Magnesium chloride
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Magnesium chloride | |
| Systematic IUPAC name
Magnesium chloride | |
| Other names
Magnesium dichloride | |
| Identifiers | |
| 7786-30-3 7791-18-6 (hexahydrate) | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:6636 |
| ChEMBL | ChEMBL1200547 |
| ChemSpider | 22987 |
| ECHA InfoCard | 100.029.176 |
| E number | E511 (acidity regulators, ...) |
| 9305 | |
| PubChem | 24584 |
| RTECS number | OM2975000 |
| |
| |
| Properties | |
| MgCl2 | |
| Molar mass | 95.211 g/mol (anhydrous) 203.31 g/mol (hexahydrate) |
| Appearance | white or colourless crystalline solid |
| Density | 2.32 g/cm3 (anhydrous) 1.569 g/cm3 (hexahydrate) |
| Melting point | 714 °C (1,317 °F; 987 K) 117 °C (243 °F; 390 K) (hexahydrate) on rapid heating: slow heating leads to decomposition from 300 °C (572 °F; 573 K) |
| Boiling point | 1,412 °C (2,574 °F; 1,685 K) |
| anhydrous 52.9 g/100 mL (0 °C) 54.3 g/100 mL (20 °C) 72.6 g/100 mL (100 °C) hexahydrate 167 g/100 mL (20 °C) | |
| Solubility | slightly soluble in acetone, pyridine |
| Solubility in ethanol | 7.4 g/100 mL (30 °C) |
| Refractive index (nD) |
1.675 (anhydrous) 1.569 (hexahydrate) |
| Structure | |
| CdCl2 | |
| (octahedral, 6-coordinate) | |
| Thermochemistry | |
| 71.09 J/mol K | |
| Std molar entropy (S |
89.88 J/mol K |
| Std enthalpy of formation (ΔfH |
-641.1 kJ/mol |
| Gibbs free energy (ΔfG˚) |
-591.6 kJ/mol |
| Pharmacology | |
| A12CC01 (WHO) B05XA11 (WHO) | |
| Hazards | |
| Main hazards | Irritant |
| Safety data sheet | ICSC 0764 |
| R-phrases | R36, R37, R38 |
| S-phrases | S26, S37, S39 |
| NFPA 704 | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
| LD50 (median dose) |
2800 mg/kg (oral, rat) |
| Related compounds | |
| Other anions |
Magnesium fluoride Magnesium bromide Magnesium iodide |
| Other cations |
Beryllium chloride Calcium chloride Strontium chloride Barium chloride Radium chloride |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Magnesium chloride is the name for the chemical compound with the formula MgCl2 and its various hydrates MgCl2(H2O)x. These salts are typical ionic halides, being highly soluble in water. The hydrated magnesium chloride can be extracted from brine or sea water. In North America, magnesium chloride is produced primarily from Great Salt Lake brine. It is extracted in a similar process from the Dead Sea in the Jordan valley. Magnesium chloride, as the natural mineral bischofite, is also extracted (via solution mining) out of ancient seabeds; for example, the Zechstein seabed in northwest Europe. Some magnesium chloride is made from solar evaporation of seawater. Anhydrous magnesium chloride is the principal precursor to magnesium metal, which is produced on a large scale. Hydrated magnesium chloride is the form most readily available.
Structure, preparation, and general properties
MgCl2 crystallizes in the cadmium chloride motif, which features octahedral Mg. A variety of hydrates are known with the formula MgCl2(H2O)x, and each loses water with increasing temperature: x = 12 (−16.4 °C), 8 (−3.4 °C), 6 (116.7 °C), 4 (181 °C), 2 (ca. 300 °C).[1] In the hexahydrate, the Mg2+ remains octahedral, but is coordinated to six water ligands.[2] The thermal dehydration of the hydrates MgCl2(H2O)x (x = 6, 12) does not occur straightforwardly.[3]
As suggested by the existence of some hydrates, anhydrous MgCl2 is a Lewis acid, although a very weak one.
