Cerebral palsy
| Cerebral palsy | |
|---|---|
|
A child with cerebral palsy | |
| Classification and external resources | |
| Specialty | Pediatrics |
| ICD-10 | G80 |
| ICD-9-CM | 343 |
| OMIM | 603513 605388 |
| DiseasesDB | 2232 |
| MedlinePlus | 000716 |
| eMedicine | neuro/533 pmr/24 |
| Patient UK | Cerebral palsy |
| MeSH | D002547 |
Cerebral palsy (CP) is a group of permanent movement disorders that appear in early childhood. Signs and symptoms vary among people. Often, symptoms include poor coordination, stiff muscles, weak muscles, and tremors. There may be problems with sensation, vision, hearing, swallowing, and speaking. Often babies with cerebral palsy do not roll over, sit, crawl, or walk as early as other children of their age. Difficulty with the ability to think or reason and seizures each occurs in about one third of people with CP. While the symptoms may get more noticeable over the first few years of life, the underlying problems do not worsen over time.[1]
Cerebral palsy is caused by abnormal development or damage to the parts of the brain that control movement, balance, and posture.[1][2] Most often the problems occur during pregnancy; however, they may also occur during childbirth, or shortly after birth. Often the cause is unknown. Risk factors include preterm birth, being a twin, certain infections during pregnancy such as toxoplasmosis or rubella, exposure to methylmercury during pregnancy, a difficult delivery, and head trauma during the first few years of life, among others.[1] About 2% of cases are believed to be due to an inherited genetic cause.[3] A number of sub-types are classified based on the specific problems present. For example, those with stiff muscles have spastic cerebral palsy, those with poor coordination have ataxic cerebral palsy, and those with writhing movements have athetoid cerebral palsy. Diagnosis is based on the child's development over time. Blood tests and medical imaging may be used to rule out other possible causes.[1]
CP is partly preventable through immunization of the mother and efforts to prevent head injuries in children such as through improved safety. There is no cure for CP; however, supportive treatments, medications, and surgery may help many individuals. This may include physical therapy, occupational therapy, and speech therapy. Medications such as diazepam, baclofen, and botulinum toxin may help relax stiff muscles. Surgery may include lengthening muscles and cutting overly active nerves. Often external braces and other assistive technology are helpful. Some affected children can achieve near normal adult lives with appropriate treatment. While alternative medicines are frequently used there is no evidence to support their use.[1]
CP is the most common movement disorder in children.[4] It occurs in about 2.1 per 1,000 live births.[5] Cerebral palsy has been documented throughout history with the first known descriptions occurring in the work of Hippocrates in the 5th century BCE. Extensive study of the condition began in the 19th century by William John Little, after whom spastic diplegia was called "Little disease".[6] William Osler first named it "cerebral palsy" from the German "zerebrale Kinderlähmung" (cerebral child-paralysis).[7] A number of potential treatments are being examined, including stem cell therapy. However, more research is required to determine if it is effective and safe.[1]
Signs and symptoms
Cerebral palsy is defined as "a group of permanent disorders of the development of movement and posture, causing activity limitation, that are attributed to non-progressive disturbances that occurred in the developing fetal or infant brain."[8] While the central feature of CP is a disorder with movement, difficulties with thinking, learning, feeling, communication and behavior often occur along with cerebral palsy.[8] Of those with CP, 28% have epilepsy, 58% have difficulties with communication, at least 42% have problems with their vision, and 23–56% have learning disabilities.[9]
Cerebral palsy is characterized by abnormal muscle tone, reflexes, or motor development and coordination. There can be joint and bone deformities and contractures (permanently fixed, tight muscles and joints). The classical symptoms are spasticity, spasms, other involuntary movements (e.g., facial gestures), unsteady gait, problems with balance, and/or soft tissue findings consisting largely of decreased muscle mass. Scissor walking (where the knees come in and cross) and toe walking (which can contribute to a gait reminiscent of a marionette) are common among people with CP who are able to walk, but taken on the whole, CP symptomatology is very diverse. The effects of cerebral palsy fall on a continuum of motor dysfunction, which may range from slight clumsiness at the mild end of the spectrum to impairments so severe that they render coordinated movement virtually impossible at the other end of the spectrum.
Babies born with severe CP often have an irregular posture; their bodies may be either very floppy or very stiff. Birth defects, such as spinal curvature, a small jawbone, or a small head sometimes occur along with CP. Symptoms may appear or change as a child gets older. Some babies born with CP do not show obvious signs right away. Classically, CP becomes evident when the baby reaches the developmental stage at 6 to 9 months and is starting to mobilise, where preferential use of limbs, asymmetry, or gross motor developmental delay is seen.[10]
Drooling is common among children with cerebral palsy, which can have a variety of impacts including social rejection, impaired speaking, damage to clothing and books, and mouth infections.[11]
An average of 55.5% of people with cerebral palsy experience lower urinary tract symptoms, more commonly excessive storage issues than voiding issues. Those with voiding issues and pelvic floor overactivity can deteriorate as adults and experience upper urinary tract dysfunction.[12]
Language
Speech and language disorders are common in people with cerebral palsy. The incidence of dysarthria is estimated to range from 31% to 88%.[13] Speech problems are associated with poor respiratory control, laryngeal and velopharyngeal dysfunction, and oral articulation disorders that are due to restricted movement in the oral-facial muscles. There are three major types of dysarthria in cerebral palsy: spastic, dyskinetic (athetosis), and ataxic.
Overall language delay is associated with problems of intellectual disability, deafness, and learned helplessness.[14] Children with cerebral palsy are at risk of learned helplessness and becoming passive communicators, initiating little communication.[14] Early intervention with this clientele, and their parents, often targets situations in which children communicate with others so that they learn that they can control people and objects in their environment through this communication, including making choices, decisions, and mistakes.[14]
Skeleton
In order for bones to attain their normal shape and size, they require the stresses from normal musculature. Osseous findings will, therefore, mirror the specific muscular deficits in a given person with CP. The shafts of the bones are often thin (gracile) and become thinner during growth. When compared to these thin shafts (diaphyses), the centres (metaphyses) often appear quite enlarged (ballooning). With a lack of use, articular cartilage may atrophy, leading to narrowed joint spaces. Depending on the degree of spasticity, a person with CP may exhibit a variety of angular joint deformities. Because vertebral bodies need vertical gravitational loading forces to develop properly, spasticity and an abnormal gait can hinder proper and/or full bone and skeletal development. People with CP tend to be shorter in height than the average person because their bones are not allowed to grow to their full potential. Sometimes bones grow to different lengths, so the person may have one leg longer than the other.
Pain and sleep
Pain is common and may result from the inherent deficits associated with the condition, along with the numerous procedures children typically face.[15] Pain is associated with tight or shortened muscles, abnormal posture, stiff joints, unsuitable orthosis, etc. There is also a high likelihood of chronic sleep disorders secondary to both physical and environmental factors.[16] Chronic pain is under-recognized in children with cerebral palsy.[17]
Eating
Those with CP may have difficulty preparing food, holding utensils, or chewing and swallowing due to sensory and motor impairments. An infant with CP may not be able to suck, swallow or chew.[18] Children with CP may have too little or too much sensitivity around and in the mouth.[18] Fine finger dexterity, like that needed for picking up a utensil, is more frequently impaired than gross manual dexterity, like that needed for spooning food onto a plate.[19] Grip strength impairments are less common.[19]
Associated disorders
Associated disorders include "intellectual disabilities, seizures, muscle contractures, abnormal gait, osteoporosis, communication disorders, malnutrition, sleep disorders, and mental health disorders, such as depression and anxiety".[20] In addition to these, "functional gastrointestinal abnormalities contributing to bowel obstruction, vomiting, and constipation" may also arise. Adults with cerebral palsy may suffer from "ischemic heart disease, cerebrovascular disease, cancer, and trauma" moreso than a non-affected population.[21]
Related conditions can include seizures, apraxia, dysarthria or other communication disorders, eating problems, sensory impairments, intellectual disability, learning disabilities, urinary incontinence, fecal incontinence, and/or behavioural disorders.
Causes
Cerebral palsy is due to abnormal development or damage occurring to the developing brain.[22] This damage can occur during pregnancy, delivery, the first month of life, or less commonly in early childhood.[22] Structural problems in the brain are seen in 80% of cases, most commonly within the white matter.[22] More than three-quarters of cases are believed to result from issues that occur during pregnancy.[22]
While in certain cases there is no identifiable cause, typical causes include problems in intrauterine development (e.g. exposure to radiation, infection, fetal growth restriction), hypoxia of the brain (thrombotic events, placental conditions), birth trauma during labor and delivery, and complications around birth or during childhood.[14][23]
Preterm birth
Between 40% and 50% of all children who develop cerebral palsy were born prematurely.[24] Most of these cases (75-90%) are believed to be due to issues that occur around the time of birth, often just after birth.[22] Multiple-birth infants are also more likely than single-birth infants to have CP.[25] They are also more likely to be born with a low birth weight.
In those who are born with a weight between 1 kg and 1.5 kg CP occurs in 6%.[5] Among those born before 28 weeks of gestation it occurs in 11%.[5] Genetic factors are believed to play an important role in prematurity and cerebral palsy generally.[26] While in those who are born between 34 and 37 weeks the risk is 0.4% (three times normal).[27]
Term infants
In babies that are born at term risk factors include problems with the placenta, birth defects, low birthweight, breathing meconium into the lungs, a delivery requiring either the use of instruments or an emergency Caesarean section, birth asphyxia, seizures just after birth, respiratory distress syndrome, low blood sugar, and infections in the baby.[28]
As of 2013 it was unclear how much of a role birth asphyxia plays as a cause.[29] It is believed, however, that only a small number of cases are caused by lack of oxygen during birth.[30] It is unclear if the size of the placenta plays a role.[31]As of 2015 it is evident that in advantaged countries, most cases of cerebral palsy in term or near-term neonates have explanations other than asphyxia.[23]
Genetics
About 2% of all CP cases are inherited, with glutamate decarboxylase-1 being one of the possible enzymes involved.[3] Most inherited cases are autosomal recessive,[3] meaning both parents must be carriers for the disorder in order to have a child with the disorder.
Early childhood
After birth, other causes include toxins, severe jaundice, lead poisoning, physical brain injury, stroke,[32] shaken baby syndrome, incidents involving hypoxia to the brain (such as near drowning), and encephalitis or meningitis.
