Nitric oxide
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Nitric oxide | |||
| Systematic IUPAC name
Oxidonitrogen(•)[1] (additive) | |||
| Other names
Nitrogen monoxide Nitrogen(II) oxide | |||
| Identifiers | |||
| 10102-43-9 | |||
| 3D model (Jmol) | Interactive image | ||
| 3DMet | B00122 | ||
| ChEBI | CHEBI:16480 | ||
| ChEMBL | ChEMBL1200689 | ||
| ChemSpider | 127983 | ||
| DrugBank | DB00435 | ||
| ECHA InfoCard | 100.030.233 | ||
| EC Number | 233-271-0 | ||
| 451 | |||
| 2509 | |||
| KEGG | D00074 | ||
| PubChem | 145068 | ||
| RTECS number | QX0525000 | ||
| UNII | 31C4KY9ESH | ||
| UN number | 1660 | ||
| |||
| |||
| Properties | |||
| NO | |||
| Molar mass | 30.01 g·mol−1 | ||
| Appearance | Colourless gas | ||
| Density | 1.3402 g dm−3 | ||
| Melting point | −164 °C (−263 °F; 109 K) | ||
| Boiling point | −152 °C (−242 °F; 121 K) | ||
| 0.0098 g/100ml (0 °C) 0.0056 g/100ml (20 °C) | |||
| Refractive index (nD) |
1.0002697 | ||
| Structure | |||
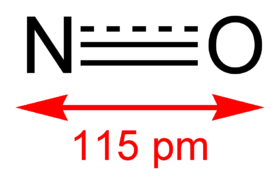
| linear (point group C∞v) | |||
| Thermochemistry | |||
| Std molar entropy (S |
210.76 J K−1 mol−1 | ||
| Std enthalpy of formation (ΔfH |
91.29 kJ mol−1 | ||
| Pharmacology | |||
| R07AX01 (WHO) | |||
| Inhalation | |||
| Pharmacokinetics: | |||
| good | |||
| via pulmonary capillary bed | |||
| 2–6 seconds | |||
| Hazards | |||
| Safety data sheet | External MSDS | ||
| EU classification (DSD) |
| ||
| R-phrases | R8, R23, R34, R44 | ||
| S-phrases | (S1), S17, S23, S36/37/39, S45 | ||
| NFPA 704 | |||
| Lethal dose or concentration (LD, LC): | |||
| LC50 (median concentration) |
315 ppm (rabbit, 15 min) 854 ppm (rat, 4 hr) 320 ppm (mouse)[2] | ||
| LCLo (lowest published) |
2500 ppm (mouse, 12 min)[2] | ||
| Related compounds | |||
| Dinitrogen pentoxide Dinitrogen tetroxide | |||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Nitric oxide (nitrogen oxide,[3] nitrogen monoxide) is a molecular, chemical compound with chemical formula of ·NO. One of several oxides of nitrogen, it is a colorless gas under standard conditions. Nitric oxide is a free radical—i.e., its bonding structure includes an unpaired electron,[4] represented by the dot (·) on the nitrogen atom—and it is in the class of heteronuclear diatomic molecules that are of historic theoretical interest (for the insights they gave in formulating early modern theories of bonding). It is a particularly important intermediate in the chemical industry. In addition, some is unavoidably produced during combustion of fossil fuels in power plants and automobile engines, with excess being created when more air or higher temperatures are present than needed for efficient and complete combustion of the fuel. It is also produced naturally by the extremely high air temperatures produced along the path of lightning in thunderstorms.
In mammals including humans, nitric oxide is an important cellular signaling molecule involved in many physiological and pathological processes.[5] It is a powerful vasodilator with a short half-life of a few seconds in the blood. Long-known pharmaceuticals such as nitroglycerine and amyl nitrite were found to be precursors to nitric oxide more than a century after their first use in medicine. Low levels of nitric oxide production are important in protecting organs such as the liver from ischemic damage. Nitric oxide production is associated with nonalcoholic fatty liver disease (NAFLD) and is essential for hepatic lipid metabolism under starvation.[6]
As a consequence of its importance in neuroscience, physiology, and immunology, nitric oxide was proclaimed "Molecule of the Year" in 1992.[7] Research into its function led to the 1998 Nobel Prize for discovering the role of nitric oxide as a cardiovascular signalling molecule. Nitric oxide should not be confused with nitrous oxide (N2O), an anesthetic, or with nitrogen dioxide (NO2), a brown toxic gas and a major air pollutant, the latter being a product to which nitric oxide is rapidly oxidized in air.
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.[8]
Reactions
- When exposed to oxygen, ntric oxide is converted into nitrogen dioxide.
- 2 ·NO + O2 → 2 NO2
- This conversion has been speculated as occurring via the ONOONO intermediate. In water, nitric oxide reacts with oxygen and water to form HNO2 or nitrous acid. The reaction is thought to proceed via the following stoichiometry:
- 4 ·NO + O2 + 2 H2O → 4 HNO2
- Nitric oxide will react with fluorine, chlorine, and bromine to form the XNO species, known as the nitrosyl halides, such as nitrosyl chloride. Nitrosyl iodide can form but is an extremely short-lived species and tends to reform I2.
- 2 ·NO + Cl2 → 2 NOCl
- Nitroxyl (HNO) is the reduced form of nitric oxide.
- Nitric oxide dimer N2O2 is formed when nitric oxide is cooled.