In the Dow process, magnesium chloride is regenerated from magnesium hydroxide using hydrochloric acid:
It can also be prepared from magnesium carbonate by a similar reaction.
In most of its derivatives, MgCl2 forms octahedral complexes. Derivatives with tetrahedral Mg2+ are less common. Examples include salts of (tetraethylammonium)2MgCl4 and adducts such as MgCl(TMEDA).[4]
Applications
Magnesium chloride serves as precursor to other magnesium compounds, for example by precipitation:
It can be electrolysed to give magnesium metal:[5]
- MgCl2(l) → Mg(l) + Cl2(g)
This process is practiced on a substantial scale.
Magnesium chloride is most commonly used for dust control and road stabilization. Its second-most common use is ice control. In addition to the production of magnesium metal, magnesium chloride also is used for a variety of other applications: fertilizer, mineral supplement for animals, wastewater treatment, wallboard, artificial seawater, feed supplement, textiles, paper, fireproofing agents, cements and refrigeration brine. Mixed with hydrated magnesium oxide, magnesium chloride forms a hard material called Sorel cement.
This compound is also used in fire extinguishers: obtained by the reaction of magnesium hydroxide and hydrochloric acid (HCl) in liquid form along with water in gaseous state.[6][7] Magnesium chloride also is used in several medical and topical (skin related) applications. It has been used in pills as supplemental sources of magnesium, where it serves as a soluble compound that is not as laxative as magnesium sulfate, and more bioavailable than magnesium hydroxide and magnesium oxide, since it does not require stomach acid to produce soluble Mg2+ ion. It can also be used as an effective anesthetic for cephalopods,[8] some species of crustaceans,[9] and several species of bivalve, including oysters.[10]
MgCl2 is also commonly utilized in the polymerase chain reaction(PCR). The magnesium ion is necessary for both in vivo/vitro DNA synthesis.
Use in dust and erosion control
Magnesium chloride is one of many substances used for dust control, soil stabilization and wind erosion mitigation.[11] When magnesium chloride is applied to roads and bare soil areas, both positive and negative performance issues occur which are related to many application factors.[12] Water absorbing magnesium chloride (deliquescent) attributes include
- it starts to absorb water from the air at 32% relative humidity, almost independent of temperature,
- more effective than calcium chloride solutions for increasing surface tension,
- treated roads can be regraded and re-compacted with less concern for losing moisture and density.
However, limitations include
- a minimum humidity level is required to absorb moisture from the air,
- it is more suitable in drier climates,
- in concentrated solutions it is very corrosive,
- it attracts moisture thereby prolonging the active period for corrosion,
- rainwater tends to leach out highly soluble chlorides,
- if there is a high fines content in treated material then the surface may become slippery when wet,
- when less than 20% solution it has performance effectiveness similar to water.[11][12]
The use of magnesium chloride on roads remains controversial. Advocates claim (1) Cleaner air, which leads to better health as fugitive dust can cause health problems in the young, elderly and people with respiratory conditions;[13] and (2) Greater safety through improved road conditions,[14][15] including increased driver visibility and decreased risks caused by loose gravel, soft spots, road roughness and flying rocks.[16] It reduces foreign sediment in nearby surface waters [17] (dust that settles in creeks and streams), helps prevent stunted crop growth caused by clogged pores in plants, and keeps vehicles and property cleaner.[18] Other studies show the use of salts for road deicing or dust suppressing can contribute substantial amounts of chloride ions to runoff from surface of roads treated with the compounds. The salts MgCl2 (and CaCl2) are very soluble in water and will dissociate.[19] The salts, when used on road surfaces, will dissolve during wet weather and be transported into the groundwater through infiltration and/or runoff into surface water bodies.[15] Groundwater infiltration can be a problem and the chloride ion in drinking water is considered a problem when concentrations exceed 250 mg/l. It is therefore is regulated by the EPA’s drinking water standards. The chloride concentration in the groundwater or surface water depends on several factors including:
- application rate,
- composition and type of soil,
- type, intensity, and amount of precipitation,
- the drainage of the road system.[20]
In addition, the chloride concentration in the surface water also depends on the size or flow rate of the water body and the resulting dilution achieved. In chloride concentration studies carried out in Wisconsin during a winter deicing period, runoff from roadside drainages were analyzed. All studies indicated that the chloride concentration increased as a result of deicing activities but the levels were still below the MCL of 250 mg/L set by the EPA.[21][22][23][24][25] Nevertheless, the long-term effect of this exposure is not known.