Others
Infections in the mother, even infections that are not easily detected, may triple the risk of the child developing the disorder.[33] Infections of the fetal membranes known as chorioamnionitis increases the risk.[34]
Intrauterine and neonatal insults (many of which are infectious in nature) increase the risk.[35]
It has been hypothesised that some cases of cerebral palsy are caused by the death in very early pregnancy of an identical twin.[36]
Diagnosis
The diagnosis of cerebral palsy has historically rested on the person's history and physical examination. A general movements assessment, which involves measuring movements that occur spontaneously among those less than four months of age, appears to be most accurate.[37][38] Symptoms and diagnosis typically occur by the age of 2.[39] It is a developmental disability.[37]
Once a person is diagnosed with cerebral palsy, further diagnostic tests are optional. Neuroimaging with CT or MRI is warranted when the cause of a person's cerebral palsy has not been established. An MRI is preferred over CT due to diagnostic yield and safety. When abnormal, the neuroimaging study can suggest the timing of the initial damage. The CT or MRI is also capable of revealing treatable conditions, such as hydrocephalus, porencephaly, arteriovenous malformation, subdural hematomas and hygromas, and a vermian tumour[40] (which a few studies suggest are present 5–22% of the time). Furthermore, an abnormal neuroimaging study indicates a high likelihood of associated conditions, such as epilepsy and intellectual disability.[41]
The age at which CP is diagnosed is important, but there is disagreement over what is the best age to make the diagnosis.[38] The earlier CP is diagnosed correctly, the better the opportunities are to provide the child with physical and education help, but there might be a greater chance that CP will be confused with another problem, especially if the child is 18 months of age or younger.[38] Infants may have temporary problems with muscle tone or control that can be confused with CP, which is permanent.[38] A metabolism disorder or tumors in the nervous system may appear to be CP; metabolic disorders, in particular, can produce brain problems that look like CP on an MRI.[30] Disorders that deteriorate the white matter in the brain and problems that cause spasms and weakness in the legs, may be mistaken for CP when they first appear early in life.[38] However, these disorders get worse over time, and CP does not[38] (although it may change in character).[30] In infancy it may not be possible to tell the difference between them.[38] Fragile X syndrome (a cause of autism and intellectual disability) and general intellectual disability must also be ruled out.[38] Cerebral palsy specialist John McLaughlin recommends waiting until the child is 36 months of age before making a diagnosis.[38]
Classification
CP is classified by the types of motor impairment of the limbs or organs, and by restrictions to the activities an affected person may perform.[42] The Gross Motor Function Classification System-Expanded and Revised and the Manual Abilities Classification System are used to describe mobility and manual dexterity in people with cerebral palsy, and recently the Communication Function Classification System, and the Eating and Drinking Ability Classification System have been proposed to describe those functions.[43] There are three main CP classifications by motor impairment: spastic, ataxic, and athetoid/dyskinetic. Additionally, there is a mixed type that shows a combination of features of the other types. These classifications reflect the areas of the brain that are damaged.
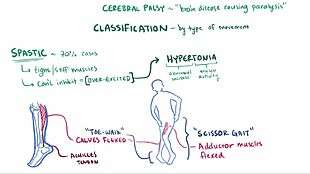
Spastic
Spastic cerebral palsy, or cerebral palsy where spasticity (muscle tightness) is the exclusive or almost exclusive impairment present, is by far the most common type of overall cerebral palsy, occurring in upwards of 70% of all cases.[44] People with this type of CP are hypertonic and have what is essentially a neuromuscular mobility impairment (rather than hypotonia or paralysis) stemming from an upper motor neuron lesion in the brain as well as the corticospinal tract or the motor cortex. This damage impairs the ability of some nerve receptors in the spine to receive gamma-Aminobutyric acid properly, leading to hypertonia in the muscles signaled by those damaged nerves.
As compared to other types of CP, and especially as compared to hypotonic or paralytic mobility disabilities, spastic CP is typically more easily manageable by the person affected, and medical treatment can be pursued on a multitude of orthopedic and neurological fronts throughout life. In any form of spastic CP, clonus of the affected limb(s) may sometimes result, as well as muscle spasms resulting from the pain and/or stress of the tightness experienced. The spasticity can and usually does lead to a very early onset of muscle stress symptoms like arthritis and tendinitis, especially in ambulatory individuals in their mid-20s and early-30s. Occupational therapy and physical therapy regimens of assisted stretching, strengthening, functional tasks, and/or targeted physical activity and exercise are usually the chief ways to keep spastic CP well-managed. If the spasticity is too much for the person to handle, other remedies may be considered, such as antispasmodic medications, botulinum toxin, baclofen, or even a neurosurgery known as a selective dorsal rhizotomy (which eliminates the spasticity by reducing the excitatory neural response in the nerves causing it).
Ataxic
Ataxia-type symptoms can be caused by damage to the cerebellum. Ataxia is a less common type of cerebral palsy, occurring between 5% and 10% of all cases.[45] Some of these individuals have hypotonia and tremors. Motor skills such as writing, typing, or using scissors might be affected, as well as balance, especially while walking. It is common for individuals to have difficulty with visual and/or auditory processing. They usually have an awkward gait and as well with some dysarthria.
Athetoid
Athetoid cerebral palsy or dyskinetic cerebral palsy is mixed muscle tone — hypertonia and hypotonia mixed with involuntary motions. People with dyskinetic CP have trouble holding themselves in an upright, steady position for sitting or walking, and often show involuntary motions. For some people with dyskinetic CP, it takes a lot of work and concentration to get their hand to a certain spot (like scratching their nose or reaching for a cup). Because of their mixed tone and trouble keeping a position, they may not be able to hold onto objects, especially small ones requiring fine motor control (such as a toothbrush or pencil). About 10% of individuals with CP are classified as dyskinetic CP but some have mixed forms with spasticity and dyskinesia. The damage occurs to the extrapyramidal motor system and/or pyramidal tract and to the basal ganglia. In newborn infants, high bilirubin levels in the blood, if left untreated, can lead to brain damage in the basal ganglia (kernicterus), which can lead to dyskinetic cerebral palsy.
Mixed
Mixed cerebral palsy has symptoms of athetoid, ataxic and spastic CP appearing simultaneously, each to varying degrees, and both with and without symptoms of each. Mixed CP is the most difficult to treat as it is extremely heterogeneous and sometimes unpredictable in its symptoms and development over the lifespan.
Prevention
Electronic fetal monitoring has not helped to prevent CP, and in 2014 the American College of Obstetricians and Gynecologists, the Royal Australian and New Zealand College of Obstetricians and Gynaecologists, and the Society of Obstetricians and Gynaecologists of Canada have acknowledged that there are no long-term benefits of electronic fetal monitoring.[23] In those at risk of an early delivery, magnesium sulphate appears to decrease the risk of cerebral palsy.[46][47] It is unclear if it helps those who are born at term.[48] Caffeine is used to treat apnea of prematurity and reduces the risk of cerebral palsy in premature babies, but there are also concerns of long term negative effects.[49]
Cooling high-risk full-term babies shortly after birth may reduce disability.[50]
Management
Over time, the approach to CP management has shifted away from narrow attempts to fix individual physical problems – such as spasticity in a particular limb – to making such treatments part of a larger goal of maximizing the person's independence and community engagement.[51]:886 However, the evidence base for the effectiveness of intervention programs reflecting this philosophy has not yet caught up: effective interventions for body structures and functions have a strong evidence base, but evidence is lacking for effective interventions targeted toward participation, environment, or personal factors.[51] There is also no good evidence to show that an intervention that is effective at the body-specific level will result in an improvement at the activity level, or vice versa.[51] Although such cross-over benefit might happen, not enough high-quality studies have been done to demonstrate it.[51]
A review on the treatment of drooling in children with cerebral palsy found that it was not possible to tell whether interventions worked or were safe, including botulin toxin A and benztropine and glycopyrrolate.[11]
Various forms of therapy are available to people living with cerebral palsy as well as caregivers and parents. Treatment may include one or more of the following: physical therapy; occupational therapy; speech therapy; water therapy; drugs to control seizures, alleviate pain, or relax muscle spasms (e.g. benzodiazepines); surgery to correct anatomical abnormalities or release tight muscles; braces and other orthotic devices; rolling walkers; and communication aids such as computers with attached voice synthesisers. The treatments with the best evidence are medications (anticonvulsants, botulinum toxin, bisphosphonates, diazepam), therapy (bimanual training, casting, constraint-induced movement therapy, context-focused therapy, fitness training, goal-directed training, hip surveillance, home programmes, occupational therapy after botulinum toxin, pressure care) and surgery (selective dorsal rhizotomy).[51]
Therapy

Physiotherapy programs are designed to encourage the patient to build a strength base for improved gait and volitional movement, together with stretching programs to limit contractures. Many experts believe that lifelong physiotherapy is crucial to maintaining muscle tone, bone structure, and prevent dislocation of the joints.
Speech therapy helps control the muscles of the mouth and jaw, and helps improve communication. Just as CP can affect the way a person moves their arms and legs, it can also affect the way they move their mouth, face and head. This can make it hard for the person to breathe; talk clearly; and bite, chew and swallow food. Speech therapy often starts before a child begins school and continues throughout the school years.[52]
Conductive education (CE) was developed in Hungary from 1945 based on the work of András Pető. It is a unified system of rehabilitation for people with neurological disorders including cerebral palsy, Parkinson's disease and multiple sclerosis, amongst other conditions. It is theorised to improve mobility, self-esteem, stamina and independence as well as daily living skills and social skills. The conductor is the professional who delivers CE in partnership with parents and children. Skills learned during CE should be applied to everyday life and can help to develop age-appropriate cognitive, social and emotional skills. It is available at specialised centres.
Biofeedback is a therapy in which people learn how to control their affected muscles. Biofeedback therapy has been found to significantly improve gait in children with cerebral palsy.[53]
Massage therapy is designed to help relax tense muscles, strengthen muscles, and keep joints flexible.[54] More research is needed to determine the health benefits of these therapies for people with CP.