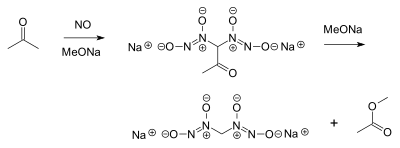
- Nitric oxide reacts with acetone and an alkoxide to a diazeniumdiolate or nitrosohydroxylamine and methyl acetate:[9]
- This reaction was discovered around 1898, and remains of interest in nitric oxide prodrug research. Nitric oxide can also react directly with sodium methoxide, forming sodium formate and nitrous oxide.[10]
Preparation
In commercial settings, nitric oxide is produced by the oxidation of ammonia at 750–900 °C (normally at 850 °C) with platinum as catalyst:
- 4 NH3 + 5 O2 → 4 ·NO + 6 H2O
The uncatalyzed endothermic reaction of oxygen (O2) and nitrogen (N2), which is performed at high temperature (>2000 °C) by lightning has not been developed into a practical commercial synthesis (see Birkeland–Eyde process):
- N2 + O2 → 2 ·NO
In the laboratory, nitric oxide is conveniently generated by reduction of dilute nitric acid with copper:
- 8 HNO3 + 3 Cu → 3 Cu(NO3)2 + 4 H2O + 2 ·NO
or by the reduction of nitrous acid in the form of sodium nitrite or potassium nitrite:
- 2 NaNO2 + 2 NaI + 2 H2SO4 → I2 + 4 NaHSO4 + 2 ·NO
- 2 NaNO2 + 2 FeSO4 + 3 H2SO4 → Fe2(SO4)3 + 2 NaHSO4 + 2 H2O + 2 ·NO
- 3 KNO2(l) + KNO3(l) + Cr2O3(s) → 2 K2CrO4(s) + 4 NO(g)
The iron(II) sulfate route is simple and has been used in undergraduate laboratory experiments. So-called NONOate compounds are also used for nitric oxide generation.
Coordination chemistry
Nitric oxide reacts with all transition metals to give complexes called metal nitrosyls. The most common bonding mode of nitric oxide is the terminal linear type (M−NO). The angle of the M−N−O group varies from 160° to 180° but is still termed "linear". In this case, the nitric oxide group is considered a 3-electron donor under the covalent (neutral) method of electron counting, or a 2-electron donor under the ionic method.[11] One can view such complexes as derived from NO+, which is isoelectronic with carbon monoxide (CO).
In the case of a bent M−N−O conformation, the ·NO group can be considered a one-electron donor using neutral counting, or a 2-electron donor using ionic counting.[12]
Nitric oxide can serve as a one-electron pseudohalide. In such complexes, the M−N−O group is characterized by an angle between 120° and 140°.
The ·NO group can also bridge between metal centers through the nitrogen atom in a variety of geometries.
Concentration measurement
_(white)_in_the_cytoplasm_(green)_of_clusters_of_conifer_cells_one_hour_after_mechanical_agitation.jpg)
Nitric oxide concentration can be determined using a simple chemiluminescent reaction involving ozone.[13] A sample containing nitric oxide is mixed with a large quantity of ozone. The nitric oxide reacts with the ozone to produce oxygen and nitrogen dioxide, accompanied with emission of light (chemiluminescence):
- ·NO + O3 → NO2 + O2 + hν
which can be measured with a photodetector. The amount of light produced is proportional to the amount of nitric oxide in the sample.
Other methods of testing include electroanalysis (amperometric approach), where ·NO reacts with an electrode to induce a current or voltage change. The detection of NO radicals in biological tissues is particularly difficult due to the short lifetime and concentration of these radicals in tissues. One of the few practical methods is spin trapping of nitric oxide with iron-dithiocarbamate complexes and subsequent detection of the mono-nitrosyl-iron complex with electron paramagnetic resonance (EPR).[14][15]
A group of fluorescent dye indicators that are also available in acetylated form for intracellular measurements exist. The most common compound is 4,5-diaminofluorescein (DAF-2).[16]
Production
From a thermodynamic perspective, ·NO is unstable with respect to O2 and N2, although this conversion is very slow at ambient temperatures in the absence of a catalyst. Because the heat of formation of ·NO is endothermic, its synthesis from molecular nitrogen and oxygen requires elevated temperatures above 1000 °C.
A major natural source is lightning. The use of internal combustion engines has drastically increased the presence of nitric oxide in the environment. One purpose of catalytic converters in cars is to minimize ·NO emission by catalytic reversion to O2 and N2.
Environmental effects
Acid deposition
Nitric oxide reacts with the hydroperoxy radical (HO2•) to form nitrogen dioxide (NO2), which then can react with a hydroxyl radical (•OH) to produce nitric acid (HNO3):
- ·NO + HO2•→ •NO2 + •OH
- ·NO2 + •OH → HNO3
Nitric acid, along with sulfuric acid, contribute acid rain deposition.
Ozone depletion
Furthermore, ·NO participates in ozone layer depletion. In this process, nitric oxide reacts with stratospheric ozone to form O2 and nitrogen dioxide:
- ·NO + O3 → NO2 + O2
As seen in the Concentration Measurement section, this reaction is also utilized to measure concentrations of ·NO in control volumes.
Precursor to NO2
As seen in the Acid deposition section, nitric oxide can transform into nitrogen dioxide (this can happen with the hydroperoxy radical, HO2•, or diatomic oxygen, O2). Symptoms of short-term nitrogen dioxide exposure include nausea, dyspnea and headache. Long-term effects could include impaired immune and respiratory function.[17]
Technical applications
Although ·NO has relatively few direct uses, it is produced on a massive scale as an intermediate in the Ostwald process for the synthesis of nitric acid from ammonia. In 2005, the US alone produced 6 million metric tons of nitric acid.[18] It finds use in the semiconductor industry for various processes. In one of its applications, it is used along with nitrous oxide to form oxynitride gates in CMOS devices.