Although the EPA has set the maximum chloride concentration in water for domestic use at 250 mg/l animals can tolerate higher levels. At excessively high levels, chloride is said to affect the health of animals.[26] As stated by the National Technical Advisory Committee to the Secretary of Interior (1968), “Salinity may have a two-fold effect on wildlife; a direct one affecting the body processes of the species involved and an indirect one altering the environment making living species perpetuation difficult or impossible.” One major problem associated with the use of deicing salt as far as wildlife is concerned is that wildlife are known to have “salt craving” and therefore are attracted to salted highways which can be a traffic hazard to both the animals and motorists.
Regarding the accumulation of chloride salts in roadside soils including the adverse effects on roadside plants and vegetation physiology and morphology, documentation dates back to World War II era times [27] and consistently continues forward to present times.[28] As far as plants and vegetation are concerned, the accumulation of salts in the soil adversely affects their physiology and morphology by: increasing the osmotic pressure of the soil solution, by altering the plant’s mineral nutrition, and by accumulating specific ions to toxic concentrations in the plants. Regarding the intentional application of excessive salts: see Salting the Earth.
Road departments and private industry may apply liquid or powdered magnesium chloride to control dust and erosion on unimproved (dirt or gravel) roads and dusty job sites such as quarries because it is relatively inexpensive to purchase and apply. Its hygroscopy makes it absorb moisture from the air, limiting the number of smaller particles (silts and clays) that become airborne. The most significant benefit of applying dust control products is the reduction in gravel road maintenance costs.[29] However, recent research and updates indicate biological toxicity in the environment in plants as an ongoing problem.[28] Since 2001, truckers have complained about "Killer Chemicals" on roads and now some states are backing away from using salt products.[30][31]
Also a small percentage of owners of indoor arenas (e.g. for horse riding) may apply magnesium chloride to sand or other "footing" materials to control dust. Although magnesium chloride use in an equestrian (horse) arena environment is generally referred to as a dust suppressant it is technically more accurate to consider it as a water augmentation activity since its performance is based on absorbing moisture from the air and from whatever else comes in contact with it.
To control or mitigate dust, chlorides need moisture to work effectively so it works better in humid than arid climates. As the humidity increases the chloride draw moisture out of the air to keep the surface damp and as humidity decreases it diffuses and releases moisture. These naturally occurring equilibrium changes also allow chlorides to also be used as a dehydrating agent including the drying out of and curing and preservation of hides.[32]
As a road stabilizer, magnesium chloride binds gravel and clay particles to keep them from leaving the road. The water-absorbing (hygroscopic) characteristics of magnesium chloride prevent the road from drying out, which keeps gravel on the ground. The road remains continually "wet" as if a water truck had just sprayed the road.[33]
Use in ice control


Magnesium chloride is used for low-temperature de-icing of highways, sidewalks, and parking lots. When highways are treacherous due to icy conditions, magnesium chloride helps to prevent the ice bond, allowing snow plows to clear the roads more efficiently.
Magnesium chloride is used in three ways for pavement ice control: Anti-icing, when maintenance professionals spread it onto roads before a snow storm to prevent snow from sticking and ice from forming; pre-wetting, which means a liquid formulation of magnesium chloride is sprayed directly onto salt as it is being spread onto roadway pavement, wetting the salt so that it sticks to the road; and pre-treating, when magnesium chloride and salt are mixed together before they are loaded onto trucks and spread onto paved roads.
