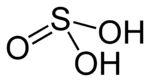
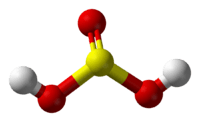
Sulfurous acid
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Sulfurous acid | |
| Identifiers | |
| 7782-99-2 | |
| 3D model (Jmol) | Interactive image |
| ChEBI | CHEBI:48854 |
| ChEMBL | ChEMBL1161699 |
| ChemSpider | 1069 |
| ECHA InfoCard | 100.029.066 |
| KEGG | C00094 |
| PubChem | 1100 |
| UNII | J1P7893F4J |
| |
| |
| Properties | |
| H2SO3 | |
| Molar mass | 82.07 g/mol |
| Acidity (pKa) | 1.857, 7.172 |
| Hazards | |
| Safety data sheet | ICSC 0074 |
| EU classification (DSD) |
|
| R-phrases | R20, R34 |
| S-phrases | (S1/2), S9, S26, S36/37/39, S45 |
| Flash point | Non-flammable |
| Related compounds | |
| Related compounds |
Sulfur dioxide Sulfuric acid |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Sulfurous acid (also sulphurous acid) is the chemical compound with the formula H2SO3. There is no evidence that sulfurous acid exists in solution, but the molecule has been detected in the gas phase.[1] The conjugate bases of this elusive acid are, however, common anions, bisulfite (or hydrogen sulfite) and sulfite. Sulfurous acid is an intermediate species in the formation of acid rain from sulfur dioxide.[2]
Raman spectra of solutions of sulfur dioxide in water show only signals due to the SO2 molecule and the bisulfite ion, HSO−
3.[3] The intensities of the signals are consistent with the following equilibrium:
- SO2 + H2O ⇌ HSO−
3 + H+ Ka = 1.54×10−2; pKa = 1.81.
- SO2 + H2O ⇌ HSO−
17O NMR spectroscopy provided evidence that solutions of sulfurous acid and protonated sulfites contains a mixture of isomers, which is in equilibrium:[4]
- [H–OSO2]− ⇌ [H–SO3]−
When trying to concentrate the solution by evaporation to produce waterless sulfurous acid it will decompose (reversing the forming reaction). In cooling down a clathrate SO2 · 5,75 H2O will cristallize which decomposes again at 7 °C. Thus sulfurous acid H2SO3 cannot be isolated.
Uses
Aqueous solutions of sulfur dioxide, which sometimes are referred to as sulfurous acid, are used as reducing agents and as disinfectants, as are solutions of bisulfite and sulfite salts. They are oxidised to sulfuric acid or sulfate by accepting another oxygen atom. [5]
They are also mild bleaches, and are used for materials which may be damaged by chlorine-containing bleaches.
References
- ↑ D. Sülzle; M. Verhoeven; J. K. Terlouw; H. Schwarz (1988). "Generation and Characterization of Sulfurous Acid (H2SO3) and of Its Radical Cation as Stable Species in the Gas Phase". Angew. Chem. Int. Ed. Engl. 27 (11): 1533–4. doi:10.1002/anie.198815331.
- ↑ McQuarrie; Rock (1987). General Chemistry (2nd ed.). New York: W.H. Freeman and Company. p. 243. ISBN 0-7167-1806-5.
- ↑ Jolly, William L. (1991), Modern Inorganic Chemistry (2nd ed.), New York: McGraw-Hill, ISBN 0-07-032768-8
- ↑ Catherine E. Housecroft; Alan G. Sharpe (2008). "Chapter 16: The group 16 elements". Inorganic Chemistry, 3rd Edition. Pearson. p. 520. ISBN 978-0-13-175553-6.
- ↑ L. Kolditz, Anorganische Chemie, VEB Deutscher Verlag der Wissenschaften, Berlin 1983, S. 476.