Phosphorus trioxide
 Phosphorus in orange, oxygen in red | |
 | |
| Names | |
|---|---|
| Other names
Phosphorus(III) oxide Phosphorus sesquioxide Phosphorous oxide Phosphorous anhydride Tetraphosphorous hexoxide | |
| Identifiers | |
| 1314-24-5 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:37372 |
| ChemSpider | 109897 |
| PubChem | 123290 |
| |
| |
| Properties | |
| P4O6 | |
| Molar mass | 219.88 g mol−1 |
| Appearance | colourless monoclinic crystals or liquid |
| Density | 2.135 g/cm3 |
| Melting point | 23.8 °C (74.8 °F; 296.9 K) |
| Boiling point | 173.1 °C (343.6 °F; 446.2 K) |
| reacts | |
| Acidity (pKa) | 9.4 |
| Structure | |
| See Text | |
| 0 | |
| Related compounds | |
| Other anions |
Phosphorus trisulfide |
| Other cations |
Dinitrogen trioxide Arsenic trioxide Antimony trioxide |
| Related compounds |
Phosphorus pentoxide Phosphorous acid |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Phosphorus trioxide is the chemical compound with the molecular formula P4O6. Although it should properly be named tetraphosphorus hexoxide, the name phosphorus trioxide preceded the knowledge of the compound's molecular structure, and its usage continues today. This colorless solid is structurally related to adamantane. It is formally the anhydride of phosphorous acid, H3PO3, but cannot be obtained by the dehydration of the acid. It is a white, waxy, crystalline and highly toxic solid.[1]
Preparation
It is obtained by the combustion of phosphorus in a limited supply of air at low temperature.
- P4(s) + 3 O2(g) → P4O6(s)
By-products include red phosphorus suboxide.[1]
Chemical Properties
Phosphorus trioxide reacts with cold water to form phosphorous acid.
- P4O6(s) + 6 H2O(l) → 4 H3PO3(aq)
It reacts vigorously with hot water, via a complex set of reactions, to form red phosphorus, phosphines, H3PO3 and H3PO4.
P4O6 reacts with hydrogen chloride to form H3PO3 and phosphorus trichloride.
- P4O6 + 6 HCl → 2 H3PO3 + 2 PCl3
With chlorine or bromine it forms the corresponding phosphoryl halide, and it reacts with iodine in a sealed tube to from diphosphorus tetraiodide.[1]
When P4O6 is allowed to react with ozone at 195 K in dichloromethane, the highly unstable compound P4O18 is formed.[2]
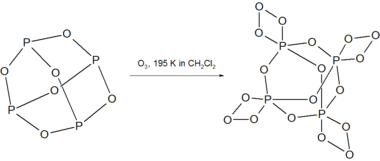
P4O18 decomposes above 238 K in solution with the release of O2 gas. Decomposition of dry P4O18 is explosive.
P4O6 is converted into the mixed P(III)P(V) species P4O8 when heated in a sealed tube at 710 K, with the side product being red phosphorus.[2]
As a ligand
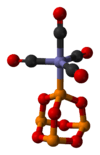
P4O6 is a ligand for transition metals, comparable to phosphite. Tetracarbonyl(tetraphosphorus hexaoxide)iron, P4O6·Fe(CO)4, is an example of a complex containing the phosphorus trioxide cage as a ligand. Its molecular structure as determined by single-crystal X-ray diffraction[3] is shown below:
P4O6 can also donate electrons to Lewis acids such as BH3. Adducts with one to two equivalents of BH3 complexed with one mole of P4O6 has been reported. When P4O6 is reacted with one molar equivalent of (CH3)2S•BH3, followed by slow crystallization from the toluene solution at 244K, P8O12(BH3)2 rather than an adduct of P4O6 is formed:[2]
References
- 1 2 3 A. F. Holleman; Wiberg, Egon; Wiberg, Nils (2001). Inorganic Chemistry. Boston: Academic Press. ISBN 0-12-352651-5.
- 1 2 3 .Catherine E. Housecroft; Alan G. Sharpe (2008). "Chapter 15: The group 15 elements". Inorganic Chemistry, 3rd Edition. Pearson. p. 473. ISBN 978-0-13-175553-6.
- ↑ M. Jansen & J. Clade (November 1996). "Tetracarbonyl(tetraphosphorus hexaoxide)iron". Acta Crystallogr. C. 52 (11): 2650–2652. doi:10.1107/S0108270196004398.
'