Occupational therapy helps adults and children maximise their function, adapt to their limitations and live as independently as possible.[55][56] A family-centred philosophy is used with children who have CP. Occupational therapists work closely with families in order to address their concerns and priorities for their child.[57]
CP commonly causes hemiplegia.[58] Those with hemiplegia have limited use of the limbs on one side of the body, and have normal use of the limbs on the other side.[58] Hemiplegics often adapt by ignoring the limited limbs, and performing nearly all activities with the unaffected limbs, which can lead to increased problems with muscle tone, motor control and range of motion.[58] An emerging technique called constraint-induced movement therapy (CIMT) is designed to address this.[58] In CIMT, the unaffected limbs are constrained, forcing the individual to learn to use the affected limbs.[58] As of 2007 there was limited, preliminary evidence that CIMT is effective, but more study is needed before it can be recommended with confidence.[58]
However, there is only some benefit from therapy. Treatment is usually symptomatic and focuses on helping the person to develop as many motor skills as possible or to learn how to compensate for the lack of them. Nonspeaking people with CP are often successful availing themselves of augmentative and alternative communication (AAC).[59]
Medication
Botulinum toxin injections are given into muscles that are spastic or sometimes dystonic, the aim being to reduce the muscle hypertonus that can be painful. A reduction in muscle tone can also facilitate bracing and the use of orthotics. Most often lower extremity muscles are injected. Botulinum toxin is focal treatment, meaning that a limited number of muscles can be injected at the same time. The effect of the toxin is reversible and a reinjection is needed every 4–6 months.[60]
In children it decreases spasticity and improve range of motion and thus has become commonly used.[61]
Surgery
Surgery usually involves one or a combination of:
- Loosening tight muscles and releasing fixed joints, most often performed on the hips, knees, hamstrings, and ankles. In rare cases, this surgery may be used for people with stiffness of their elbows, wrists, hands, and fingers. Selective Percutaneous Myofascial Lengthening (SPML) is one example.
- The insertion of a baclofen pump usually during the stages while a person is a young adult. This is usually placed in the left abdomen. It is a pump that is connected to the spinal cord, whereby it releases doses of baclofen to alleviate continuous muscle flexion. Baclofen is a muscle relaxant and is often given by mouth to patients to help counter the effects of spasticity.
- Straightening abnormal twists of the leg bones, i.e. femur (termed femoral anteversion or antetorsion) and tibia (tibial torsion). This is a secondary complication caused by the spastic muscles generating abnormal forces on the bones, and often results in intoeing (pigeon-toed gait). The surgery is called derotation osteotomy, in which the bone is broken (cut) and then set in the correct alignment.[62]
- Cutting nerves on the limbs most affected by movements and spasms. This procedure, called a rhizotomy ("rhizo" meaning root and "tomy" meaning "a cutting of" from the Greek suffix tomia), reduces spasms and allows more flexibility and control of the affected limbs and joints.[63][64]
Other surgical procedures are available to try to help with other problems. Those who have serious difficulties with eating may undergo a procedure called a gastrostomy: a hole is cut through the belly skin and into the stomach to allow for a feeding tube.[65] There is no good evidence about the effectiveness or safety of gastrostomy.[65]
Orthotics
Orthotic devices such as ankle-foot orthoses (AFOs) are often prescribed to achieve the following objectives: correct and/or prevent deformity, provide a base of support, facilitate training in skills, and improve the efficiency of gait.[66]
The available evidence suggests that orthoses can have positive effects on all temporal and spatial parameters of gait, i.e. velocity, cadence, step length, stride length, single and double support.[67] AFOs have also been found to reduce energy expenditure.[68]
Assistive technology
There are now assistive technologies designed to help when dealing with cerebral palsy, most often to aid with meal times. There are manual feeding aids, for example, one designed using viscous fluid damping to smooth out essential tremors associated with cerebral palsy. There are also electronic feeding aids on the market suitable for anyone who can chew and swallow but unable to feed themselves. There are manual drinking aids available, designed with non-return valves and holders or non-slip surfaces, to enable users who lack the ability to drink from a cup or glass, due to tremors, weakness or limited head mobility, to gain a route to hydration and nutrition.
Other
Hyperbaric oxygen therapy (HBOT), in which pressurised oxygen is inhaled inside a hyperbaric chamber, has been studied under the theory that improving oxygen availability to damaged brain cells can reactivate some of them to function normally. HBOT results in no significant difference from that of pressurised room air, however, and some children undergoing HBOT may experience adverse events such as seizures and the need for ear pressure equalisation tubes.[69]
Patterning is a controversial form of alternative therapy for people with CP. The method is promoted by The Institutes for the Achievement of Human Potential (IAHP), a Philadelphia nonprofit organisation, but has been criticised by the American Academy of Pediatrics.[70]
Music therapy has been used in CP to motivate or relax children, or used as auditory feedback.[71]
While there is great interest in using video game rehabilitation with children with cerebral palsy, it is difficult to compare outcomes between studies, and therefore to reach evidence-based conclusions on its effectiveness.[72]
Reviews disagree on the usefulness of therapy with horses – one found there was a positive effect on large scale motor function and another found that there was no evidence of improvements.[73][74]
Family centered care is a paradigm that is often used with families with a child with CP. A review of how parents facilitate their child's participation found that parents typically "enable and support performance of meaningful activities" and "enable, change and use the environment", but that there is little written on parents' needs.[75]
Prognosis
CP is not a progressive disorder (meaning the brain damage does not worsen), but the symptoms can become more severe over time. A person with the disorder may improve somewhat during childhood if he or she receives extensive care, but once bones and musculature become more established, orthopedic surgery may be required. The full intellectual potential of a child born with CP will often not be known until the child starts school. People with CP are more likely to have learning disabilities, although these may be unrelated to IQ, including varying degrees of intellectual disability. Intellectual level among people with CP varies from genius to intellectually impaired, as it does in the general population, and experts have stated that it is important not to underestimate the capabilities of a person with CP and to give them every opportunity to learn.[76]
The ability to live independently with CP varies widely, depending partly on the severity of each person's impairment and partly on the capability of each person to self-manage the logistics of life. Some individuals with CP require personal assistant services for all activities of daily living. Others only need assistance with certain activities, and still others do not require any physical assistance. But regardless of the severity of a person's physical impairment, a person's ability to live independently often depends primarily on the person's capacity to manage the physical realities of his or her life autonomously. In some cases, people with CP recruit, hire, and manage a staff of personal care assistants (PCAs). PCAs facilitate the independence of their employers by assisting them with their daily personal needs in a way that allows them to maintain control over their lives.
CP can significantly reduce a person's life expectancy, depending on the severity of their condition and the quality of care with which they are provided.[22][77] The ability to ambulate, roll, and self-feed has been associated with increased life expectancy.[78] There is currently no evidence that CP affects fertility, although some of the secondary symptoms have been shown to affect sexual desire and performance.[79]
Self-care
For many children with CP, parents are heavily involved in self-care activities. Self-care activities, such as bathing, dressing, grooming, can be difficult for children with CP as self-care depends primarily on use of the upper limbs.[80] For those living with CP, impaired upper limb function affects almost 50% of children and is considered the main factor contributing to decreased activity and participation.[81] As the hands are used for many self-care tasks, sensory and motor impairments of the hands make daily self-care more difficult.[19][82] Motor impairments cause more problems than sensory impairments.[19] The most common impairment is that of finger dexterity, which is the ability to manipulate small objects with the fingers.[19]
Productivity
The effects of sensory, motor and cognitive impairments affect self-care occupations in children with CP and productivity occupations. Productivity can include, but is not limited to, school, work, household chores or contributing to the community.[83]
Play is included as a productive occupation as it is often the primary activity for children.[84] If play becomes difficult due to a disability, like CP, this can cause problems for the child.[85] These difficulties can affect a child's self-esteem.[85] In addition, the sensory and motor problems experienced by children with CP affect how the child interacts with their surroundings, including the environment and other people.[85] Not only do physical limitations affect a child's ability to play, the limitations perceived by the child's caregivers and playmates also impact the child's play activities.[86] Some children with disabilities spend more time playing by themselves.[87] When a disability prevents a child from playing, there may be social, emotional and psychological problems[88] which can lead to increased dependence on others, less motivation and poor social skills.[89]
In school, students are asked to complete many tasks and activities, many of which involve handwriting. Many children with CP have the capacity to learn and write in the school environment.[90] However, students with CP may find it difficult to keep up with the handwriting demands of school and their writing may be difficult to read.[90] In addition, writing may take longer and require greater effort on the student's part.[90] Factors linked to handwriting include postural stability, sensory and perceptual abilities of the hand, and writing tool pressure.[90]
Speech impairments may be seen in children with CP depending on the severity of brain damage.[91] Communication in a school setting is important because communicating with peers and teachers is very much a part of the "school experience" and enhances social interaction. Problems with language or motor dysfunction can lead to underestimating a student's intelligence.[92] In summary, children with CP may experience difficulties in school, such as difficulty with handwriting, carrying out school activities, communicating verbally and interacting socially.
Leisure
Leisure activities can have several positive effects on physical health, mental health, life satisfaction and psychological growth for people with physical disabilities like CP.[93] Common benefits identified are stress reduction, development of coping skills, companionship, enjoyment, relaxation and a positive effect on life satisfaction.[94] In addition, for children with CP, leisure appears to enhance adjustment to living with a disability.[94]
Leisure can be divided into structured (formal) and unstructured (informal) activities.[95] Studies show that children with disabilities, like CP, participate mainly in informal activities that are carried out in the family environment and are organised by adults.[96] Typically, children with disabilities carry out leisure activities by themselves or with their parents rather than with friends. Therefore, children may experience limited diversity of activities and social engagements, as well as a more passive lifestyle than their peers.[96] Although leisure is important for children with CP, they may have difficulties carrying out leisure activities due to social and physical barriers.
Participation and barriers
Participation is involvement in life situations and everyday activities.[97] Participation includes the domains of self-care, productivity and leisure. In fact, communication, mobility, education, home life, leisure and social relationships require participation and are indicators of the extent to which a child functions in his or her environment.[97] Barriers can exist on three levels: micro, meso and macro.[98] First, the barriers at the micro level involve the person.[98] Barriers at the micro level include the child's physical limitations (motor, sensory and cognitive impairments) or their subjective feelings regarding their ability to participate.[99] For example, the child may not participate in group activities due to lack of confidence. Second, barriers at the meso level include the family and community.[98] These may include negative attitudes of people toward disability or lack of support within the family or in the community.[100] One of the main reasons for this limited support appears to be the result of a lack of awareness and knowledge regarding the child's ability to engage in activities despite his or her disability.[100] Third, barriers at the macro level incorporate the systems and policies that are not in place or hinder children with CP. These may be environmental barriers to participation such as architectural barriers, lack of relevant assistive technology and transportation difficulties due to limited wheelchair access or public transit that can accommodate children with CP.[100] For example, a building without an elevator will prevent the child from accessing higher floors.
