also be defined as "gamma rays".[13] The only naming-convention that is still universally respected is the rule that electromagnetic radiation that is known to be of atomic nuclear origin is always referred to as "gamma rays," and never as X-rays. However, in physics and astronomy, the converse convention (that all gamma rays are considered to be of nuclear origin) is frequently violated.
In astronomy, higher energy gamma and X-rays are defined by energy, since the processes that produce them may be uncertain and photon energy, not origin, determines the required astronomical detectors needed.[14] High energy photons occur in nature that are known to be produced by processes other than nuclear decay but are still referred to as gamma radiation. An example is "gamma rays" from lightning discharges at 10 to 20 MeV, and known to be produced by the bremsstrahlung mechanism.
Another example is gamma-ray bursts, now known to be produced from processes too powerful to involve simple collections of atoms undergoing radioactive decay. This has led to the realization that many gamma rays produced in astronomical processes result not from radioactive decay or particle annihilation, but rather in much the same manner as the production of X-rays. Although gamma rays in astronomy are discussed below as non-radioactive events, in fact a few gamma rays are known in astronomy to originate explicitly from gamma decay of nuclei (as demonstrated by their spectra and emission half life). A classic example is that of supernova SN 1987A, which emits an "afterglow" of gamma-ray photons from the decay of newly made radioactive nickel-56 and cobalt-56. Most gamma rays in astronomy, however, arise by other mechanisms. Astronomical literature tends to write "gamma-ray" with a hyphen, by analogy to X-rays, rather than in a way analogous to alpha rays and beta rays. This notation tends to subtly stress the non-nuclear source of most astronomical "gamma-rays".
Units of measurement and exposure
The measure of the ionizing effect of gamma rays is called the exposure:
- The coulomb per kilogram (C/kg) is the SI unit of ionizing radiation exposure, and is the amount of radiation required to create 1 coulomb of charge of each polarity in 1 kilogram of matter.
- The röntgen (R) is an obsolete traditional unit of exposure, which represented the amount of radiation required to create 1 esu of charge of each polarity in 1 cubic centimeter of dry air. 1 röntgen = 2.58×10−4 C/kg
However, the effect of gamma and other ionizing radiation on living tissue is more closely related to the amount of energy deposited rather than the charge. This is called the absorbed dose:
- The gray (Gy), which has units of joules per kilogram (J/kg), is the SI unit of absorbed dose, and is the amount of radiation required to deposit 1 joule of energy in 1 kilogram of any kind of matter.
- The rad is the deprecated CGS unit, numerically equal to 0.01 J deposited per kilogram of matter; thus, 100 rad = 1 Gy.
The equivalent dose is the measure of the biological effect of radiation on human tissue. For gamma rays, it is equal to the absorbed dose.
- The sievert (Sv) is the SI unit of equivalent dose, which for gamma rays is numerically equal to the absorbed dose in grays (Gy).
- The rem is the deprecated CGS unit of equivalent dose. For gamma rays, an equivalent dose in rems is numerically equal to the absorbed dose in rads, or 0.01 J of energy absorbed per kilogram of matter; thus, 1 Sv = 100 rem.
Properties
Shielding
Shielding from gamma rays requires large amounts of mass, in contrast to alpha particles, which can be blocked by paper or skin, and beta particles, which can be shielded by foil. Gamma rays are better absorbed by materials with high atomic numbers and high density, although neither effect is important compared to the total mass per area in the path of the gamma ray. For this reason, a lead shield is only modestly better (20–30% better) as a gamma shield than an equal mass of another shielding material, such as aluminium, concrete, water, or soil; lead's major advantage is not in lower weight, but rather its compactness due to its higher density. Protective clothing, goggles and respirators can protect from internal contact with or ingestion of alpha or beta emitting particles, but provide no protection from gamma radiation from external sources.