Miscellaneous applications
Nitric oxide can be used for detecting surface radicals on polymers. Quenching of surface radicals with nitric oxide results in incorporation of nitrogen, which can be quantified by means of X-ray photoelectron spectroscopy.
Biological functions
·NO is one of the few gaseous signalling molecules known and is additionally exceptional due to the fact that it is a radical gas. It is a key vertebrate biological messenger, playing a role in a variety of biological processes.[19] It is a known bioproduct in almost all types of organisms, ranging from bacteria to plants, fungi, and animal cells.[20]
Nitric oxide, known as an endothelium-derived relaxing factor (EDRF), is biosynthesized endogenously from L-arginine, oxygen, and NADPH by various nitric oxide synthase (NOS) enzymes. Reduction of inorganic nitrate may also serve to make nitric oxide. The endothelium (inner lining) of blood vessels uses nitric oxide to signal the surrounding smooth muscle to relax, thus resulting in vasodilation and increasing blood flow. Nitric oxide is highly reactive (having a lifetime of a few seconds), yet diffuses freely across membranes. These attributes make nitric oxide ideal for a transient paracrine (between adjacent cells) and autocrine (within a single cell) signaling molecule.[21]
Independent of nitric oxide synthase, an alternative pathway, coined the nitrate-nitrite-nitric oxide pathway, elevates nitric oxide through the sequential reduction of dietary nitrate derived from plant-based foods.[22] Nitrate-rich vegetables, in particular leafy greens, such as spinach and arugula, and beetroot, have been shown to increase cardioprotective levels of nitric oxide with a corresponding reduction in blood pressure in pre-hypertensive persons.[23][24] For the body to generate nitric oxide through the nitrate-nitrite-nitric oxide pathway, the reduction of nitrate to nitrite (by nitrate reductase, a bacterial enzyme) occurs in the mouth, by commensal bacteria, an obligatory and necessary step.[25] Monitoring nitric oxide status by saliva testing detects the bioconversion of plant-derived nitrate into nitric oxide. A rise in salivary levels is indicative of diets rich in leafy vegetables which are often abundant in anti-hypertensive diets such as the DASH diet.[26]
The production of nitric oxide is elevated in populations living at high altitudes, which helps these people avoid hypoxia by aiding in pulmonary vasculature vasodilation. Effects include vasodilatation, neurotransmission (see gasotransmitters), modulation of the hair cycle,[27] production of reactive nitrogen intermediates and penile erections (through its ability to vasodilate). Nitroglycerin and amyl nitrite serve as vasodilators because they are converted to nitric oxide in the body. The vasodilating antihypertensive drug minoxidil contains an ·NO moiety and may act as an NO agonist. Likewise, Sildenafil citrate, popularly known by the trade name Viagra, stimulates erections primarily by enhancing signaling through the nitric oxide pathway in the penis.
Nitric oxide (NO) contributes to vessel homeostasis by inhibiting vascular smooth muscle contraction and growth, platelet aggregation, and leukocyte adhesion to the endothelium. Humans with atherosclerosis, diabetes, or hypertension often show impaired NO pathways.[28] A high salt intake was demonstrated to attenuate NO production in patients with essential hypertension, although bioavailability remains unregulated.[29]
Nitric oxide is also generated by phagocytes (monocytes, macrophages, and neutrophils) as part of the human immune response.[30] Phagocytes are armed with inducible nitric oxide synthase (iNOS), which is activated by interferon-gamma (IFN-γ) as a single signal or by tumor necrosis factor (TNF) along with a second signal.[31][32][33] On the other hand, transforming growth factor-beta (TGF-β) provides a strong inhibitory signal to iNOS, whereas interleukin-4 (IL-4) and IL-10 provide weak inhibitory signals. In this way, the immune system may regulate the armamentarium of phagocytes that play a role in inflammation and immune responses.[34] Nitric oxide is secreted as free radicals in an immune response and is toxic to bacteria and intracellular parasites, including Leishmania[35] and malaria;[36][37][38] the mechanism for this includes DNA damage[39][40][41] and degradation of iron sulfur centers into iron ions and iron-nitrosyl compounds.[42]
The inducible pathway (iNOS) of nitrogen oxide synthesis in phagocytes can generated large amounts of ·NO that trigger apoptosis in and thereby the death of cells. In vitro studies indicate that phagocyte-dependent generation of ·NO at concentrations greater than 400-500 NM triggers apoptosis in nearby cells and that this effect may act in a manner similar to Specialized pro-resolving mediators to dampen and reverse inflammatory responses by neutralizing and then speeding the clearance of pro-inflammatory cells from inflamed tissues.[43] However, the role of ·NO in inflammation is complex with model studies involving viral infection suggesting that this gaseous mediator can also promote inflammation.[44]
In response, many bacterial pathogens have evolved mechanisms for nitric oxide resistance.[45] Because nitric oxide might serve as an inflammometer (meter of inflammation) in conditions like asthma, there has been increasing interest in the use of exhaled nitric oxide as a breath test in diseases with airway inflammation. Reduced levels of exhaled NO have been associated with exposure to air pollution in cyclists and smokers, but, in general, increased levels of exhaled NO are associated with exposure to air pollution.[46]
Nitric oxide can contribute to reperfusion injury when an excessive amount produced during reperfusion (following a period of ischemia) reacts with superoxide to produce the damaging oxidant peroxynitrite. In contrast, inhaled nitric oxide has been shown to help survival and recovery from paraquat poisoning, which produces lung tissue-damaging superoxide and hinders NOS metabolism.