A 2013 review stated that outcomes for adults with cerebral palsy without intellectual disability in the 2000s were that "60-80% completed high school, 14-25% completed college, up to 61% were living independently in the community, 25-55% were competitively employed, and 14-28% were involved in long term relationships with partners or had established families".[101]
Aging
Because children with cerebral palsy are often told that it is a non-progressive disease, they may be unprepared for the greater impacts of the aging process as they head into their 30s.[102] 25% or more adults with cerebral palsy who can walk experience increasing difficulties walking with age.[103] Chronic disease risk, such as obesity, is also higher among adults with cerebral palsy than the general population.[104]
Like they did in childhood, adults with cerebral palsy experience psychosocial issues related to their CP, chiefly the need for social support, self-acceptance, and acceptance by others. Workplace accommodations may be needed to enhance continued employment for adults with CP as they age. Rehabilitation or social programs that include Salutogenesis may improve the coping potential of adults with CP as they age.[105]
Epidemiology
Cerebral palsy occurs in about 2.1 per 1000 live births.[5] In those born at term rates are lower at 1 per 1000 live births.[22] Rates appear to be similar in both the developing and developed world.[22] Within a population it may occur more often in poorer people.[106] The rate is higher in males than in females; in Europe it is 1.3 times more common in males.[107] Variances in reported rates of incidence or prevalence across different geographical areas in industrialised countries are thought to be caused primarily by discrepancies in the criteria used for inclusion and exclusion. When such discrepancies are taken into account in comparing two or more registers of patients with cerebral palsy (for example, the extent to which children with mild cerebral palsy are included), the prevalence rates converge toward the average rate of 2:1000.
Overall, advances in care of pregnant mothers and their babies has not resulted in a noticeable decrease in CP. This is generally attributed to medical advances in areas related to the care of premature babies (which results in a greater survival rate). Only the introduction of quality medical care to locations with less-than-adequate medical care has shown any decreases. The incidence of CP increases with premature or very low-weight babies regardless of the quality of care.[108]
Prevalence of cerebral palsy is best calculated around the school entry age of about 6 years, the prevalence in the U.S. is estimated to be 2.4 out of 1000 children.[109]
According to the Australian Bureau of Statistics, in 2014, 104 Australians died of cerebral palsy.[110]
History
Cerebral palsy has affected humans since antiquity. A decorated grave marker dating from around the 15th to 14th century BCE shows a figure with one small leg and using a crutch, possibly due to cerebral palsy. The oldest likely physical evidence of the condition comes from the mummy of Siptah, an Egyptian Pharaoh who ruled from about 1196 to 1190 BCE and died at about 20 years of age. The presence of cerebral palsy has been suspected due to his deformed foot and hands.[6]
The medical literature of the ancient Greeks discusses paralysis and weakness of the arms and legs; the modern word palsy comes from the Ancient Greek words παράλυση or πάρεση, meaning paralysis or paresis respectively. The works of the school of Hippocrates (460–c. 370 BCE), and the manuscript On the Sacred Disease in particular, describe a group of problems that matches up very well with the modern understanding of cerebral palsy. The Roman Emperor Claudius (10 BCE–54 CE) is suspected of having CP, as historical records describe him as having several physical problems in line with the condition. Medical historians have begun to suspect and find depictions of CP in much later art. Several paintings from the 16th century and later show individuals with problems consistent with it, such as Jusepe de Ribera's 1642 painting The Clubfoot.[6]
The modern understanding of CP as resulting from problems within the brain began in the early decades of the 1800s with a number of publications on brain abnormalities by Johann Christian Reil, Claude François Lallemand and Philippe Pinel. Later physicians used this research to connect problems in the brain with specific symptoms. The English surgeon William John Little (1810–1894) was the first person to study CP extensively. In his doctoral thesis he stated that CP was a result of a problem around the time of birth. He later identified a difficult delivery, a preterm birth and perinatal asphyxia in particular as risk factors. The spastic diplegia form of CP came to be known as Little's disease. In the 1880s British neurologist William Gowers built on Little's work by linking paralysis in newborns to difficult births. He named the problem "birth palsy" and classified birth palsies into two types: peripheral and cerebral.[6]
Working in Pennsylvania in the 1880s, Canadian-born physician William Osler (1849–1919) reviewed dozens of CP cases to further classify the disorders by the site of the problems on the body and by the underlying cause. Osler made further observations tying problems around the time of delivery with CP, and concluded that problems causing bleeding inside the brain were likely the root cause. Osler also suspected polioencephalitis as an infectious cause. Through the 1890s, scientists commonly confused CP with polio.[6]
Before moving to psychiatry, Austrian neurologist Sigmund Freud (1856–1939) made further refinements to the classification of the disorder. He produced the system still being used today. Freud's system divides the causes of the disorder into problems present at birth, problems that develop during birth, and problems after birth. Freud also made a rough correlation between the location of the problem inside the brain and the location of the affected limbs on the body, and documented the many kinds of movement disorders.[6]
In the early 20th century, the attention of the medical community generally turned away from CP until orthopedic surgeon Winthrop Phelps became the first physician to treat the disorder. He viewed CP from a musculoskeletal perspective instead of a neurological one. Phelps developed surgical techniques for operating on the muscles to address issues such as spasticity and muscle rigidity. Hungarian physical rehabilitation practitioner András Pető developed a system to teach children with CP how to walk and perform other basic movements. Pető's system became the foundation for conductive education, widely used for children with CP today. Through the remaining decades, physical therapy for CP has evolved, and has become a core component of the CP management program.[6]
In 1997, Robert Palisano et al. introduced the Gross Motor Function Classification System (GMFCS) as an improvement over the previous rough assessment of limitation as either mild, moderate or severe.[42] The GMFCS grades limitation based on observed proficiency in specific basic mobility skills such as sitting, standing and walking, and takes into account the level of dependency on aids such as wheelchairs or walkers. The GMFCS was further revised and expanded in 2007.[42]
Society and culture
Economic impact
Access Economics has released a report on the economic impact of cerebral palsy in Australia. The report found that, in 2007, the financial cost of cerebral palsy (CP) in Australia was $1.47 billion or 0.14% of GDP.[111] Of this:
- 1.03 billion (69.9%) was productivity lost due to lower employment, absenteeism and premature death of Australians with CP
- 141 million (9.6%) was the DWL from transfers including welfare payments and taxation forgone
- 131 million (9.0%) was other indirect costs such as direct program services, aides and home modifications and the bringing-forward of funeral costs
- 129 million (8.8%) was the value of the informal care for people with CP
- 40 million (2.8%) was direct health system expenditure
The value of lost well-being (disability and premature death) was a further $2.4 billion.
In per capita terms, this amounts to a financial cost of $43,431 per person with CP per annum. Including the value of lost well-being, the cost is over $115,000 per person per annum.
Individuals with CP bear 37% of the financial costs, and their families and friends bear a further 6%. Federal government bears around one-third (33%) of the financial costs (mainly through taxation revenues forgone and welfare payments). State governments bear under 1% of the costs, while employers bear 5% and the rest of society bears the remaining 19%. If the burden of disease (lost well-being) is included, individuals bear 76% of the costs.
The average lifetime cost for people with CP in the US is $921,000 per individual, including lost income.[112]
In the United States many states allow Medicaid beneficiaries to use their Medicaid funds to hire their own PCAs, instead of forcing them to use institutional or managed care.[113]
In India, the government-sponsored program called "NIRAMAYA" for the medical care of children with neurological and muscular deformities has proved to be an ameliorating economic measure for persons with such disabilities. It has shown that persons with mental or physically debilitating congenital disabilities can lead better lives if they have financial independence.
Use of the term
The term palsy in modern language refers to a disorder of movement, but the word root "palsy" technically means "paralysis", even though it is not used as such within the meaning of cerebral palsy. The use of "palsy" in the term cerebral palsy makes it important to note that paralytic disorders are in fact not cerebral palsy – meaning that the condition of tetraplegia, which comes from spinal cord injury or traumatic brain injury, should not be confused with spastic quadriplegia, which doesn't, nor should tardive dyskinesia be confused with dyskinetic cerebral palsy or the condition of (paralytic) "diplegia" with spastic diplegia. In fact, as of the early 21st century some clinicians have become so distressed at common incorrect use of these terms that they have resorted to new naming schemes rather than trying to reclaim the classic ones; one such example of this evolution is the increasing use of the term bilateral spasticity to refer to spastic diplegia. Such clinicians even argue quite often that the "new" term is technically more clinically accurate than the established term.
Many people would rather be referred to as a person with a disability instead of handicapped. "Cerebral Palsy: A Guide for Care" at the University of Delaware offers the following guidelines:
Impairment is the correct term to use to define a deviation from normal, such as not being able to make a muscle move or not being able to control an unwanted movement. Disability is the term used to define a restriction in the ability to perform a normal activity of daily living which someone of the same age is able to perform. For example, a three-year-old child who is not able to walk has a disability because a normal three-year-old can walk independently. A handicapped child or adult is one who, because of the disability, is unable to achieve the normal role in society commensurate with his age and socio-cultural milieu. As an example, a sixteen-year-old who is unable to prepare his own meal or care for his own toilet or hygiene needs is handicapped. On the other hand, a sixteen-year-old who can walk only with the assistance of crutches but who attends a regular school and is fully independent in activities of daily living is disabled but not handicapped. All disabled people are impaired, and all handicapped people are disabled, but a person can be impaired and not necessarily be disabled, and a person can be disabled without being handicapped.[114]
The term "spastic" denotes the attribute of spasticity in types of spastic CP. In 1952 a UK charity called The Spastics Society was formed.[115] The term "spastics" was used by the charity as a term for people with CP. The word "spastic" has since been used extensively as a general insult to disabled people, which some see as extremely offensive. They are also frequently used to insult able-bodied people when they seem overly uncoordinated, anxious, or unskilled in sports. The charity changed its name to Scope in 1994.[115] In the United States the word spaz has the same usage as an insult, but is not generally associated with CP.[116]
It is not taken as derogatory in the Indian context. Rather, "spasticity" and "cerebral palsy" are used interchangeably. The term is widely used to connote cerebral palsy and is accepted for usage in medical fraternity as well as in social life. Many organisations known as "Spastic Societies" viz. Spastic Society of Gurgaon are working in different areas in India as charitable bodies for people with cerebral palsy, in care-taking, rehabilitation and medical support of children with neurological muscular development disabilities.