The higher the energy of the gamma rays, the thicker the shielding made from the same shielding material is required. Materials for shielding gamma rays are typically measured by the thickness required to reduce the intensity of the gamma rays by one half (the half value layer or HVL). For example, gamma rays that require 1 cm (0.4″) of lead to reduce their intensity by 50% will also have their intensity reduced in half by 4.1 cm of granite rock, 6 cm (2½″) of concrete, or 9 cm (3½″) of packed soil. However, the mass of this much concrete or soil is only 20–30% greater than that of lead with the same absorption capability. Depleted uranium is used for shielding in portable gamma ray sources, but here the savings in weight over lead are larger, as portable sources' shape resembles a sphere to some extent, and the volume of a sphere is dependent on the cube of the radius; so a source with its radius cut in half will have its volume reduced by a factor of eight, which will more than compensate uranium's greater density (as well as reducing bulk). In a nuclear power plant, shielding can be provided by steel and concrete in the pressure and particle containment vessel, while water provides a radiation shielding of fuel rods during storage or transport into the reactor core. The loss of water or removal of a "hot" fuel assembly into the air would result in much higher radiation levels than when kept under water.
Matter interaction
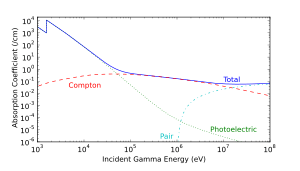
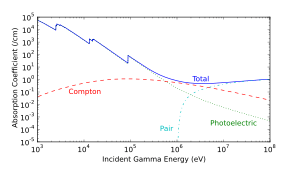
When a gamma ray passes through matter, the probability for absorption is proportional to the thickness of the layer, the density of the material, and the absorption cross section of the material. The total absorption shows an exponential decrease of intensity with distance from the incident surface:
where x is the thickness of the material from the incident surface, μ = nσ is the absorption coefficient, measured in cm−1, n the number of atoms per cm3 of the material (atomic density) and σ the absorption cross section in cm2.
As it passes through matter, gamma radiation ionizes via three processes: the photoelectric effect, Compton scattering, and pair production.
- Photoelectric effect: This describes the case in which a gamma photon interacts with and transfers its energy to an atomic electron, causing the ejection of that electron from the atom. The kinetic energy of the resulting photoelectron is equal to the energy of the incident gamma photon minus the energy that originally bound the electron to the atom (binding energy). The photoelectric effect is the dominant energy transfer mechanism for X-ray and gamma ray photons with energies below 50 keV (thousand electron volts), but it is much less important at higher energies.
- Compton scattering: This is an interaction in which an incident gamma photon loses enough energy to an atomic electron to cause its ejection, with the remainder of the original photon's energy emitted as a new, lower energy gamma photon whose emission direction is different from that of the incident gamma photon, hence the term "scattering". The probability of Compton scattering decreases with increasing photon energy. Compton scattering is thought to be the principal absorption mechanism for gamma rays in the intermediate energy range 100 keV to 10 MeV. Compton scattering is relatively independent of the atomic number of the absorbing material, which is why very dense materials like lead are only modestly better shields, on a per weight basis, than are less dense materials.
- Pair production: This becomes possible with gamma energies exceeding 1.02 MeV, and becomes important as an absorption mechanism at energies over 5 MeV (see illustration at right, for lead). By interaction with the electric field of a nucleus, the energy of the incident photon is converted into the mass of an electron-positron pair. Any gamma energy in excess of the equivalent rest mass of the two particles (totaling at least 1.02 MeV) appears as the kinetic energy of the pair and in the recoil of the emitting nucleus. At the end of the positron's range, it combines with a free electron, and the two annihilate, and the entire mass of these two is then converted into two gamma photons of at least 0.51 MeV energy each (or higher according to the kinetic energy of the annihilated particles).
The secondary electrons (and/or positrons) produced in any of these three processes frequently have enough energy to produce much ionization themselves.
Additionally, gamma rays, particularly high energy ones, can interact with atomic nuclei resulting in ejection of particles in photodisintegration, or in some cases, even nuclear fission (photofission).
Light interaction
High-energy (from 80 GeV to ~10 TeV) gamma rays arriving from far-distant quasars are used to estimate the extragalactic background light in the universe: The highest-energy rays interact more readily with the background light photons and thus the density of the background light may be estimated by analyzing the incoming gamma ray spectra.[15][16]
Gamma ray production
Gamma rays can be produced by a wide range of phenomena, both nuclear and non-nuclear.
Radioactive decay (gamma decay)
Gamma rays are produced during gamma decay, which normally occurs after other forms of decay occur, such as alpha or beta decay. An excited nucleus can decay by the emission of an
α
or
β
particle. The daughter nucleus that results is usually left in an excited state. It can then decay to a lower energy state by emitting a gamma ray photon, in a process called gamma decay.