While it is generally accepted that ongoing use of any de-icer (ice melter) will eventually contribute to some degradation of the concrete surface to which it is applied, some de-icers are gentler on concrete than others. Conflicting information in regards to magnesium chloride- and calcium chloride-based liquid de-icers have left many unsure which choice is best-suited for their needs.
Past studies have often utilized high temperatures to accelerate the impact to concrete. By setting parameters that more closely represent real-world de-icing conditions, Purdue University researchers measured the impact of magnesium chloride and calcium chloride to concrete. Their study concluded that calcium chloride damages concrete twice as fast as magnesium chloride.[34]
Nutritional supplement
Magnesium chloride is a type of neutraceutical preparation of magnesium.
Use in hydrogen storage
Magnesium chloride has shown promise as a storage material for hydrogen. Ammonia, which is rich in hydrogen atoms, is used as an intermediate storage material. Ammonia can be effectively absorbed onto solid magnesium chloride, forming Mg(NH3)6Cl2. Ammonia is released by mild heat, and is then passed through a catalyst to give hydrogen gas.[35]
Niche uses
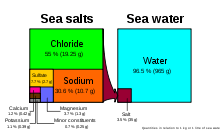
Magnesium in natural seawater values are between 1250 mg/L and 1350 mg/L. (approximately 3.7% of the total seawater mineral content. See thumbnail chart.) Dead Sea minerals contain a significantly higher magnesium chloride ratio (50.8%). Carbonates and calcium are essential for all growth of corals, coralline algae, clams, and invertebrates. Magnesium can be depleted by mangrove plants and the use of excessive limewater or by going beyond natural calcium, alkalinity, and pH values.[36]
Culinary use
Magnesium chloride (E511[37]) is an important coagulant used in the preparation of tofu from soy milk. In Japan it is sold as nigari (にがり, derived from the Japanese word for "bitter"), a white powder produced from seawater after the sodium chloride has been removed, and the water evaporated. In China, it is called lushui (卤水). Nigari or lushui consists mostly of magnesium chloride, with some magnesium sulfate and other trace elements. It is also an ingredient in baby formula milk.[38]
Gardening and horticulture
Because magnesium is a mobile nutrient, magnesium chloride can be effectively used as a substitute for magnesium sulfate (Epsom salt) to help correct magnesium deficiency in plants via foliar feeding. It is advisable to use a smaller dose when using magnesium chloride compared to the recommended dose of magnesium sulfate (20 g/L).[39] This is due primarily to the chlorine present in magnesium chloride, which can easily reach toxic levels if over-applied and/or applied too often.[40]
It has been found that higher concentrations of magnesium in tomato and some pepper plants can make them more susceptible to disease caused by infection of the bacteria Xanthomonas campestris, since Mg is essential for bacterial growth.[41]
Toxicology
Magnesium ions are bitter-tasting, and magnesium chloride solutions are bitter in varying degrees, depending on the concentration of magnesium.
Magnesium toxicity from magnesium salts is rare in healthy individuals with a normal diet, because excess magnesium is readily excreted in urine by the kidneys. A few cases of oral magnesium toxicity have been described in persons with normal renal function ingesting large amounts of magnesium salts, but it is rare. If a large amount of magnesium chloride is eaten, it will have effects similar to magnesium sulfate, causing diarrhea, although the sulfate also contributes to the laxative effect in magnesium sulfate, so the effect from the chloride is not as severe.
Plant Toxicity: Chloride (Cl−) and magnesium (Mg2+) are both essential nutrients important for normal plant growth. Too much of either nutrient may harm a plant, although foliar chloride concentrations are more strongly related with foliar damage than magnesium. High concentrations of MgCl2 ions in the soil may be toxic or change water relationships such that the plant cannot easily accumulate water and nutrients. Once inside the plant, chloride moves through the water-conducting system and accumulates at the margins of leaves or needles, where dieback occurs first. Leaves are weakened or killed, which can lead to the death of the tree.[28][42]
Eco Toxicity levels related to terrestrial and aquatic organisms for magnesium chloride are listed in the Pesticide Action Network Pesticide Database.[43][44]
Locomotive boiler problem
The presence of dissolved magnesium chloride in the well water (bore water) used in locomotive boilers on the Trans-Australian Railway caused serious and expensive maintenance problems during the steam era. At no point along its route does the line cross a permanent fresh watercourse, so bore water had to be relied on. No inexpensive treatment for the highly mineralised water was available and locomotive boilers were lasting less than a quarter of the time normally expected.[45] In the days of steam locomotion, about half the total train load was water for the engine. The line's operator, Commonwealth Railways was an early adopter of the diesel-electric locomotive.