In plants, nitric oxide can be produced by any of four routes: (i) L-arginine-dependent nitric oxide synthase,[47][48][49] (although the existence of animal NOS homologs in plants is debated),[50] (ii) plasma membrane-bound nitrate reductase, (iii) mitochondrial electron transport chain, or (iv) non-enzymatic reactions. It is a signaling molecule, acts mainly against oxidative stress and also plays a role in plant pathogen interactions. Treating cut flowers and other plants with nitric oxide has been shown to lengthen the time before wilting.[51]
Two important biological reaction mechanisms of nitric oxide are S-nitrosation of thiols, and nitrosylation of transition metal ions. S-nitrosation involves the (reversible) conversion of thiol groups, including cysteine residues in proteins, to form S-nitrosothiols (RSNOs). S-Nitrosation is a mechanism for dynamic, post-translational regulation of most or all major classes of protein.[52] The second mechanism, nitrosylation, involves the binding of ·NO to a transition metal ion like iron or copper. In this function, ·NO is referred to as a nitrosyl ligand. Typical cases involve the nitrosylation of heme proteins like cytochromes, thereby disabling the normal enzymatic activity of the enzyme. Nitrosylated ferrous iron is particularly stable, as the binding of the nitrosyl ligand to ferrous iron (Fe(II)) is very strong. Hemoglobin is a prominent example of a heme protein that may be modified by ·NO by both pathways: ·NO may attach directly to the heme in the nitrosylation reaction, and independently form S-nitrosothiols by S-nitrosation of the thiol moieties.[53]
Mechanism of action
There are several mechanisms by which ·NO has been demonstrated to affect the biology of living cells. These include oxidation of iron-containing proteins such as ribonucleotide reductase and aconitase, activation of the soluble guanylate cyclase, ADP ribosylation of proteins, protein sulfhydryl group nitrosylation, and iron regulatory factor activation.[54] ·NO has been demonstrated to activate NF-κB in peripheral blood mononuclear cells, an important transcription factor in iNOS gene expression in response to inflammation.[55]
It was found that ·NO acts through the stimulation of the soluble guanylate cyclase, which is a heterodimeric enzyme with subsequent formation of cyclic-GMP. Cyclic-GMP activates protein kinase G, which causes reuptake of Ca2+ and the opening of calcium-activated potassium channels. The fall in concentration of Ca2+ ensures that the myosin light-chain kinase (MLCK) can no longer phosphorylate the myosin molecule, thereby stopping the crossbridge cycle and leading to relaxation of the smooth muscle cell.[56]
Medical use
Neonatal use
Nitric oxide/oxygen blends are used in critical care to promote capillary and pulmonary dilation to treat primary pulmonary hypertension in neonatal patients[57][58] post-meconium aspiration and related to birth defects. These are often a last-resort gas mixture before the use of extracorporeal membrane oxygenation (ECMO). Nitric oxide therapy has the potential to significantly increase the quality of life and, in some cases, save the lives of infants at risk for pulmonary vascular disease.[59]
Pediatric and adult use
Currently in the United States, nitric oxide use is not approved for any population other than neonates. In the adult ICU setting, inhaled ·NO can improve hypoxemia in acute lung injury, acute respiratory distress syndrome, and severe pulmonary hypertension, although the effects are short-lived and there are no studies demonstrating improved clinical outcomes. It is used on an individualized basis in ICUs as an adjunct to other definitive therapies for reversible causes of hypoxemic respiratory distress.[60]
Dosage and strength
Currently in the United States, nitric oxide is a gas available in concentrations of only 100 ppm and 800 ppm. Overdosage with inhaled nitric oxide will be seen by elevations in methemoglobin and pulmonary toxicities associated with inspired ·NO. Elevated NO may cause acute lung injury.
Contraindications
Inhaled nitric oxide is contraindicated in the treatment of neonates known to be dependent on right-to-left shunting of blood. This is as the nitric oxide decreases the pulmonary circulation's resistance by dilating pulmonary blood vessels. The increased pulmonary return increases pressure in the left atrium, causing closure of the foramen ovale and reducing the blood flow through the ductus arteriosus. Closing these shunts can kill neonates with heart malformations that rely on the right-to-left shunting of blood.
Pulmonary embolism
Nitric oxide is also administered as salvage therapy in patients with acute right ventricular failure secondary to pulmonary embolism.[61]
Pharmacology
Nitric oxide is considered an antianginal drug: It causes vasodilation, which can help with ischemic pain, known as angina, by decreasing the cardiac workload. By dilating (expanding) the arteries, nitric oxide drugs lower arterial pressure and left ventricular filling pressure.[62]
This vasodilation does not decrease the volume of blood the heart pumps, but rather it decreases the force the heart muscle must exert to pump the same volume of blood. Nitroglycerin pills, taken sublingually (under the tongue), are used to prevent or treat acute chest pain. The nitroglycerin reacts with a sulfhydryl group (–SH) to produce nitric oxide, which eases the pain by causing vasodilation. There is a potential role for the use of nitric oxide in alleviating bladder contractile dysfunctions,[63][64] and recent evidence suggests that nitrates may be beneficial for treatment of angina due to reduced myocardial oxygen consumption both by decreasing preload and afterload and by some direct vasodilation of coronary vessels.[62]
Associated problems
There are some associated complaints with utilization of nitric oxide in neonatal patients. Some of them include dose errors associated with the delivery system, headaches associated with environmental exposure of nitric oxide in hospital staff, hypotension associated with acute withdrawal of the drug, hypoxemia associated with acute withdrawal of the drug, and pulmonary edema in patients with CREST syndrome.