The emission of a gamma ray from an excited nucleus typically requires only 10−12 seconds. Gamma decay may also follow nuclear reactions such as neutron capture, nuclear fission, or nuclear fusion. Gamma decay is also a mode of relaxation of many excited states of atomic nuclei following other types of radioactive decay, such as beta decay, so long as these states possess the necessary component of nuclear spin. When high-energy gamma rays, electrons, or protons bombard materials, the excited atoms emit characteristic "secondary" gamma rays, which are products of the creation of excited nuclear states in the bombarded atoms. Such transitions, a form of nuclear gamma fluorescence, form a topic in nuclear physics called gamma spectroscopy. Formation of fluorescent gamma rays are a rapid subtype of radioactive gamma decay.
In certain cases, the excited nuclear state that follows the emission of a beta particle or other type of excitation, may be more stable than average, and is termed a metastable excited state, if its decay takes (at least) 100 to 1000 times longer than the average 10−12 seconds. Such relatively long-lived excited nuclei are termed nuclear isomers, and their decays are termed isomeric transitions. Such nuclei have half-lifes that are more easily measurable, and rare nuclear isomers are able to stay in their excited state for minutes, hours, days, or occasionally far longer, before emitting a gamma ray. The process of isomeric transition is therefore similar to any gamma emission, but differs in that it involves the intermediate metastable excited state(s) of the nuclei. Metastable states are often characterized by high nuclear spin, requiring a change in spin of several units or more with gamma decay, instead of a single unit transition that occurs in only 10−12 seconds. The rate of gamma decay is also slowed when the energy of excitation of the nucleus is small.[17]
An emitted gamma ray from any type of excited state may transfer its energy directly to any electrons, but most probably to one of the K shell electrons of the atom, causing it to be ejected from that atom, in a process generally termed the photoelectric effect (external gamma rays and ultraviolet rays may also cause this effect). The photoelectric effect should not be confused with the internal conversion process, in which a gamma ray photon is not produced as an intermediate particle (rather, a "virtual gamma ray" may be thought to mediate the process).

Co
Gamma rays, X-rays, visible light, and radio waves are all forms of electromagnetic radiation. The only difference is the frequency and hence the energy of those photons. Gamma rays are generally the most energetic of these, although a broad overlap with X-ray energies occurs. An example of gamma ray production follows:
First 60
Co
decays to excited 60
Ni
by beta decay emission of an electron of 0.31 MeV. Then the excited 60
Ni
decays to the ground state (see nuclear shell model) by emitting gamma rays in succession of 1.17 MeV followed by 1.33 MeV. This path is followed 99.88% of the time:
Another example is the alpha decay of 241
Am
to form 237
Np
; which is followed by gamma emission. In some cases, the gamma emission spectrum of the daughter nucleus is quite simple, (e.g. 60
Co
/60
Ni
) while in other cases, such as with (241
Am
/237
Np
and 192
Ir
/192
Pt
), the gamma emission spectrum is complex, revealing that a series of nuclear energy levels exist.
Gamma spectroscopy
Because a beta decay is accompanied by the emission of a neutrino that also carries a varying amount of energy away, the beta emission spectrum does not have sharp lines, but instead is broad. Hence, it is not possible to describe the different energy levels found in the nucleus using beta decay energies alone.
Gamma spectroscopy is the study of the energetic transitions in atomic nuclei, which are generally associated with the absorption or emission of gamma rays. As in optical spectroscopy (see Franck Condon effect) the absorption of gamma rays by a nucleus is especially likely (i.e., peaks in a "resonance") when the energy of the gamma ray is the same as that of an energy transition in the nucleus. In the case of gamma rays, such a resonance is seen in the technique of Mössbauer spectroscopy. In the Mössbauer effect the narrow resonance absorption for nuclear gamma absorption can be successfully attained by physically immobilizing atomic nuclei in a crystal. The immobilization of nuclei at both ends of a gamma resonance interaction is required so that no gamma energy is lost to the kinetic energy of recoiling nuclei at either the emitting or absorbing end of a gamma transition. Such loss of energy causes gamma ray resonance absorption to fail. However, when emitted gamma rays carry essentially all of the energy of the atomic nuclear de-excitation that produces them, this energy is also sufficient to excite the same energy state in a second immobilized nucleus of the same type.