See also
Notes & references
- Notes
- ↑ Holleman, A. F.; Wiberg, E. Inorganic Chemistry Academic Press: San Diego, 2001. ISBN 0-12-352651-5.
- ↑ Wells, A. F. (1984) Structural Inorganic Chemistry, Oxford: Clarendon Press. ISBN 0-19-855370-6.
- ↑ see notes in Rieke, R. D.; Bales, S. E.; Hudnall, P. M.; Burns, T. P.; Poindexter, G. S. "Highly Reactive Magnesium for the Preparation of Grignard Reagents: 1-Norbornane Acid", Organic Syntheses, Collected Volume 6, p. 845 (1988). http://www.orgsyn.org/orgsyn/pdfs/CV6P0845.pdf
- ↑ N. N. Greenwood, A. Earnshaw, Chemistry of the Elements, Pergamon Press, 1984.
- ↑ Hill, Petrucci, McCreary, Perry, General Chemistry, 4th ed., Pearson/Prentice Hall, Upper Saddle River, New Jersey, USA.
- ↑ http://www.ask.com/question/what-is-magnesium-chloride
- ↑ http://www.ilo.org/oshenc/part-ix/metals-chemical-properties-and-toxicity/item/142-magnesium
- ↑ Messenger JB, Nixon M, Ryan KP. (1985 ). Magnesium chloride as an anaesthetic for cephalopods. Comp Biochem Physiol C. 82(1):203-5.
- ↑ Archived October 10, 2009, at the Wayback Machine.
- ↑ Culloty, S. C. & Mukahy, M. F. 1992. An evaluation of anaesthetics for Ostrea edulis (L.). Aquaculture. 107: 249–252.
- 1 2 http://www.fs.fed.us/eng/pubs/html/99771207/99771207.html#EI
- 1 2 http://www.nrcs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb1043546.pdf
- ↑ Schwendeman, T., Dust Control Study, Dust Palliative Evaluation, Gallatin National Forest”, USDA Forest Service, 1981
- ↑ http://www.cdc.gov/niosh/nas/rdrp/appendices/chapter3/a3-23.pdf
- 1 2 http://www.mountain-plains.org/pubs/pdf/MPC04-156.pdf
- ↑ Lohnes, R.A. and Coree, B.J., Determination and Evaluation of Alternate Methods for Managing and Controlling Highway-related Dust, Dept of Civil and Construction Engineering, Iowa State University, 2002
- ↑ Hass, R.A.., “Dustproofing Unsurfaced Tank Trails Grafenwohr Training Area, Federal Republic of Germany. June 15–29, 1985.” U. S. Army Corps of Engineers, Paper GL-86-40, 1986; the appendix of this report summarized the environmental effects of the use of magnesium chloride, saying: "A comprehensive literature search (Toxline, Medline, Chemline, Hazard Lie, Biological Abstracts, Toxic Data Bank and other available sources) was made. There appears to be no reported evidence that MgCl2 has had or will produce any effects on the groundwater, the water table, or vegetation following single or repeated applications to soil."
- ↑ Han, C. Dust Control on Unpaved Roads, Minnesota Local Road Research Board, 1992
- ↑ Snoeyink, V.L. and D. Jenkins. Water Chemistry. John Wiley & Sons, Inc., New York. 1980
- ↑ Pollock, S.J., and L.G. Toler. Effects of Highway Deicing Salts on Groundwater and Water Supplies in Massachusetts. Highway Research Board, No. 425 17-21. 1973
- ↑ Schraufnagel, F.H. Chlorides. Commission on Water Pollution, Madison, Wisconsin. 1965.