Mechanism of action
Nitric oxide is a compound produced by many cells of the body. It relaxes vascular smooth muscle by binding to the heme moiety of cytosolic guanylate cyclase, activating guanylate cyclase and increasing intracellular levels of cyclic-guanosine 3’,5’-monophosphate, which then leads to vasodilation. When inhaled, nitric oxide dilates the pulmonary vasculature and, because of efficient scavenging by hemoglobin, has minimal effect on the vasculature of the entire body.[65]
Inhaled nitric oxide appears to increase the partial pressure of arterial oxygen (PaO2) by dilating pulmonary vessels in better-ventilated areas of the lung, moving pulmonary blood flow away from lung segments with low ventilation/perfusion (V/Q) ratios toward segments with normal or better ratios.[66]
Pharmacokinetics
Nitric oxide is absorbed systemically after inhalation. Most of it moves across the pulmonary capillary bed where it combines with hemoglobin that is 60% to 100% oxygen-saturated.
Nitrate has been identified as the predominant nitric oxide metabolite excreted in the urine, accounting for >70% of the nitric oxide dose inhaled. Nitrate is cleared from the plasma by the kidney at rates approaching the rate of glomerular filtration.
Occupational safety and health
People can be exposed to nitric oxide in the workplace by breathing it in. The Occupational Safety and Health Administration (OSHA) has set the legal limit (permissible exposure limit) for nitric oxide exposure in the workplace as 25 ppm (30 mg/m3) over an 8-hour workday. The National Institute for Occupational Safety and Health (NIOSH) has set a recommended exposure limit (REL) of 25 ppm (30 mg/m3) over an 8-hour workday. At levels of 100 ppm, nitric oxide is immediately dangerous to life and health.[67]
References
- ↑ "Nitric Oxide (CHEBI:16480)". Chemical Entities of Biological Interest (ChEBI). UK: European Bioinformatics Institute.
- 1 2 "Nitric oxide". Immediately Dangerous to Life and Health. National Institute for Occupational Safety and Health (NIOSH).
- ↑ IUPAC nomenclature of inorganic chemistry 2005. PDF.
- ↑ Lund, Anders; Shimada, Shigetaka; Shiotani, Masaru (2011). Principles and Applications of ESR Spectroscopy. Springer. ISBN 978-1-4020-5344-3.
- ↑ Hou, YC; Janczuk, A; Wang, PG (1999). "Current trends in the development of nitric oxide donors". Current pharmaceutical design. 5 (6): 417–41. PMID 10390607.
- ↑ Gu, Qilin; Yang, Xiaojie; Lin, Li; Li, Shaoyang; Li, Qing; Zhong, Shan; Peng, Jinrong; Cui, Zongbin (December 2014). "Genetic ablation of solute carrier family 7a3a leads to hepatic steatosis in zebrafish during fasting". Hepatology. 60 (6): 1929–1941. doi:10.1002/hep.27356.
- ↑ Culotta, Elizabeth; Koshland, Daniel E. Jr (1992). "NO news is good news". Science. 258 (5090): 1862–1864. doi:10.1126/science.1361684. PMID 1361684.
- ↑ "40 C.F.R.: Appendix A to Part 355—The List of Extremely Hazardous Substances and Their Threshold Planning Quantities" (PDF) (July 1, 2008 ed.). Government Printing Office. Retrieved October 29, 2011.
- ↑ Traube, Wilhelm (1898). "Ueber Synthesen stickstoffhaltiger Verbindungen mit Hülfe des Stickoxyds". Justus Liebig's Annalen der Chemie. 300: 81–128. doi:10.1002/jlac.18983000108.
- ↑ Derosa, Frank; Keefer, Larry K.; Hrabie, Joseph A. (2008). "Nitric Oxide Reacts with Methoxide". The Journal of Organic Chemistry. 73 (3): 1139–42. doi:10.1021/jo7020423. PMID 18184006.
- ↑ Crabtree, Robert H. (2005). The Organometallic Chemistry of the Transition Metals. John Wiley and Sons. p. 32. ISBN 9780471718758.
- ↑ Crabtree, Robert H. (2005). The Organometallic Chemistry of the Transition Metals. John Wiley and Sons. pp. 96–98. ISBN 9780471718758.
- ↑ Fontijn, Arthur.; Sabadell, Alberto J.; Ronco, Richard J. (1970). "Homogeneous chemiluminescent measurement of nitric oxide with ozone. Implications for continuous selective monitoring of gaseous air pollutants". Analytical Chemistry. 42 (6): 575–579. doi:10.1021/ac60288a034.
- ↑ Vanin, A; Huisman, A; Van Faassen, E (2002). "Iron dithiocarbamate as spin trap for nitric oxide detection: Pitfalls and successes". Methods in enzymology. Methods in Enzymology. 359: 27–42. doi:10.1016/S0076-6879(02)59169-2. ISBN 9780121822620. PMID 12481557.
- ↑ Nagano, T; Yoshimura, T (2002). "Bioimaging of nitric oxide". Chemical Reviews. 102 (4): 1235–70. doi:10.1021/cr010152s. PMID 11942795.
- ↑ Kojima H, Nakatsubo N, Kikuchi K, Kawahara S, Kirino Y, Nagoshi H, Hirata Y, Nagano T (1998). "Detection and imaging of nitric oxide with novel fluorescent indicators: diaminofluoresceins". Anal. Chem. 70 (13): 2446–2453. doi:10.1021/ac9801723. PMID 9666719.
- ↑ "Centers for Disease Control and Prevention". NIOSH. 1 July 2014. Retrieved 10 December 2015.
- ↑ "Production: Growth is the Norm". Chemical and Engineering News. 84 (28): 59–68. July 10, 2006. doi:10.1021/cen-v084n028.p059.
