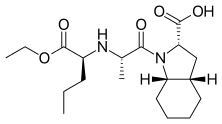
Perindopril
 | |
| Clinical data | |
|---|---|
| Trade names | Coversyl, Coversum, Preterax, Aceon |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a602017 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code |
C09AA04 (WHO) C09BA04 (WHO) (with diuretics) C09BB04 (WHO) (with amlodipine) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 24% |
| Protein binding | 20% |
| Metabolism | Renal |
| Biological half-life | 1–17 hours for perindoprilat (active metabolite) |
| Identifiers | |
| |
| CAS Number |
82834-16-0 |
| PubChem (CID) | 107807 |
| IUPHAR/BPS | 6367 |
| DrugBank |
DB00790 |
| ChemSpider |
96956 |
| UNII |
1964X464OJ |
| KEGG |
D03753 |
| ChEBI |
CHEBI:8024 |
| ChEMBL |
CHEMBL1581 |
| ECHA InfoCard | 100.124.027 |
| Chemical and physical data | |
| Formula | C19H32N2O5 |
| Molar mass | 368.468 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Perindopril is a long-acting ACE inhibitor used to treat high blood pressure, heart failure, or stable coronary artery disease[1] in form of perindopril arginine (trade names include Coversyl, Coversum) or perindopril erbumine (Aceon). According to the Australian government's Pharmaceutical Benefits Scheme website, based on data provided to the Australian Department of Health and Aging by the manufacturer, perindopril arginine and perindopril erbumine are therapeutically equivalent and may be interchanged without differences in clinical effect.[2] However, the dose prescribed to achieve the same effect differs due to different molecular weights for the two forms.
Medical uses
Perindopril shares the indications of ACE inhibitors as a class, including essential hypertension, stable coronary artery disease (reduction of risk of cardiac events in patients with a history of myocardial infarction and/or revascularization) and treatment of symptomatic heart disease or failure. In addition, the Perindopril pROtection aGainst REcurrent Stroke Study (PROGRESS) found that perindopril reduces the risk of stroke in both hypertensive and nonhypertensive individuals with a history of stroke or transient ischemic attack.[3]
The Anglo-Scandinavian Cardiac Outcomes Trial showed the benefits of taking the two drugs perindopril and amlodipine together. About 9000 British patients aged 40 to 79 were involved in the five-year trial. Half were given the new drug combination; the rest were given traditional drugs. Perindopril and amlodipine were found to be so effective, the trial was stopped early so all patients could receive the combination.[4][5]
Composition
Each tablet contains 2, 4, or 8 mg of the tert-butylamine salt of perindopril. Perindopril is also available under the trade name Coversyl Plus, containing 4 mg of perindopril combined with 1.25 mg indapamide.
In Australia, each tablet contains 2.5, 5, or 10 mg of perindopril arginine. Perindopril is also available under the trade name Coversyl Plus, containing 5 mg of perindopril arginine combined with 1.25 mg indapamide and Coversyl Plus LD, containing 2.5 mg of perindopril arginine combined with 0.625 mg indapamide.
The efficacy and tolerability of a fixed-dose combination of 4 mg perindopril and 5 mg amlodipine, a calcium channel antagonist, has been confirmed in a prospective, observational multicenter trial of 1,250 hypertensive patients.[6] A preparation of the two drugs is available commercially as Coveram.
Trade names
Also, it is available under the brand names:
- Aceon
- Acertil
- Actiprex
- Armix
- Coverene
- Coverex
- Coversum
- Coversyl
- Covinace
- Perineva
- Prenessa
- Prestarium
- Preterax
- Prexanil
- Prexum
- Procaptan
- Provinace
- Pericard
- Percarnil
Contraindications
- Children
- Pregnancy
- Lactation
- Situations where a patient has a history of hypersensitivity
- Renal failure
Precautions
- Assess renal function before and during treatment where appropriate.