Media
Maverick documentary filmmaker Kazuo Hara criticises the mores and customs of Japanese society in an unsentimental portrait of adults with cerebral palsy in his 1972 film Goodbye CP (Sayonara CP). Focusing on how the CP victims are generally ignored or disregarded in Japan, Hara challenges his society's taboos about physical handicaps. Using a deliberately harsh style, with grainy black-and-white photography and out-of-sync sound, Hara brings a stark realism to his subject.[117]
Spandan (2012), a film by Vegitha Reddy and Aman Tripathi, delves into the dilemma of parents whose child suffers from cerebral palsy. While films made with children with special needs as central characters have been attempted before, the predicament of parents dealing with the stigma associated with the condition and beyond is dealt in Spandan. In one of the songs of Spandan "Chal chaal chaal tu bala" more than 50 CP kids have acted. The famous classical singer Devaki Pandit has given her voice to the song penned by Prof. Jayant Dhupkar and composed by National Film Awards winner Isaac Thomas Kottukapally.[118][119][120][121]
My Left Foot (1989) is a drama film directed by Jim Sheridan and starring Daniel Day-Lewis. It tells the true story of Christy Brown, an Irishman born with cerebral palsy, who could control only his left foot. Christy Brown grew up in a poor, working-class family, and became a writer and artist. It won the Academy Award for Best Actor (Daniel Day-Lewis) and Best Actress in a Supporting Role (Brenda Fricker). It was also nominated for Best Director, Best Picture and Best Writing, Screenplay Based on Material from Another Medium. It also won the New York Film Critics Circle Award for Best Film for 1989.[122]
Call the Midwife (2012–) has featured two episodes with actor Colin Young, who he himself has cerebral palsy, playing a character with the same disability. His story lines have focused on the segregation of those with disabilities in the UK in the 1950s, and also romantic relationships between people with disabilities.[123]
Notable cases
- Josh Blue, winner of the fourth season of NBC's Last Comic Standing, whose act revolves around his CP.[124] Blue was also on the 2004 U.S. Paralympic soccer team.[125]
- Jason Benetti, Play-by-play broadcaster for ESPN, Fox Sports, Westwood One, and Time Warner covering football, baseball, lacrosse, hockey, and basketball. Beginning in 2016, he will also be the television play-by-play announcer for Chicago White Sox home games.[126]
- Jack Carroll, British comedian and runner-up in the seventh season of Britain's Got Talent.[127]
- Abbey Curran, American beauty queen who represented Iowa at Miss USA 2008 and was the first contestant with a disability to compete. She also made an appearance on The Ellen DeGeneres Show and CBS The Early Show.[128][129][130]
- Geri Jewell had a regular role in the prime-time series The Facts of Life.[131] She has had roles on Sesame Street, 21 Jump Street, The Young and the Restless and Deadwood.[132]
- Francesca Martinez, British stand-up comedian and actress.[133]
- Harold Elwood Yuker, a psychologist and educator at Hofstra University, Distinguished Professor of Psychology and a founding director of the Center for the Study of Attitudes Toward Persons with Disabilities, widely recognised as a critic of the tendency of some disabled people to keep to themselves. His motto was The most important thing for anyone with a disability is to learn to get along in a nondisabled world.[134]
- Evan O'Hanlon, Australian Paralympian, the fastest athlete with cerebral palsy in the world.[135]
- Arun Shourie's son Aditya about whom he has written a book Does He Know a Mother's Heart[136]
- Maysoon Zayid, the self-described "Palestinian Muslim virgin with cerebral palsy, from New Jersey", who is an actress, stand-up comedian and activist.[137] Zayid has been a resident of Cliffside Park, New Jersey.[138] She is considered one of America's first Muslim women comedians and the first person ever to perform standup in Palestine and Jordan.[139]
- RJ Mitte, an American actor best known for his role as Walter White Jr. in Breaking Bad. He is also a celebrity ambassador for United Cerebral Palsy.[140]
- Zach Anner, an American comedian, actor and writer. He had a television series on Oprah Winfrey's OWN called Rollin' With Zach and is the author of If at Birth You Don't Succeed.[141]
- Kaine, a member of the popular Atlanta, Georgia-based hip-hop duo The Ying Yang Twins, has a mild form of cerebral palsy that causes him to limp.[142]
- Ashwin Karthik, born with cerebral palsy, became first quadriplegic student in India to become a Computer Science engineering graduate[143] in 2013.[144]
- Hannah Cockroft is a British wheelchair athlete specialising in sprint distances in the T34 classification. She holds the Paralympic and world records for the 100 metres, 200 metres and 400 metres in her classification.[145][146][147]
Research
Stem cell therapy is being studied as a treatment.[148] A potential treatment for some forms of cerebral palsy may be deep brain stimulation.[149]
As of 2016 it is thought that research in genetics and genomics, teratology, and developmental neuroscience is going to yield greater understanding of cerebral palsy.[23]
See also
- Cerebral palsy sport classification - describes the disability sport classification for cerebral palsy.
References
- 1 2 3 4 5 6 "Cerebral Palsy: Hope Through Research". National Institute of Neurological Disorders and Stroke. February 2, 2015. Retrieved 4 March 2015.
- ↑ "Cerebral Palsy: Overview". National Institutes of Health. September 5, 2014. Retrieved 4 March 2015.
- 1 2 3 "CEREBRAL PALSY, SPASTIC QUADRIPLEGIC, 1; CPSQ1". Online Mendelian Inheritance in Man. November 13, 2012. Retrieved 4 March 2015.
- ↑ "How many people are affected?". National Institutes of Health. September 5, 2014. Retrieved 4 March 2015.
- 1 2 3 4 Oskoui, M; Coutinho, F; Dykeman, J; Jetté, N; Pringsheim, T (Jun 2013). "An update on the prevalence of cerebral palsy: a systematic review and meta-analysis.". Developmental Medicine & Child Neurology. 55 (6): 509–19. doi:10.1111/dmcn.12080. PMID 23346889.
- 1 2 3 4 5 6 7 Panteliadis, C; Panteliadis, P; Vassilyadi, F (Apr 2013). "Hallmarks in the history of cerebral palsy: from antiquity to mid-20th century.". Brain & Development. 35 (4): 285–92. doi:10.1016/j.braindev.2012.05.003. PMID 22658818.
- ↑ "What is cerebral palsy?". Archived from the original on 7 July 2015.
- 1 2 Rosenbaum, P; Paneth, N; Leviton, A; Goldstein, M; Bax, M; Damiano, D; Dan, B; Jacobsson, B (2007). "A report: The definition and classification of cerebral palsy April 2006". Developmental Medicine & Child Neurology Supplement. 109: 8–14. doi:10.1111/j.1469-8749.2007.tb12610.x. PMID 17370477.; Corrected in Rosenbaum, P; Paneth, N; Leviton, A; Goldstein, M; Bax, M; Damiano, D; Dan, B; Jacobsson, B (2007). "A report: The definition and classification of cerebral palsy April 2006". Developmental Medicine & Child Neurology. 49: 480. doi:10.1111/j.1469-8749.2007.00480.x.
- ↑ Kent, Ruth (2013). "Chapter 38: Cerebral Palsy". In Barnes MP, Good DC. Handbook of Clinical Neurology. 3. 110. Elsevier. pp. 443–459. ISBN 978-0444529015.
- ↑ Agarwal, Anil; Verma, Indreshwar (December 2012). "Cerebral palsy in children: An overview". Journal of Clinical Orthopaedics and Trauma. 3 (2): 77–81. doi:10.1016/j.jcot.2012.09.001.
- 1 2 Walshe, M; Smith, M; Pennington, L (14 November 2012). "Interventions for drooling in children with cerebral palsy.". The Cochrane database of systematic reviews. 11: CD008624. doi:10.1002/14651858.CD008624.pub3. PMID 23152263.
- ↑ Samijn, Bieke; Van Laecke, Erik; Renson, Catherine; Hoebeke, Piet; Plasschaert, Frank; Vande Walle, Johan; Van den Broeck, Christine (February 2016). "Lower urinary tract symptoms and urodynamic findings in children and adults with cerebral palsy: A systematic review". Neurourology and Urodynamics: n/a–n/a. doi:10.1002/nau.22982.
- ↑ Hirsh Adam T.; Gallegos Juan C.; Gertz Kevin J.; Engel Joyce M.; Jensen Mark P. (2010). "Symptom Burden in Individuals with Cerebral Palsy". Journal of Rehabilitation Research & Development. 47 (9): 863–67. doi:10.1682/jrrd.2010.03.0024.
- 1 2 3 4 Beukelman, David R.; Mirenda, Pat (1999). Augmentative and Alternative Communication: Management of severe communication disorders in children and adults (2nd ed.). Baltimore: Paul H Brookes Publishing Co. pp. 246–249. doi:10.1080/07434619912331278735. ISBN 1-55766-333-5.
- ↑ McKearnan K.A.; Kieckhefer G.M.; Engel J.M.; Jensen M.P.; Labyak S. (2004). "Pain in children with cerebral palsy: A review". Journal of Neuroscience Nursing. 26 (5): 252–259. doi:10.1097/01376517-200410000-00004.
- ↑ Newman C.J.; O'Regan M.; Hensey O. (2006). "Sleep disorders in children with cerebral palsy". Developmental Medicine & Child Neurology. 48 (7): 564–8. doi:10.1017/S0012162206001198. PMID 16780625.
- ↑ Kingsnorth, S.; Orava, T.; Provvidenza, C.; Adler, E.; Ami, N.; Gresley-Jones, T.; Mankad, D.; Slonim, N.; Fay, L.; Joachimides, N.; Hoffman, A.; Hung, R.; Fehlings, D. (28 September 2015). "Chronic Pain Assessment Tools for Cerebral Palsy: A Systematic Review". PEDIATRICS. 136 (4): e947–e960. doi:10.1542/peds.2015-0273.
- 1 2 Klingels, K.; De Cock, P.; Molenaers, G.; Desloovere, K.; Huenaerts, C.; Jaspers, E.; Feys, H. (2010). "Upper limb motor and sensory impairments in children with hemiplegic cerebral palsy. Can they be measured reliably?". Disability & Rehabilitation. 32 (5): 409–416. doi:10.3109/09638280903171469. PMID 20095955.
- 1 2 3 4 5 Donkervoort, M.; Roebroeck, M.; Wiegerink, D.; Van der Heijden-Maessen, H.; Stam, H.; The Transition Research Group South (2007). "Determinants of functioning of adolescents and young adults with cerebral palsy". Disability & Rehabilitation. 29 (6): 453–463. doi:10.1080/09638280600836018. PMID 17364800.