Gamma rays from sources other than radioactive decay
A few gamma rays in astronomy are known to arise from gamma decay (see discussion of SN1987A) but most do not.
Photons from astrophysical sources that carry energy in the gamma radiation range are often explicitly called gamma-radiation. In addition to nuclear emissions, they are often produced by sub-atomic particle and particle-photon interactions. Those include electron-positron annihilation, neutral pion decay, bremsstrahlung, inverse Compton scattering, and synchrotron radiation.
- Terrestrial thunderstorms: Thunderstorms can produce a brief pulse of gamma radiation called a terrestrial gamma-ray flash. These gamma rays are thought to be produced by high intensity static electric fields accelerating electrons, which then produce gamma rays by bremsstrahlung as they collide with and are slowed by atoms in the atmosphere. Gamma rays up to 100 MeV can be emitted by terrestrial thunderstorms, and were discovered by space-borne observatories. This raises the possibility of health risks to passengers and crew on aircraft flying in or near thunderclouds.[18]
Extraterrestrial, high energy gamma rays include the gamma ray background produced when cosmic rays (either high speed electrons or protons) collide with ordinary matter, producing pair-production gamma rays at 511 keV. Alternatively, bremsstrahlung are produced at energies of tens of MeV or more when cosmic ray electrons interact with nuclei of sufficiently high atomic number (see gamma ray image of the Moon at the beginning of this article, for illustration).
- Pulsars and magnetars: The gamma ray sky (see illustration at right) is dominated by the more common and longer-term production of gamma rays that emanate from pulsars within the Milky Way. Sources from the rest of the sky are mostly quasars. Pulsars are thought to be neutron stars with magnetic fields that produce focused beams of radiation, and are far less energetic, more common, and much nearer sources (typically seen only in our own galaxy) than are quasars or the rarer gamma-ray burst sources of gamma rays. Pulsars have relatively long-lived magnetic fields that produce focused beams of relativistic speed charged particles, which emit gamma rays (bremsstrahlung) when those strike gas or dust in their nearby medium, and are decelerated. This is a similar mechanism to the production of high energy photons in megavoltage radiation therapy machines (see bremsstrahlung). The "inverse Compton effect", in which charged particles (usually electrons) impart energy to low-energy photons boosting them to higher energy photons. Such impacts of photons on relativistic charged particle beams is another possible mechanism of gamma ray production. Neutron stars with a very high magnetic field (magnetars), thought to produce astronomical soft gamma repeaters, are another relatively long-lived star-powered source of gamma radiation.
- Quasars and active galaxies: More powerful gamma rays from very distant quasars and closer active galaxies are thought to have a gamma ray production source similar to a particle accelerator. High energy electrons produced by the quasar, and subjected to inverse Compton scattering, synchrotron radiation, or bremsstrahlung, are the likely source of the gamma rays from those objects. It is thought that as a supermassive black hole at the center of such galaxies provide the power source that intermittently destroys stars and focuses the resulting charged particles into beams that emerge from their rotational poles. When those beams interact with gas, dust, and lower energy photons they produce X-rays and gamma rays. These sources are known to fluctuate with durations of a few weeks, suggesting their relatively small size (less than a few light-weeks across). Such sources of gamma and X-rays are the most commonly visible high intensity sources outside our galaxy. They shine not in bursts (see illustration), but relatively continuously when viewed with gamma ray telescopes. The power of a typical quasar is about 1040 watts, a small fraction of which is gamma radiation. Much of the rest is emitted as electromagnetic waves of all frequencies, including radio waves.

- Gamma-ray bursts: The most intense sources of gamma rays, are also the most intense sources of any type of electromagnetic radiation presently known. They are the "long duration burst" sources of gamma rays in astronomy ("long" in this context, meaning a few tens of seconds), and they are rare compared with the sources discussed above. By contrast, "short" gamma-ray bursts, which are not associated with supernovae, are thought to produce gamma rays during the collision of pairs of neutron stars, or a neutron star and a black hole. Such bursts last two seconds or less, and are of far lower energy than the "long" bursts (only sources in our galaxy are detectable for that reason).[19]
The so-called long-duration gamma-ray bursts produce a total energy output of about 1044 joules (as much energy as our Sun will produce in its entire life-time) but in a period of only 20 to 40 seconds. Gamma rays are approximately 50% of the total energy output. The leading hypotheses for the mechanism of production of these highest-known intensity beams of radiation, are inverse Compton scattering and synchrotron radiation from high-energy charged particles. These processes occur as relativistic charged particles leave the region of the event horizon of a newly formed black hole created during supernova explosion. The beam of particles moving at relativistic speeds are focused for a few tens of seconds by the magnetic field of the exploding hypernova. The fusion explosion of the hypernova drives the energetics of the process. If the narrowly directed beam happens to be pointed toward the Earth, it shines at gamma ray frequencies with such intensity, that it can be detected even at distances of up to 10 billion light years, which is close to the edge of the visible universe.