- Renovascular hypertension
- Surgery/anesthesia
- Renal failure: the dose should be cautiously adjusted in accordance with the creatinine clearance (refer to complete data sheet).
- Symptomatic hypotension is rarely seen, but is more likely in volume-depleted patients, those receiving diuretics, or with the first two doses. In diuretic-treated patients, stop the diuretic three days before starting perindopril. A diuretic may later be given in combination if necessary; potassium-sparing diuretics are not recommended. Combination with neuroleptics or imipramine-type drugs may increase the hypotensive effect. Serum lithium concentrations may rise during lithium therapy.
Side effects
Side effects are mild, usually at the start of treatment; they include:
- Cough
- Fatigue
- Asthenia
- Headache
- Disturbances of mood and/or sleep
Less often
- Taste impairment
- Epigastric discomfort
- Nausea
- Abdominal pain
- Rash
Reversible increases in blood urea and creatinine may be observed. Proteinuria has occurred in some patients. Rarely, angioneurotic edema and decreases in hemoglobin, red cells, and platelets have been reported.
Marketing
On 9 July 2014, the European Commission imposed fines of €427,700,000 on Laboratoires Servier and 5 companies which produce generics due to Servier's abuse of their dominant market position, in breach of European Union Competition law. Servier's strategy had included acquiring the principal source of generic production of Perindopril and entering into several pay-for-delay agreements with potential generic competitors.[7]
References
- ↑ Royal Australian College of General Practitioners. "Consumer Medicine Information, GenRx Perindopril" (PDF). Clinical Resources, Medicine information for health professionals.
- ↑ Australian Government Department of Health and Ageing (2008). "PBS For Health Professionals". Pharmaceutical Benefits Scheme. Retrieved 2008-09-04.
- ↑ PROGRESS Collaborative Group. "Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack". The Lancet. 2001 Sep 29;. 358 (9287): 1033–1041. doi:10.1016/s0140-6736(01)06178-5. PMID 11589932.
- ↑ Charlotte Harding (2005). "What you need to know about the new wonder drug cocktail for high blood pressure...". JADN Repository. Retrieved 2007-07-01.
- ↑ Linda Brookes (2003). "ASCOT: Anglo-Scandinavian Cardiac Outcomes Trial -- Results From The Lipid-Lowering Arm". Medscape Today. Retrieved 2007-07-01.
- ↑ Bahl VK, Jadhav UM, Thacker HP. Management of Hypertension with the Fixed Combination of Perindopril and Amlodipine in Daily Clinical Practice: Results from the STRONG Prospective, Observational, Multicenter Study. American Journal of Cardiovascular Drugs May 22, 2009; 9 (3): 135-42 Link text
- ↑ http://europa.eu/rapid/press-release_IP-14-799_en.htm
Further reading
- Bounhoure JP, Bottineau G, Lechat P, et al.. "Value of perindopril in the treatment of chronic congestive heart failure: multicentre double-blind placebo-controlled study." Clin Exp Hypertens. 1989;A11(suppl 2):575-586.
- Lechat P, Granham SP, Desche P, et al.. "Efficacy and acceptability of perindopril in mild-to-moderate chronic congestive heart failure." Am Heart J. 1993;126:798-806.
- Morgan T and Anderson A; "Clinical efficacy of perindopril in hypertension." Clin Exp Pharmacol Physiol. 1992;19:61-65.
- Myers MG; (on behalf of the perindopril multicentre dose-response study group) "A dose-response study of perindopril in hypertension: effects on blood pressure 6 and 24h after dosing." Can J Cardiol. 1996;12:1191-1196.
- "The European trial on reduction of cardiac events with perindopril in stable coronary artery disease investigators. Efficacy of perindopril in reduction of cardiovascular events among patients with stable coronary artery disease: randomized, double-blind, placebo controlled, multicentre trial (the EUROPA study)." The Lancet 2003;362:782-788.