- ↑ Hutchinson, F.E. The Influences of Salts Applied to Highways on the Levels of Sodium and Chloride Ions Present in Water and Soil Samples – Progress Report I. Project No. R1084-8. 1966.
- ↑ Pollock, S.J., and L.G. Toler. Effects of Highway Deicing Salts on Groundwater and Water Supplies in Massachusetts. Highway Research Board, No. 425 17-21. 1973.
- ↑ Hutchinson, F.E. The Influences of Salts Applied to Highways on the Levels of Sodium and Chloride Ions Present in Water and Soil Samples – Progress Report I. Project No.R1084-8. 1966.
- ↑ Schraufnagel, F.H. Chlorides. Commission on Water Pollution, Madison, Wisconsin. 1965
- ↑ Heller, V.G. “Saline and Alkaline Drinking Waters.” Journal of Nutrition, 5:421-429 1932
- ↑ Strong, F.C. A Study of Calcium Chloride Injury to Roadside Trees. Michigan Agr. Exp. Station, Quarterly Bulletin, 27:209-224. 1944
- 1 2 3 http://www.ext.colostate.edu/pubs/garden/07425.html
- ↑ http://www.epa.gov/owow/NPS/gravelroads/sec4.pdf
- ↑ http://www.truckinginfo.com/blog/all-thats-trucking/story/2011/12/some-states-are-backing-away-from-killer-chemical-de-icers.aspx
- ↑ http://www.truckinginfo.com/magazine/issue/2001/09.aspx
- ↑ http://wyndmoor.arserrc.gov/Page/1999%5C6706.pdf
- ↑ http://www.soilworks.com/docs/soilworks-army-dust-control-field-handbook-2006.pdf
- ↑
- Jain, J., Olek, J., Janusz, A., and Jozwiak-Niedzwiedzka, D., "Effects of Deicing Salt Solutions on Physical Properties of Pavement Concretes", Transportation Research Record: Journal of the Transportation Research Board, No. 2290, Transportation Research Board of the National Academies, Washington, D.C., 2012, pp. 69-75. DOI: 10.3141/2290-09
- ↑
- Christensen, C. H., Sorensen, R. Z., Johannessen, T., Quaade,U. J.,Honkala, K., Elmøe, T. D., Køhler, R., and Nørskov, J. K., "Metal ammine complexes for hydrogen storage, " J. Mater. Chem. 15(2005) 4106–4108
- ↑ "Aquarium Chemistry: Magnesium In Reef Aquaria — Advanced Aquarist | Aquarist Magazine and Blog". Advancedaquarist.com. 2003-10-15. Retrieved 2013-01-17.
- ↑ Food Standard Agency. "Current EU approved additives and their E Numbers". Retrieved 22 March 2010.
- ↑ "Listed under ingredients for Similac Hypoallergenic Infant Formula with Iron (Abbott Nutrition)". abbottnutrition.com. Retrieved 2013-07-22.
- ↑ Comparison of Magnesium Sulfate and THIS Mg Chelate Foliar Sprays
- ↑ Magnesium Chloride Toxicity in Trees
- ↑ Effect of Foliar and Soil Magnesium Application on Bacterial Leaf Spot of Peppers
- ↑ Pesticide Action Network Database, http://www.pesticideinfo.org/Detail_Chemical.jsp?Rec_Id=PC32916
- ↑ http://www.pesticideinfo.org/Detail_Chemical.jsp?Rec_Id=PC32916#Ecotoxicity
- ↑ http://www.pesticideinfo.org/Docs/ref_ecotoxicity3.html#AvgGroupToxicity
- ↑ "Overland Locomotive:Feed Water Problems". The Argus. 1927-03-21. Retrieved 2014-03-11.
- References
- Handbook of Chemistry and Physics, 71st edition, CRC Press, Ann Arbor, Michigan, 1990.