- ↑ Weller, Richard, Could the sun be good for your heart? TedxGlasgow. Filmed March 2012, posted January 2013
- ↑ Roszer, T (2012) The Biology of Subcellular Nitric Oxide. ISBN 978-94-007-2818-9
- ↑ Stryer, Lubert (1995). Biochemistry, 4th Edition. W.H. Freeman and Company. p. 732. ISBN 0-7167-2009-4.
- ↑ "Plant-based Diets | Plant-based Foods | Beetroot Juice | Nitric Oxide Vegetables". Berkeley Test. Retrieved 2013-10-04.
- ↑ Ghosh, S. M.; Kapil, V.; Fuentes-Calvo, I.; Bubb, K. J.; Pearl, V.; Milsom, A. B.; Khambata, R.; Maleki-Toyserkani, S.; Yousuf, M.; Benjamin, N.; Webb, A. J.; Caulfield, M. J.; Hobbs, A. J.; Ahluwalia, A. (2013). "Enhanced Vasodilator Activity of Nitrite in Hypertension: Critical Role for Erythrocytic Xanthine Oxidoreductase and Translational Potential". Hypertension. 61 (5): 1091–102. doi:10.1161/HYPERTENSIONAHA.111.00933. PMID 23589565.
- ↑ Webb, A. J.; Patel, N.; Loukogeorgakis, S.; Okorie, M.; Aboud, Z.; Misra, S.; Rashid, R.; Miall, P.; Deanfield, J.; Benjamin, N.; MacAllister, R.; Hobbs, A. J.; Ahluwalia, A. (2008). "Acute Blood Pressure Lowering, Vasoprotective, and Antiplatelet Properties of Dietary Nitrate via Bioconversion to Nitrite". Hypertension. 51 (3): 784–90. doi:10.1161/HYPERTENSIONAHA.107.103523. PMC 2839282
 . PMID 18250365.
. PMID 18250365. - ↑ Hezel, MP; Weitzberg, E (2013). "The oral microbiome and nitric oxide homoeostasis". Oral Diseases. 21: n/a. doi:10.1111/odi.12157.
- ↑ Green, Shawn J. (2013-07-25). "Turning DASH Strategy into Reality for Improved Cardio Wellness Outcomes: Part II". Real World Health Care. Retrieved 2013-10-04.
- ↑ Proctor, PH (August 1989). "Endothelium-Derived Relaxing Factor and Minoxidil: Active Mechanisms in Hair Growth". Archives of Dermatology. 125 (8): 1146. doi:10.1001/archderm.1989.01670200122026. PMID 2757417.
- ↑ Dessy, C.; Ferron, O. (2004). "Pathophysiological Roles of Nitric Oxide: In the Heart and the Coronary Vasculature". Current Medical Chemistry – Anti-Inflammatory & Anti-Allergy Agents in Medicinal Chemistry. 3 (3): 207–216. doi:10.2174/1568014043355348.
- ↑ Osanai, T; Fujiwara, N; Saitoh, M; Sasaki, S; Tomita, H; Nakamura, M; Osawa, H; Yamabe, H; Okumura, K (2002). "Relationship between salt intake, nitric oxide, and asymmetric dimethylarginine and its relevance to patients with end-stage renal disease". Blood purification. 20 (5): 466–8. doi:10.1159/000063555. PMID 12207094.
- ↑ Green, SJ; Mellouk, S; Hoffman, SL; Meltzer, MS; Nacy, CA (1990). "Cellular mechanisms of nonspecific immunity to intracellular infection: Cytokine-induced synthesis of toxic nitrogen oxides from L-arginine by macrophages and hepatocytes". Immunology letters. 25 (1–3): 15–9. doi:10.1016/0165-2478(90)90083-3. PMID 2126524.
- ↑ Gorczyniski and Stanely, Clinical Immunology. Landes Bioscience; Austin, TX. ISBN 1-57059-625-5
- ↑ Green, SJ; Nacy, CA; Schreiber, RD; Granger, DL; Crawford, RM; Meltzer, MS; Fortier, AH (1993). "Neutralization of gamma interferon and tumor necrosis factor alpha blocks in vivo synthesis of nitrogen oxides from L-arginine and protection against Francisella tularensis infection in Mycobacterium bovis BCG-treated mice". Infection and immunity. 61 (2): 689–98. PMC 302781
 . PMID 8423095.
. PMID 8423095. - ↑ Kamijo, R; Gerecitano, J; Shapiro, D; Green, SJ; Aguet, M; Le, J; Vilcek, J (1995). "Generation of nitric oxide and clearance of interferon-gamma after BCG infection are impaired in mice that lack the interferon-gamma receptor". Journal of inflammation. 46 (1): 23–31. PMID 8832969.
- ↑ Green, SJ; Scheller, LF; Marletta, MA; Seguin, MC; Klotz, FW; Slayter, M; Nelson, BJ; Nacy, CA (1994). "Nitric oxide: Cytokine-regulation of nitric oxide in host resistance to intracellular pathogens". Immunology letters. 43 (1–2): 87–94. doi:10.1016/0165-2478(94)00158-8. PMID 7537721.
- ↑ Green, SJ; Crawford, RM; Hockmeyer, JT; Meltzer, MS; Nacy, CA (1990). "Leishmania major amastigotes initiate the L-arginine-dependent killing mechanism in IFN-gamma-stimulated macrophages by induction of tumor necrosis factor-alpha". Journal of immunology. 145 (12): 4290–7. PMID 2124240.
- ↑ Seguin, M. C.; Klotz, FW; Schneider, I; Weir, JP; Goodbary, M; Slayter, M; Raney, JJ; Aniagolu, JU; Green, SJ (1994). "Induction of nitric oxide synthase protects against malaria in mice exposed to irradiated Plasmodium berghei infected mosquitoes: Involvement of interferon gamma and CD8+ T cells". Journal of Experimental Medicine. 180 (1): 353–8. doi:10.1084/jem.180.1.353. PMC 2191552
 . PMID 7516412.