Health effects
Gamma rays cause damage at a cellular level and are penetrating, causing diffuse damage throughout the body. However, they are less ionising than alpha or beta particles, which are less penetrating.
Low levels of gamma rays cause a stochastic health risk, which for radiation dose assessment is defined as the probability of cancer induction and genetic damage.[20] High doses produce deterministic effects, which is the severity of acute tissue damage that is certain to happen. These effects are compared to the physical quantity absorbed dose measured by the unit gray (Gy).[21]
Uses
Gamma rays provide information about some of the most energetic phenomena in the universe; however, they are largely absorbed by the Earth's atmosphere. Instruments aboard high-altitude balloons and satellites missions, such as the Fermi Gamma-ray Space Telescope, provide our only view of the universe in gamma rays.
Gamma-induced molecular changes can also be used to alter the properties of semi-precious stones, and is often used to change white topaz into blue topaz.
Non-contact industrial sensors commonly use sources of gamma radiation in refining, mining, chemicals, food, soaps and detergents, and pulp and paper industries, for the measurement of levels, density, and thicknesses. Typically, these use Co-60 or Cs-137 isotopes as the radiation source.
In the US, gamma ray detectors are beginning to be used as part of the Container Security Initiative (CSI). These machines are advertised to be able to scan 30 containers per hour.
Gamma radiation is often used to kill living organisms, in a process called irradiation. Applications of this include the sterilization of medical equipment (as an alternative to autoclaves or chemical means), the removal of decay-causing bacteria from many foods and the prevention of the sprouting of fruit and vegetables to maintain freshness and flavor.
Despite their cancer-causing properties, gamma rays are also used to treat some types of cancer, since the rays kill cancer cells also. In the procedure called gamma-knife surgery, multiple concentrated beams of gamma rays are directed to the growth in order to kill the cancerous cells. The beams are aimed from different angles to concentrate the radiation on the growth while minimizing damage to surrounding tissues.
Gamma rays are also used for diagnostic purposes in nuclear medicine in imaging techniques. A number of different gamma-emitting radioisotopes are used. For example, in a PET scan a radiolabeled sugar called fludeoxyglucose emits positrons that are annihilated by electrons, producing pairs of gamma rays that highlight cancer as the cancer often has a higher metabolic rate than the surrounding tissues. The most common gamma emitter used in medical applications is the nuclear isomer technetium-99m which emits gamma rays in the same energy range as diagnostic X-rays. When this radionuclide tracer is administered to a patient, a gamma camera can be used to form an image of the radioisotope's distribution by detecting the gamma radiation emitted (see also SPECT). Depending on which molecule has been labeled with the tracer, such techniques can be employed to diagnose a wide range of conditions (for example, the spread of cancer to the bones via bone scan).
Body response
When gamma radiation breaks DNA molecules, a cell may be able to repair the damaged genetic material, within limits. However, a study of Rothkamm and Lobrich has shown that this repair process works well after high-dose exposure but is much slower than in the case of a low-dose exposure.[22]
Risk assessment
The natural outdoor exposure in Great Britain ranges from 0.1 to 0.5 µSv/h with significant increase around known nuclear and contaminated sites.[23] Natural exposure to gamma rays is about 1 to 2 mSv per year, and the average total amount of radiation received in one year per inhabitant in the USA is 3.6 mSv.[24] There is a small increase in the dose, due to naturally occurring gamma radiation, around small particles of high atomic number materials in the human body caused by the photoelectric effect.[25]
By comparison, the radiation dose from chest radiography (about 0.06 mSv) is a fraction of the annual naturally occurring background radiation dose.[26] A chest CT delivers 5 to 8 mSv. A whole-body PET/CT scan can deliver 14 to 32 mSv depending on the protocol.[27] The dose from fluoroscopy of the stomach is much higher, approximately 50 mSv (14 times the annual background).