- ↑ Jones, KB; Wilson, B; Weedon, D; Bilder, D (December 2015). "Care of Adults With Intellectual and Developmental Disabilities: Cerebral Palsy.". FP essentials. 439: 26–30. PMID 26669212.
- ↑ Krigger, KW (1 January 2006). "Cerebral palsy: an overview.". American Family Physician. 73 (1): 91–100. PMID 16417071.
- 1 2 3 4 5 6 7 8 John Yarnell (2013). Epidemiology and Disease Prevention: A Global Approach (02 ed.). Oxford University Press. p. 190. ISBN 9780199660537.
- 1 2 3 4 Nelson KB, Blair E. (3 September 2015). "Prenatal Factors in Singletons with Cerebral Palsy Born at or near Term.". NEJM. 373 (10): 946–53. doi:10.1056/NEJMra1505261.
- ↑ William B. Carey, ed. (2009). Developmental-behavioral pediatrics (4th ed.). Philadelphia, PA: Saunders/Elsevier. p. 264. ISBN 9781416033707.
- ↑ Saunders, NR; Hellmann, J; Farine, D (Oct 2011). "Cerebral palsy and assisted conception.". Journal of Obstetrics and Gynaecology Canada. 33 (10): 1038–43. PMID 22014781.
- ↑ Hallman, M (Apr 2012). "Premature birth and diseases in premature infants: common genetic background?". The Journal of Maternal-Fetal & Neonatal Medicine. 25 Suppl 1: 21–4. doi:10.3109/14767058.2012.667600. PMID 22385349.
- ↑ Poets, CF; Wallwiener, D; Vetter, K (Oct 2012). "Risks associated with delivering infants 2 to 6 weeks before term--a review of recent data.". Deutsches Arzteblatt International. 109 (43): 721–6. doi:10.3238/arztebl.2012.0721. PMC 3498472
 . PMID 23181136.
. PMID 23181136. - ↑ McIntyre, S; Taitz, D; Keogh, J; Goldsmith, S; Badawi, N; Blair, E (Jun 2013). "A systematic review of risk factors for cerebral palsy in children born at term in developed countries.". Developmental Medicine & Child Neurology. 55 (6): 499–508. doi:10.1111/dmcn.12017. PMID 23181910.
- ↑ Ellenberg, JH; Nelson, KB (Mar 2013). "The association of cerebral palsy with birth asphyxia: a definitional quagmire.". Developmental Medicine & Child Neurology. 55 (3): 210–6. doi:10.1111/dmcn.12016. PMID 23121164.
- 1 2 3 "Cerebral Palsy: Hope Through Research". National Institute of Neurological Disorders and Stroke (U.S.). NIH Publication No. 13-159. August 2013. Retrieved 2014-01-23.
- ↑ Teng, J; Chang, T; Reyes, C; Nelson, KB (Oct 2012). "Placental weight and neurologic outcome in the infant: a review.". The Journal of Maternal-Fetal & Neonatal Medicine. 25 (10): 2082–7. doi:10.3109/14767058.2012.671871. PMID 22394270.
- ↑ Kieffer, Sara. "Cerebral Palsy | Johns Hopkins Pediatric Neurosurgery". Retrieved 2015-09-18.
- ↑ "Infection in the Newborn as a Cause of Cerebral Palsy, 12/2004". United Cerebral Palsy Research and Education Foundation (U.S.). Retrieved 2007-07-05.
- ↑ Bersani, I; Thomas, W; Speer, CP (Apr 2012). "Chorioamnionitis--the good or the evil for neonatal outcome?". The Journal of Maternal-Fetal & Neonatal Medicine. 25 Suppl 1: 12–6. doi:10.3109/14767058.2012.663161. PMID 22309119.
- ↑ Mwaniki, MK; Atieno, M; Lawn, JE; Newton, CR (Feb 4, 2012). "Long-term neurodevelopmental outcomes after intrauterine and neonatal insults: a systematic review.". Lancet. 379 (9814): 445–52. doi:10.1016/s0140-6736(11)61577-8. PMC 3273721
 . PMID 22244654.
. PMID 22244654. - ↑ Pharoah PO (December 2005). "Causal hypothesis for some congenital anomalies". Twin Res Hum Genet. 8 (6): 543–550. doi:10.1375/183242705774860141. PMID 16354495.
- 1 2 McIntyre, S; Morgan, C; Walker, K; Novak, I (Nov 2011). "Cerebral palsy--don't delay.". Developmental Disabilities Research Reviews. 17 (2): 114–29. doi:10.1002/ddrr.1106. PMID 23362031.
- 1 2 3 4 5 6 7 8 9 Bosanquet, M; Copeland, L; Ware, R; Boyd, R (May 2013). "A systematic review of tests to predict cerebral palsy in young children.". Developmental Medicine & Child Neurology. 55 (5): 418–26. doi:10.1111/dmcn.12140. PMID 23574478.
- ↑ Lungu, Codrin; Hirtz, Deborah; Damiano, Diane; Gross, Paul; Mink, Jonathan W. (20 September 2016). "Report of a workshop on research gaps in the treatment of cerebral palsy". Neurology (journal). 87 (12): 1293–1298. doi:10.1212/WNL.0000000000003116.
- ↑ Kolawole TM, Patel PJ, Mahdi AH (1989). "Computed tomographic (CT) scans in cerebral palsy (CP)". Pediatr Radiol. 20 (1–2): 23–27. doi:10.1007/BF02010628. PMID 2602010.
- ↑ Ashwal S, Russman BS, Blasco PA, et al. (2004). "Practice parameter: diagnostic assessment of the child with cerebral palsy: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society". Neurology. 62 (6): 851–63. doi:10.1212/01.WNL.0000117981.35364.1B. PMID 15037681.
- 1 2 3 Rethlefsen, SA.; Ryan, DD.; Kay, RM. (Oct 2010). "Classification systems in cerebral palsy.". Orthop Clin North Am. 41 (4): 457–67. doi:10.1016/j.ocl.2010.06.005. PMID 20868878.
- ↑ Trabacca, Antonio; Vespino, Teresa; Di Liddo, Antonella; Russo, Luigi (September 2016). "Multidisciplinary rehabilitation for patients with cerebral palsy: improving long-term care". Journal of Multidisciplinary Healthcare. 9: 455–462. doi:10.2147/JMDH.S88782.
- ↑ Stanley F, Blair E, Alberman E. Cerebral Palsies: Epidemiology and Causal Pathways. London, United Kingdom: MacKeith Press; 2000.
- ↑ http://www.emedicinehealth.com/cerebral_palsy/article_em.htm
- ↑ Doyle, LW; Crowther, CA; Middleton, P; Marret, S; Rouse, D (Jan 21, 2009). "Magnesium sulphate for women at risk of preterm birth for neuroprotection of the fetus.". The Cochrane Database of Systematic Reviews (1): CD004661. doi:10.1002/14651858.CD004661.pub3. PMID 19160238.
- ↑ Wolf, HT; Hegaard, HK; Greisen, G; Huusom, L; Hedegaard, M (Feb 2012). "Treatment with magnesium sulphate in pre-term birth: a systematic review and meta-analysis of observational studies.". Journal of Obstetrics and Gynaecology. 32 (2): 135–40. doi:10.3109/01443615.2011.638999. PMID 22296422.
- ↑ Nguyen, TM; Crowther, CA; Wilkinson, D; Bain, E (Feb 28, 2013). "Magnesium sulphate for women at term for neuroprotection of the fetus.". The Cochrane Database of Systematic Reviews. 2: CD009395. doi:10.1002/14651858.cd009395.pub2. PMID 23450601.
- ↑ Atik, Anzari; Harding, Richard; De Matteo, Robert; Kondos-Devcic, Delphi; Cheong, Jeanie; Doyle, Lex W.; Tolcos, Mary (January 2017). "Caffeine for apnea of prematurity: Effects on the developing brain". NeuroToxicology. 58: 94–102. doi:10.1016/j.neuro.2016.11.012.
- ↑ Jacobs, SE; Berg, M; Hunt, R; Tarnow-Mordi, WO; Inder, TE; Davis, PG (Jan 31, 2013). "Cooling for newborns with hypoxic ischaemic encephalopathy.". The Cochrane Database of Systematic Reviews. 1: CD003311. doi:10.1002/14651858.CD003311.pub3. PMID 23440789.
- 1 2 3 4 5 Novak, I; McIntyre, S; Morgan, C; Campbell, L; Dark, L; Morton, N; Stumbles, E; Wilson, SA; Goldsmith, S (Oct 2013). "A systematic review of interventions for children with cerebral palsy: state of the evidence.". Developmental Medicine & Child Neurology. 55 (10): 885–910. doi:10.1111/dmcn.12246. PMID 23962350.
- ↑ Pennington L, Goldbart J, Marshall J (2004). Pennington L, ed. "Speech and language therapy to improve the communication skills of children with cerebral palsy". Cochrane Database of Systematic Reviews (2): CD003466. doi:10.1002/14651858.CD003466.pub2. PMID 15106204.
- ↑ Dursun, E; Dursun, N; Alican, D (21 January 2004). "Effects of biofeedback treatment on gait in children with cerebral palsy.". Disability and Rehabilitation. 26 (2): 116–20. doi:10.1080/09638280310001629679. PMID 14668149.
- ↑ Macgregor R, Campbell R, Gladden MH, Tennant N, Young D (2007). "Effects of massage on the mechanical behaviour of muscles in adolescents with spastic diplegia: a pilot study". Developmental Medicine & Child Neurology. 49 (3): 187–191. doi:10.1111/j.1469-8749.2007.00187.x. PMID 17355474.
- ↑ Hansen, Ruth A.; Atchison, Ben (2000). Conditions in occupational therapy: effect on occupational performance. Hagerstown, MD: Lippincott Williams & Wilkins. ISBN 0-683-30417-8.
- ↑ Crepeau, Elizabeth Blesedell; Willard, Helen S.; Spackman, Clare S.; Neistadt, Maureen E. (1998). Willard and Spackman's occupational therapy. Philadelphia: Lippincott-Raven Publishers. ISBN 0-397-55192-4.
- ↑ Mulligan, Shelley (2003). Occupational therapy evaluation for children : a pocket guide. Philadelphia: Lippincott Williams & Wilkins. ISBN 9780781731638.
- 1 2 3 4 5 6 Hoare, BJ.; Wasiak, J.; Imms, C.; Carey, L. (2007). "Constraint-induced movement therapy in the treatment of the upper limb in children with hemiplegic cerebral palsy.". Cochrane Database Syst Rev (2): CD004149. doi:10.1002/14651858.CD004149.pub2. PMID 17443542.