. PMID 7516412. - ↑ Mellouk, S; Green, SJ; Nacy, CA; Hoffman, SL (1991). "IFN-gamma inhibits development of Plasmodium berghei exoerythrocytic stages in hepatocytes by an L-arginine-dependent effector mechanism". Journal of immunology. 146 (11): 3971–6. PMID 1903415.
- ↑ Klotz, FW; Scheller, LF; Seguin, MC; Kumar, N; Marletta, MA; Green, SJ; Azad, AF (1995). "Co-localization of inducible-nitric oxide synthase and Plasmodium berghei in hepatocytes from rats immunized with irradiated sporozoites". Journal of immunology. 154 (7): 3391–5. PMID 7534796.
- ↑ Wink, D.; Kasprzak, K.; Maragos, C.; Elespuru, R.; Misra, M; Dunams, T.; Cebula, T.; Koch, W.; Andrews, A.; Allen, J.; Et, al. (1991). "DNA deaminating ability and genotoxicity of nitric oxide and its progenitors". Science. 254 (5034): 1001–3. doi:10.1126/science.1948068. PMID 1948068.
- ↑ Nguyen, T.; Brunson, D.; Crespi, C. L.; Penman, B. W.; Wishnok, J. S.; Tannenbaum, S. R. (1992). "DNA Damage and Mutation in Human Cells Exposed to Nitric Oxide in vitro". Proceedings of the National Academy of Sciences. 89 (7): 3030–3034. doi:10.1073/pnas.89.7.3030. Free text.
- ↑ Li, Chun-Qi; Pang, Bo; Kiziltepe, Tanyel; Trudel, Laura J.; Engelward, Bevin P.; Dedon, Peter C.; Wogan, Gerald N. (2006). "Threshold Effects of Nitric Oxide-Induced Toxicity and Cellular Responses in Wild-Type and p53-Null Human Lymphoblastoid Cells". Chemical Research in Toxicology. 19 (3): 399–406. doi:10.1021/tx050283e. PMC 2570754
 . PMID 16544944. free text
. PMID 16544944. free text - ↑ Hibbs, John B.; Taintor, Read R.; Vavrin, Zdenek; Rachlin, Elliot M. (1988). "Nitric oxide: A cytotoxic activated macrophage effector molecule". Biochemical and Biophysical Research Communications. 157 (1): 87–94. doi:10.1016/S0006-291X(88)80015-9. PMID 3196352.
- ↑ Wallace JL, Ianaro A, Flannigan KL, Cirino G (2015). "Gaseous mediators in resolution of inflammation". Seminars in Immunology. 27 (3): 227–33. doi:10.1016/j.smim.2015.05.004. PMID 26095908.
- ↑ Uehara EU, Shida Bde S, de Brito CA (2015). "Role of nitric oxide in immune responses against viruses: beyond microbicidal activity". Inflammation Research : Official Journal of the European Histamine Research Society ... [Et Al.] 64 (11): 845–52. doi:10.1007/s00011-015-0857-2. PMID 26208702.
- ↑ Janeway, C. A.; et al. (2005). Immunobiology: the immune system in health and disease (6th ed.). New York: Garland Science. ISBN 0-8153-4101-6.
- ↑ Jacobs, Lotte; Nawrot, Tim S; De Geus, Bas; Meeusen, Romain; Degraeuwe, Bart; Bernard, Alfred; Sughis, Muhammad; Nemery, Benoit; Panis, Luc (2010). "Subclinical responses in healthy cyclists briefly exposed to traffic-related air pollution: An intervention study". Environmental Health. 9: 64. doi:10.1186/1476-069X-9-64. PMC 2984475
 . PMID 20973949.
. PMID 20973949. - ↑ Corpas, F. J.; Barroso, JB; Carreras, A; Quirós, M; León, AM; Romero-Puertas, MC; Esteban, FJ; Valderrama, R; Palma, JM; Sandalio, LM; Gómez, M; Del Río, LA (2004). "Cellular and subcellular localization of endogenous nitric oxide in young and senescent pea plants". Plant Physiology. 136 (1): 2722–33. doi:10.1104/pp.104.042812. PMC 523336
 . PMID 15347796.
. PMID 15347796. - ↑ Corpas, F. J.; Barroso, Juan B.; Carreras, Alfonso; Valderrama, Raquel; Palma, José M.; León, Ana M.; Sandalio, Luisa M.; Del Río, Luis A (2006). "Constitutive arginine-dependent nitric oxide synthase activity in different organs of pea seedlings during plant development". Planta. 224 (2): 246–54. doi:10.1007/s00425-005-0205-9. PMID 16397797.
- ↑ Valderrama, R.; Corpas, Francisco J.; Carreras, Alfonso; Fernández-Ocaña, Ana; Chaki, Mounira; Luque, Francisco; Gómez-Rodríguez, María V.; Colmenero-Varea, Pilar; Del Río, Luis A.; Barroso, Juan B. (2007). "Nitrosative stress in plants". FEBS Lett. 581 (3): 453–61. doi:10.1016/j.febslet.2007.01.006. PMID 17240373.
- ↑ Corpas, F. J.; Barroso, Juan B.; Del Rio, Luis A. (2004). "Enzymatic sources of nitric oxide in plant cells – beyond one protein–one function". New Phytologist. 162 (2): 246–7. doi:10.1111/j.1469-8137.2004.01058.x.