An acute full-body equivalent single exposure dose of 1 Sv (1000 mSv) causes slight blood changes, but 2.0–3.5 Sv (2.0–3.5 Gy) causes very severe syndrome of nausea, hair loss, and hemorrhaging, and will cause death in a sizable number of cases—-about 10% to 35% without medical treatment. A dose of 5 Sv[28] (5 Gy) is considered approximately the LD50 (lethal dose for 50% of exposed population) for an acute exposure to radiation even with standard medical treatment. A dose higher than 5 Sv (5 Gy) brings an increasing chance of death above 50%. Above 7.5–10 Sv (7.5–10 Gy) to the entire body, even extraordinary treatment, such as bone-marrow transplants, will not prevent the death of the individual exposed (see Radiation poisoning). (Doses much larger than this may, however, be delivered to selected parts of the body in the course of radiation therapy.)
For low dose exposure, for example among nuclear workers, who receive an average yearly radiation dose of 19 mSv, the risk of dying from cancer (excluding leukemia) increases by 2 percent. For a dose of 100 mSv, the risk increase is 10 percent. By comparison, risk of dying from cancer was increased by 32 percent for the survivors of the atomic bombing of Hiroshima and Nagasaki. [29]
See also
References
- ↑ Aharonian, F.; Akhperjanian, A.; Barrio, J.; Bernlohr, K.; Borst, H.; Bojahr, H.; Bolz, O.; Contreras, J.; Cortina, J.; Denninghoff, S.; Fonseca, V.; Gonzalez, J.; Gotting, N.; Heinzelmann, G.; Hermann, G.; Heusler, A.; Hofmann, W.; Horns, D.; Iserlohe, C.; Ibarra, A.; Jung, I.; Kankanyan, R.; Kestel, M.; Kettler, J.; Kohnle, A.; Konopelko, A.; Kornmeyer, H.; Kranich, D.; Krawczynski, H.; Lampeitl, H. (2001). "The TeV Energy Spectrum of Markarian 501 Measured with the Stereoscopic Telescope System of HEGRA during 1998 and 1999". The Astrophysical Journal. 546 (2): 898–902. Bibcode:2001ApJ...546..898A. doi:10.1086/318321.
- ↑ P. Villard (1900) "Sur la réflexion et la réfraction des rayons cathodiques et des rayons déviables du radium," Comptes rendus, vol. 130, pages 1010-1012. See also: P. Villard (1900) "Sur le rayonnement du radium," Comptes rendus, vol. 130, pages 1178-1179.
- ↑ L'Annunziata, Michael F. (2007). Radioactivity: introduction and history. Amsterdam, Netherlands: Elsevier BV. pp. 55–58. ISBN 978-0-444-52715-8.
- ↑ Rutherford named γ rays on page 177 of: E. Rutherford (1903) "The magnetic and electric deviation of the easily absorbed rays from radium," Philosophical Magazine, Series 6, vol. 5, no. 26, pages 177-187.
- 1 2 "Rays and Particles". Galileo.phys.virginia.edu. Retrieved 2013-08-27.
- 1 2 Dendy, P. P.; B. Heaton (1999). Physics for Diagnostic Radiology. USA: CRC Press. p. 12. ISBN 0-7503-0591-6.
- ↑ Charles Hodgman, Ed. (1961). CRC Handbook of Chemistry and Physics, 44th Ed. USA: Chemical Rubber Co. p. 2850.
- ↑ Feynman, Richard; Robert Leighton; Matthew Sands (1963). The Feynman Lectures on Physics, Vol.1. USA: Addison-Wesley. pp. 2–5. ISBN 0-201-02116-1.
- ↑ L'Annunziata, Michael; Mohammad Baradei (2003). Handbook of Radioactivity Analysis. Academic Press. p. 58. ISBN 0-12-436603-1.
- ↑ Grupen, Claus; G. Cowan; S. D. Eidelman; T. Stroh (2005). Astroparticle Physics. Springer. p. 109. ISBN 3-540-25312-2.
- ↑ "CGRO SSC >> EGRET Detection of Gamma Rays from the Moon". Heasarc.gsfc.nasa.gov. 2005-08-01. Retrieved 2011-11-08.