- ↑ Clarke, Michael; Price, Katie (2012). "Augmentative and alternative communication for children with cerebral palsy". Pediatrics and Child Health. 22: 367–371. doi:10.1016/j.paed.2012.03.002.
- ↑ Heinen F, Desloovere K, Schroeder AS, et al. (January 2010). "The updated European Consensus 2009 on the use of Botulinum toxin for children with cerebral palsy". Eur. J. Paediatr. Neurol. 14 (1): 45–66. doi:10.1016/j.ejpn.2009.09.005. PMID 19914110.
- ↑ Apkon, SD; Cassidy, D (April 2010). "Safety considerations in the use of botulinum toxins in children with cerebral palsy.". PM&R. 2 (4): 282–4. doi:10.1016/j.pmrj.2010.02.006. PMID 20430330.
- ↑ Schejbalová A (2006). "[Derotational subtrochanteric osteotomy of the femur in cerebral palsy patients]". Acta Chirurgiae Orthopaedicae et Traumatologiae Cechoslovaca (in Czech). 73 (5): 334–9. PMID 17140515.
- ↑ Farmer JP, Sabbagh AJ (2007). "Selective dorsal rhizotomies in the treatment of spasticity related to cerebral palsy". Child's Nervous System. 23 (9): 991–1002. doi:10.1007/s00381-007-0398-2. PMID 17643249.
- ↑ Carraro, Elena; Zeme, Sergio; Ticcinelli, Valentina; Massaroni, Carlo; Santin, Michela; Peretta, Paola; Martinuzzi, Andrea; Trevisi, Enrico (November 2014). "Multidimensional outcome measure of selective dorsal rhizotomy in spastic cerebral palsy". European Journal of Paediatric Neurology. 18 (6): 704–713. doi:10.1016/j.ejpn.2014.06.003.
- 1 2 Gantasala, S.; Sullivan, PB.; Thomas, AG. (2013). "Gastrostomy feeding versus oral feeding alone for children with cerebral palsy.". Cochrane Database Syst Rev. 7: CD003943. doi:10.1002/14651858.CD003943.pub3. PMID 23900969.
- ↑ Condie DN, Meadows CB. Conclusions and recommendations. In: Condie DN, Meadows CB, eds. Report of a Consensus Conference on the Lower Limb Orthotic Management of Cerebral Palsy. Copenhagen: International Society of Prosthetics & Orthotics; 1995:15-19
- ↑ Ross, K; Bowers, R (2009). "A review of the effectiveness of lower limb orthoses used in cerebral palsy". Recent developments in healthcare for cerebral palsy : implications and opportunities for orthotics : report of an ISPO conference held at Wolfson College, Oxford, 8-11 September 2008. Copenhagen: International Society for Prosthetics and Orthotics (ISPO). pp. 235–297. ISBN 87-89809-28-9. PMC 2696330
 .
. - ↑ Balaban B, Yasar E, Dal U, Yazicioglu K, Mohur H, Kalyon TA (2007). "The effect of hinged ankle-foot orthosis on gait and energy expenditure in spastic hemiplegic cerebral palsy". Disability and Rehabilitation. 29 (2): 139–144. doi:10.1080/17483100600876740. PMID 17373095.
- ↑ McDonagh MS, Morgan D, Carson S, Russman BS (2007). "Systematic review of hyperbaric oxygen therapy for cerebral palsy: the state of the evidence". Dev Med Child Neurol. 49 (12): 942–947. doi:10.1111/j.1469-8749.2007.00942.x. PMID 18039243.
- ↑ American Academy of Pediatrics. Committee on Children with Disabilities (1999). "The treatment of neurologically impaired children using patterning". Pediatrics. 104 (5): 1149–1151. doi:10.1542/peds.104.5.1149. PMID 10545565.
- ↑ Alves-Pinto, Ana; Turova, Varvara; Blumenstein, Tobias; Lampe, Renée (2016). "The Case for Musical Instrument Training in Cerebral Palsy for Neurorehabilitation". Neural Plasticity. 2016: 1–9. doi:10.1155/2016/1072301.
- ↑ Bonnechère, B.; Jansen, B.; Omelina, L.; Degelaen, M.; Wermenbol, V.; Rooze, M.; Van Sint Jan, S. (August 2014). "Can serious games be incorporated with conventional treatment of children with cerebral palsy? A review". Research in Developmental Disabilities. 35 (8): 1899–1913. doi:10.1016/j.ridd.2014.04.016.
- ↑ Whalen, Cara N.; Case-Smith, Jane (29 November 2011). "Therapeutic Effects of Horseback Riding Therapy on Gross Motor Function in Children with Cerebral Palsy: A Systematic Review". Physical & Occupational Therapy In Pediatrics. 32 (3): 229–242. doi:10.3109/01942638.2011.619251.
- ↑ Tseng, Sung-Hui; Chen, Hung-Chou; Tam, Ka-Wai (26 May 2012). "Systematic review and meta-analysis of the effect of equine assisted activities and therapies on gross motor outcome in children with cerebral palsy". Disability and Rehabilitation. 35 (2): 89–99. doi:10.3109/09638288.2012.687033.
- ↑ Piškur, Barbara; Beurskens, Anna JHM; Jongmans, Marian J; Ketelaar, Marjolijn; Norton, Meghan; Frings, Christina A; Hemmingsson, Helena; Smeets, Rob JEM (8 November 2012). "Parents' actions, challenges, and needs while enabling participation of children with a physical disability: a scoping review". BMC Pediatrics. 12 (1). doi:10.1186/1471-2431-12-177.
- ↑ Jenks KM, de Moor J, van Lieshout EC, Maathuis KG, Keus I, Gorter JW (2007). "The effect of cerebral palsy on arithmetic accuracy is mediated by working memory, intelligence, early numeracy, and instruction time". Dev Neuropsychol. 32 (3): 861–79. doi:10.1080/87565640701539758. PMID 17956186.
- ↑ Hutton, Jane L. (June 2006). "Cerebral Palsy Life Expectancy". Clinics in Perinatology. 33 (2): 545–555. doi:10.1016/j.clp.2006.03.016. PMID 16765736.
- ↑ Strauss D, Brooks J, Rosenbloom R, Shavelle R (2008). "Life Expectancy in cerebral palsy: an update". Developmental Medicine & Child Neurology. 50 (7): 487–493. doi:10.1111/j.1469-8749.2008.03000.x. PMID 18611196.
- ↑ Wiegerink, Diana; Roebroeck, Marij; Bender, Jim; Stam, Henk; Cohen-Kettenis, Peggy & Transition Research Group South West Netherlands (June 2011). "Sexuality of Young Adults with Cerebral Palsy: Experienced Limitations and Needs". Sexuality and Disability. 29 (2): 119–128. doi:10.1007/s11195-010-9180-6.
- ↑ Van Zelst, B.; Miller, M.; Russo, R.; Murchland, S.; Crotty, M. (2006). "Activities of daily living in children with hemiplegic cerebral palsy: a cross-sectional evaluation using the Assessment of Motor and Process Skills". Developmental Medicine & Child Neurology. 48 (9): 723–7. doi:10.1017/S0012162206001551. PMID 16904017.
- ↑ Nieuwenhuijsen, C.; Donkervoort, M.; Nieuwstraten, W.; Stam, H.J.; Roebroeck, M.E.; Transition Research Group South West Netherlands (2009). "Experienced problems of young adults with cerebral palsy: targets for rehabilitation care". Archives of Physical Medicine and Rehabilitation. 90 (11): 1891–1897. doi:10.1016/j.apmr.2009.06.014. PMID 19887214.
- ↑ Arnould, C.; Penta, M.; Thonnard, J. (2008). "Hand impairments and their relationship with manual ability in children with cerebral palsy". Journal of Rehabilitation Medicine. 39 (9): 708–714. doi:10.2340/16501977-0111. PMID 17999009.
- ↑ Fedrizzi E, Pagliano E, Andreucci E, Oleari G (2003). "Hand function in children with hemiplegic cerebral palsy: prospective follow-up and functional outcome in adolescence". Dev. Med. Child Neurol. 45 (2): 85–91. doi:10.1017/s0012162203000173.
- ↑ Blesedell CE, Cohn ES, Schell AB. Willard and Spackman's occupational therapy. Philadelphia, PA: Lippincot, Williams and Wilkins; 2003. p. 705-709.
- 1 2 3 Townsend E, Stanton S, Law M. Enabling occupation: An occupational therapy perspective. Canadian Association of Occupational Therapists Ottawa, ON, Canada; 1997/2002. p. 34.
- ↑ Parham, LD; Primeau, LA (2008). "Play and occupational therapy". In Parham, LD; Fazio, LS. Play in occupational therapy for children (2nd ed.). St. Louis, Mo.: Mosby Elsevier. ISBN 978-0-323-02954-4.
- ↑ Miller, S; Reid, D (December 2003). "Doing play: competency, control, and expression.". Cyberpsychology & Behavior. 6 (6): 623–32. doi:10.1089/109493103322725397. PMID 14756927.
- ↑ Okimoto AM, Bundy A, Hanzlik J (2000). "Playfulness in children with and without disability: measurement and intervention". Am. J. Occup. Ther. 54 (1): 73–82. doi:10.5014/ajot.54.1.73.
- ↑ Hestenes LL, Carroll DE (2000). "The play interactions of young children with and without disabilities: Individual and environmental influences". Early Childhood Research Quarterly. 15 (2): 229–246. doi:10.1016/s0885-2006(00)00052-1.
- 1 2 3 4 Missiuna, C; Pollock N (October 1991). "Play deprivation in children with physical disabilities: the role of the occupational therapist in preventing secondary disability". Am. J. Occup. Ther. 45 (10): 882–8. doi:10.5014/ajot.45.10.882. PMID 1835302.
- ↑ Howard, L (December 1996). "A Comparison of Leisure-Time Activities between Able-Bodied Children and Children with Physical Disabilities". British Journal of Occupational Therapy. 59 (12): 570–574. doi:10.1177/030802269605901208.
- ↑ Rigby, Patricia; Schwellnus, Heidi (January 1999). "Occupational Therapy Decision Making Guidelines for Problems in Written Productivity". Physical & Occupational Therapy in Pediatrics. 19 (1): 5–27. doi:10.1080/J006v19n01_02.
- ↑ Smith M, Sandberg AD, Larsson M (2009). "Reading and spelling in children with severe speech and physical impairments: a comparative study". Int. J. Lang. Commun. Disord. 44 (6): 864–882. doi:10.1080/13682820802389873.