- ↑ Siegel-Itzkovich, J. (1999). "Viagra makes flowers stand up straight". BMJ. 319 (7205): 274. doi:10.1136/bmj.319.7205.274a. PMC 1126920
 . PMID 10426722.
. PMID 10426722. - ↑ van Faassen, E. and Vanin, A. (eds.) (2007) Radicals for life: The various forms of nitric oxide. Elsevier, Amsterdam, ISBN 978-0-444-52236-8
- ↑ van Faassen, E. and Vanin, A. (2004) "Nitric Oxide", in Encyclopedia of Analytical Science, 2nd ed., Elsevier, ISBN 0127641009.
- ↑ Shami, PJ; Moore, JO; Gockerman, JP; Hathorn, JW; Misukonis, MA; Weinberg, JB (1995). "Nitric oxide modulation of the growth and differentiation of freshly isolated acute non-lymphocytic leukemia cells". Leukemia research. 19 (8): 527–33. doi:10.1016/0145-2126(95)00013-E. PMID 7658698.
- ↑ Kaibori M.; Sakitani K.; Oda M.; Kamiyama Y.; Masu Y.; Okumura T. (1999). "Immunosuppressant FK506 inhibits inducible nitric oxide synthase gene expression at a step of NF-κB activation in rat hepatocytes". J. Hepatol. 30 (6): 1138–1145. doi:10.1016/S0168-8278(99)80270-0. PMID 10406194.
- ↑ Rhoades, RA; Tanner, GA (2003). Medical physiology 2nd edition. PMID 174.
- ↑ Finer NN, Barrington KJ; Barrington (2006). Finer, Neil, ed. "Nitric oxide for respiratory failure in infants born at or near term". Cochrane Database Syst Rev (4): CD000399. doi:10.1002/14651858.CD000399.pub2. PMID 17054129.
- ↑ Chotigeat U, Khorana M, Kanjanapattanakul W (2007). "Inhaled nitric oxide in newborns with severe hypoxic respiratory failure". J Med Assoc Thai. 90 (2): 266–71. PMID 17375630.
- ↑ Hayward, CS; Kelly, RP; MacDonald, PS (1999). "Inhaled nitric oxide in cardiology practice". Cardiovascular research. 43 (3): 628–38. doi:10.1016/S0008-6363(99)00114-5. PMID 10690334.
- ↑ Mark J.D. Griffiths, M.R.C.P.; Timothy W. Evans, M.D. (December 22, 2005). "Inhaled Nitric Oxide Therapy in Adults". N Engl J Med. 353 (25): 2683–2695. doi:10.1056/NEJMra051884. PMID 16371634.
- ↑ Summerfield DT, Desai H, Levitov A, Grooms DA, Marik PE (2011). "Inhaled Nitric Oxide as Salvage Therapy in Massive Pulmonaryembolism: A Case Series". Respir Care. 57 (3): 444–8. doi:10.4187/respcare.01373. PMID 22005573.
- 1 2 Abrams, J (1996). "Beneficial actions of nitrates in cardiovascular disease". The American Journal of Cardiology. 77 (13): 31C–7C. doi:10.1016/S0002-9149(96)00186-5. PMID 8638524.
- ↑ Moro, C; Leeds, C; Chess-Williams, R (January 2012). "Contractile activity of the bladder urothelium/lamina propria and its regulation by nitric oxide". Eur J Pharmacol. 674 (2–3): 445–449. doi:10.1016/j.ejphar.2011.11.020. PMID 22119378.
- ↑ Andersson, M.C; Tobin, G; Giglio, D (February 2008). "Cholinergic nitric oxide release from the urinary bladder mucosa in cyclophosphamide-induced cystitis of the anaesthetized rat". Br J Pharmacol. 153 (7): 1438–44. doi:10.1038/bjp.2008.6. PMC 2437908
 .
. - ↑ Kinsella JP, Cutter GR, Walsh WF, Gerstmann DR, Bose CL, Hart C, et al. (2006). "Early inhaled nitric oxide therapy in premature newborns with respiratory failure". N Engl J Med. 355 (4): 354–64. doi:10.1056/NEJMoa060442. PMID 16870914.
- ↑ Ballard RA, Truog WE, Cnaan A, Martin RJ, Ballard PL, Merrill JD, et al. (2006). "Inhaled nitric oxide in preterm infants undergoing mechanical ventilation". N Engl J Med. 355 (4): 343–53. doi:10.1056/NEJMoa061088. PMID 16870913.
- ↑ "CDC - NIOSH Pocket Guide to Chemical Hazards - Nitric oxide". www.cdc.gov. Retrieved 2015-11-20.
Further reading
- Butler A. and Nicholson R.; "Life, death and NO." Cambridge 2003. ISBN 978-0-85404-686-7.
- van Faassen, E. E.; Vanin, A. F. (eds); "Radicals for life: The various forms of Nitric Oxide." Elsevier, Amsterdam 2007. ISBN 978-0-444-52236-8.
- Ignarro, L. J. (ed.); "Nitric oxide:biology and pathobiology." Academic Press, San Diego 2000. ISBN 0-12-370420-0.
External links
- International Chemical Safety Card 1311
- CDC – NIOSH Pocket Guide to Chemical Hazards
- 1998 Nobel Prize in Physiology/Medicine for discovery of NO's role in cardiovascular regulation
- "Nitric oxide and its role in health and diabetes." (PDF).
- Microscale Gas Chemistry: Experiments with Nitrogen Oxides
- Your Brain Boots Up Like a Computer – new insights about the biological role of nitric oxide.
- Assessing The Potential of Nitric Oxide in the Diabetic Foot
- New Discoveries About Nitric Oxide Can Provide Drugs For Schizophrenia
- Nitric Oxide at the Chemical Database