- ↑ "Bremsstrahlung radiation" is "braking radiation", but "acceleration" is being used here in the specific sense of the deflection of an electron from its course: Serway, Raymond A; et al. (2009). College Physics. Belmont, CA: Brooks Cole. p. 876. ISBN 978-0-03-023798-0.
- ↑ Shaw, R. W.; Young, J. P.; Cooper, S. P.; Webb, O. F. (1999). "Spontaneous Ultraviolet Emission from 233Uranium/229Thorium Samples". Physical Review Letters. 82 (6): 1109–1111. Bibcode:1999PhRvL..82.1109S. doi:10.1103/PhysRevLett.82.1109.
- ↑ "Gamma-Ray Telescopes & Detectors". NASA GSFC. Retrieved 2011-11-22.
- ↑ Bock, R. K.; et al. (2008-06-27). "Very-High-Energy Gamma Rays from a Distant Quasar: How Transparent Is the Universe?". Science. 320 (5884): 1752–1754. arXiv:0807.2822
 . Bibcode:2008Sci...320.1752M. doi:10.1126/science.1157087. ISSN 0036-8075. PMID 18583607.
. Bibcode:2008Sci...320.1752M. doi:10.1126/science.1157087. ISSN 0036-8075. PMID 18583607. - ↑ Domínguez, Alberto; et al. (2015-06-01). "All the Light There Ever Was". Scientific American. Vol. 312 no. 6. pp. 38–43. ISSN 0036-8075.
- ↑ Gamma decay review Accessed Sept. 29, 2014
- ↑ Smith, Joseph; David M. Smith (August 2012). "Deadly Rays From Clouds". Scientific American. Vol. 307 no. 2. pp. 55–59. Bibcode:2012SciAm.307b..54D. doi:10.1038/scientificamerican0812-54.
- ↑ Announcement of first close study of a short gamma-ray burst.
- ↑ The ICRP says "In the low dose range, below about 100 mSv, it is scientifically plausible to assume that the incidence of cancer or heritable effects will rise in direct proportion to an increase in the equivalent dose in the relevant organs and tissues" ICRP publication 103 paragraph 64
- ↑ ICRP report 103 para 104 and 105
- ↑ Rothkamm, K; Löbrich, M (2003). "Evidence for a lack of DNA double-strand break repair in human cells exposed to very low x-ray doses". Proceedings of the National Academy of Sciences of the United States of America. 100 (9): 5057–62. Bibcode:2003PNAS..100.5057R. doi:10.1073/pnas.0830918100. PMC 154297
 . PMID 12679524.
. PMID 12679524. - ↑ ENVIRONMENT AGENCY UK Radioactivity in Food and the Environment, 2012
- ↑ United Nations Scientific Committee on the Effects of Atomic Radiation Annex E: Medical radiation exposures – Sources and Effects of Ionizing – 1993, p. 249, New York, UN
- ↑ Pattison, J. E.; Hugtenburg, R. P.; Green, S. (2009). "Enhancement of natural background gamma-radiation dose around uranium microparticles in the human body". Journal of the Royal Society Interface. 7 (45): 603–611. doi:10.1098/rsif.2009.0300.
- ↑ US National Council on Radiation Protection and Measurements – NCRP Report No. 93 – pp 53–55, 1987. Bethesda, Maryland, USA, NCRP
- ↑ "PET/CT total radiation dose calculations. Accessed June 23, 2011." (PDF). Retrieved 2011-11-08.
- ↑ "Lethal dose", NRC Glossary (October 18, 2011)
- ↑ Cardis, E (9 July 2005). "Risk of cancer after low doses of ionising radiation: retrospective cohort study in 15 countries". BMJ. 331 (7508): 77–0. doi:10.1136/bmj.38499.599861.E0.
External links
- Basic reference on several types of radiation
- Radiation Q & A
- GCSE information
- Radiation information
- Gamma-ray bursts
- The Lund/LBNL Nuclear Data Search – Contains information on gamma-ray energies from isotopes.
- Mapping soils with airborne detectors
 The LIVEChart of Nuclides – IAEA with filter on gamma-ray energy
The LIVEChart of Nuclides – IAEA with filter on gamma-ray energy- Health Physics Society Public Education Website