- 1 2 Pruitt, DW; Tsai, T (August 2009). "Common medical comorbidities associated with cerebral palsy.". Physical Medicine and Rehabilitation Clinics of North America. 20 (3): 453–67. doi:10.1016/j.pmr.2009.06.002. PMID 19643347.
- ↑ Cassidy, Tony (March 1996). "All work and no play: A focus on leisure time as a means for promoting health". Counselling Psychology Quarterly. 9 (1): 77–90. doi:10.1080/09515079608256354.
- 1 2 Reynolds, F (August 1997). "Coping with chronic illness and disability through creative needlecraft" (PDF). British Journal of Occupational Therapy. 60 (8): 352–356. doi:10.1177/030802269706000806.
- 1 2 King, G; Law, M; King, S; Rosenbaum, P; Kertoy, MK; Young, NL (2003). "A conceptual model of the factors affecting the recreation and leisure participation of children with disabilities.". Physical & Occupational Therapy in Pediatrics. 23 (1): 63–90. doi:10.1080/J006v23n01_05. PMID 12703385.
- 1 2 3 Aitchison, Cara (December 2003). "From leisure and disability to disability leisure: developing data, definitions and discourses". Disability & Society. 18 (7): 955–969. doi:10.1080/0968759032000127353.
- ↑ Imms, C (2008). "Children with cerebral palsy participate: a review of the literature.". Disability and Rehabilitation. 30 (24): 1867–84. doi:10.1080/09638280701673542. PMID 19037780.
- 1 2 3 Specht J, King G, Brown E, Foris C (2002). "The importance of leisure in the lives of persons with congenital physical disabilities". Am. J. Occup. Ther. 56 (4): 436–445. doi:10.5014/ajot.56.4.436.
- ↑ Frisch, Dana; Msall, Michael E. (August 2013). "Health, functioning, and participation of adolescents and adults with cerebral palsy: A review of outcomes research". Developmental Disabilities Research Reviews. 18 (1): 84–94. doi:10.1002/ddrr.1131.
- ↑ Turk, M.A., Overeynder, J.C., & Janicki, M.P. (Eds.). (1995). Uncertain Future - Aging and Cerebral Palsy: Clinical Concerns. Albany: New York State Developmental Disabilities Planning Council.
- ↑ Morgan, P.; McGinley, J. (17 April 2013). "Gait function and decline in adults with cerebral palsy: a systematic review". Disability and Rehabilitation. 36 (1): 1–9. doi:10.3109/09638288.2013.775359.
- ↑ Peterson, M. D.; Gordon, P. M.; Hurvitz, E. A. (February 2013). "Chronic disease risk among adults with cerebral palsy: the role of premature sarcopoenia, obesity and sedentary behaviour". Obesity Reviews. 14 (2): 171–182. doi:10.1111/j.1467-789X.2012.01052.x.
- ↑ Horsman, M.; Suto, M.; Dudgeon, B.; Harris, S. R. (22 February 2010). "Ageing with cerebral palsy: psychosocial issues". Age and Ageing. 39 (3): 294–299. doi:10.1093/ageing/afq018.
- ↑ Odding, E; Roebroeck, ME; Stam, HJ (28 February 2006). "The epidemiology of cerebral palsy: incidence, impairments and risk factors.". Disability and Rehabilitation. 28 (4): 183–91. doi:10.1080/09638280500158422. PMID 16467053.
- ↑ Johnson, Ann (2002). "Prevalence and characteristics of children with cerebral palsy in Europe". Developmental Medicine & Child Neurology. 44 (9): 633–40. doi:10.1017/S0012162201002675. PMID 12227618.
- ↑ Bax Martin; Goldstein Murray; Rosenbaum Peter; Leviton Alan; Paneth Nigel; Dan Bernard; Jacobsson Bo; Damiano Diane (2005). "Developmental Medicine & Child Neurology". Cambridge Journal. 47 (8): 571–76.
- ↑ Hirtz D, Thurman DJ, Gwinn-Hardy K, Mohamed M, Chaudhuri AR, Zalutsky R (2007). "How common are the "common" neurologic disorders?". Neurology. 68 (5): 326–337. doi:10.1212/01.wnl.0000252807.38124.a3. PMID 17261678.
- ↑ Australian Bureau of Statistics 2014, 3303.0 - Causes of Death, Australia - 1. Underlying causes of death (Australia), 2014, data cube: Excel spreadsheet, http://abs.gov.au/AUSSTATS/[email protected]/DetailsPage/3303.02014?OpenDocument
- ↑ Access Economics (2008) The Economic Impact of Cerebral Palsy in Australia in 2007. Access Economics, Canberra, ACT Archived November 21, 2008, at the Wayback Machine.
- ↑ Centers for Disease Control and Prevention (CDC) (2004). "Economic costs associated with mental retardation, cerebral palsy, hearing loss, and vision impairment—United States, 2003". MMWR Morb. Mortal. Wkly. Rep. 53 (3): 57–9. PMID 14749614. Retrieved 2007-08-12.
- ↑ "Medicaid.gov: Self Directed Services". Retrieved 2012-03-03.
- ↑ "Cerebral Palsy: a Guide for Care". Retrieved 2007-07-29.
- 1 2 Brindle, David (2002-05-22). "A very telling tale". The Guardian. London. Retrieved 2007-07-29.
- ↑ Zimmer, Benjamin (2007-02-05). "A brief history of "spaz"". Language Log. University of Pennsylvania. Retrieved 2007-07-29.
- ↑ Erickson, Glenn (May 22, 2007). "DVD Savant Review: Goodbye CP". DVDtalk.
- ↑ Borah, Prabalika (June 28, 2011). "Movie with a twist". The Hindu. Chennai, India.
- ↑ Alawadhi, Neha (March 26, 2011). "Charting unexplored territory in Indian cinema". The Hindu. Chennai, India.
- ↑ Rai, Ashutosh (Jan 3, 2012). "spandan" "Review of Marathi Film "SPANDAN"". The Newsleak.in.
- ↑ "Vegitha, carving a niche for herself". The Deccan Chronicle. June 7, 2011.
- ↑ Champlin, Charles (December 21, 1989). "My Left Foot". Los Angeles Times.
- ↑ "Being on Call the Midwife gave me the sense of being an actor in my own right". Retrieved August 12, 2015.
- ↑ "Josh Blue wins "Last Comic Standing"". Retrieved 2010-12-06.
- ↑ ""Last Comic Standing" biography". Retrieved 2010-12-06.
- ↑ "Jason Benetti Is Voice of Hope in Face of Cerebral Palsy". Archived from the original on 17 March 2013.
- ↑ "Britain's Got Talent: Jack Carroll to coin it with TV show, DVD and book deals," The Mirror, accessed August 1, 2013.
- ↑ "Abbey Curran, junior". St Ambrose University.
- ↑ "Abbey Curran Competing at the Miss USA Pageant!". Easter Seals. Peoria, Illinois.
- ↑ "Pageant Is Her Crowning Achievement: Miss Iowa, In Las Vegas For Miss USA Pageant, Was Born With Cerebral Palsy". Las Vegas: CBS. April 10, 2008. Retrieved 5 June 2010.
- ↑ "Comedian Geri Jewell headlines diversity event". WMU News. May 16, 2002. Retrieved 6 December 2010.
- ↑ Hartlaub, Peter (Jan 1, 2007). "No 2nd acts in film? Guess again". The San Francisco Chronicle. Associated Press. Retrieved 6 December 2010.
- ↑ Tim Lewis (7 August 2011). "Comedian Francesca Martinez: "Surely I can't be more unfunny than Patsy Palmer"". The Guardian. London.
- ↑ Saxon, Wolfgang (7 September 1997). "Harold E. Yuker, 73, Authority On Attitudes Toward Disabled". The New York Times. New York: NYTC. ISSN 0362-4331. Retrieved 30 July 2012.
- ↑ "O'Hanlon heads to Melbourne in career-best form". Athletics Australia. 1 March 2012. Retrieved 18 April 2012.
- ↑ "HarperCollins Publishers India Ltd.".
- ↑ Franks, Tim (2008-07-07). "Jerusalem Diary: Monday 7 July". BBC News.
- ↑ Heydarpour, Roja. "The Comic Is Palestinian, the Jokes Bawdy", The New York Times, November 21, 2006. Accessed March 22, 2011. "Ms. Zayid, who has a home in Cliffside Park, N.J., recently returned from Hollywood, where she lived while working on developing her one-woman show."
- ↑ IMEU | Institute for Middle East Understanding
- ↑ "Celebrity Ambassadors". Retrieved June 18, 2015.
- ↑ "REVIEW: Zach Anner's If at Birth You Don't Succeed Is an Inspiring, Hilarious Memoir About One Man's Life with Cerebral Palsy". People. March 11, 2016. Retrieved 16 March 2016.
- ↑ 10 Celebrities With Physical Deformities You May Have Never Noticed
- ↑ "THE THIRTEENTH NCPEDP - SHELL Helen Keller Awards" (PDF).
- ↑ "Celebrating Life against All odds". Press Information Bureau with inputs from the Ministry of Social Justice & Empowerment. National Informatics Centre (NIC). December 3, 2013.
- ↑ "IPC Athletics Records". www.paralympic.org. Retrieved 10 September 2012.
- ↑ "Paralympics 2012: Cockroft wins first GB track gold". BBC Sport. 31 August 2012. Retrieved 1 September 2012.
- ↑ "Paralympics 2012: Hannah Cockroft wins second sprint gold". BBC Sport. 6 September 2012. Retrieved 6 September 2012.
- ↑ Bennet, L; Tan, S; Van den Heuij, L; Derrick, M; Groenendaal, F; van Bel, F; Juul, S; Back, SA; Northington, F; Robertson, NJ; Mallard, C; Gunn, AJ (May 2012). "Cell therapy for neonatal hypoxia-ischemia and cerebral palsy.". Annals of Neurology. 71 (5): 589–600. doi:10.1002/ana.22670. PMID 22522476.
- ↑ Koy, A; Hellmich, M; Pauls, KAM; Marks, W; Lin, J-P; Fricke, O; Timmermann, L (May 2013). "Effects of deep brain stimulation in dyskinetic cerebral palsy: A meta-analysis.". Movement Disorders. 28 (5): 647–654. doi:10.1002/mds.25339.
External links
| Wikimedia Commons has media related to Cerebral palsy. |
| Look up cerebral palsy in Wiktionary, the free dictionary. |
_2008.jpg)