Graft-versus-host disease
| Graft-versus-host disease | |
|---|---|
| Classification and external resources | |
| Specialty | emergency medicine |
| ICD-10 | T86.0 |
| ICD-9-CM | 279.50 |
| DiseasesDB | 5388 |
| MedlinePlus | 001309 |
| eMedicine | med/926 ped/893 derm/478 |
| MeSH | D006086 |
Graft-versus-host disease (GvHD) is a medical complication following the receipt of transplanted tissue from a genetically different person. GvHD is commonly associated with stem cell transplant (bone marrow transplant), but the term also applies to other forms of tissue graft. Immune cells (white blood cells) in the donated tissue (the graft) recognize the recipient (the host) as foreign (nonself). The transplanted immune cells then attack the host's body cells. GvHD can also occur after a blood transfusion if the blood products used have not been irradiated or treated with an approved pathogen reduction system.
Whereas transplant rejection occurs when the host rejects the graft, GvHD occurs when the graft rejects the host. The underlying principle (alloimmunity) is the same, but the details and course may differ.
Signs and symptoms
In the classical sense, acute graft-versus-host-disease is characterized by selective damage to the liver, skin (rash), mucosa, and the gastrointestinal tract. Newer research indicates that other graft-versus-host-disease target organs include the immune system (the hematopoietic system, e.g., the bone marrow and the thymus) itself, and the lungs in the form of immune-mediated pneumonitis. Biomarkers can be used to identify specific causes of GvHD, such as elafin in the skin.[1] Chronic graft-versus-host-disease also attacks the above organs, but over its long-term course can also cause damage to the connective tissue and exocrine glands.
Acute GvHD of the GI tract can result in severe intestinal inflammation, sloughing of the mucosal membrane, severe diarrhea, abdominal pain, nausea, and vomiting. This is typically diagnosed via intestinal biopsy. Liver GvHD is measured by the bilirubin level in acute patients. Skin GvHD results in a diffuse red maculopapular rash, sometimes in a lacy pattern.
Mucosal damage to the vagina can result in severe pain and scarring, and appears in both acute and chronic GvHD. This can result in an inability to have sexual intercourse.[2]
Acute GvHD is staged as follows: overall grade (skin-liver-gut) with each organ staged individually from a low of 1 to a high of 4. Patients with grade IV GvHD usually have a poor prognosis. If the GvHD is severe and requires intense immunosuppression involving steroids and additional agents to get under control, the patient may develop severe infections as a result of the immunosuppression and may die of infection.
In the oral cavity, chronic graft-versus-host-disease manifests as lichen planus with a higher risk of malignant transformation to oral squamous cell carcinoma in comparison to the classical oral lichen planus. Graft-versus-host-disease-associated oral cancer may have more aggressive behavior with poorer prognosis, when compared to oral cancer in non-hematopoietic stem cell transplantation patients.[3]
Types
In the clinical setting, graft-versus-host-disease is divided into acute and chronic forms, and scored or graded on the basis of the tissue affected and the severity of the reaction.
- The acute or fulminant form of the disease (aGvHD) is normally observed within the first 100 days post-transplant,[6] and is a major challenge to transplants owing to associated morbidity and mortality.[7]
- The chronic form of graft-versus-host-disease (cGvHD) normally occurs after 100 days. The appearance of moderate to severe cases of cGVHD adversely influences long-term survival.[8]
Causes
Billingham Criteria: 3 criteria must be met in order for GvHD to occur.[2]
- An immuno-competent graft is administered, with viable and functional immune cells.
- The recipient is immunologically different from the donor - histo-incompatible.
- The recipient is immuno-compromised and therefore cannot destroy or inactivate the transplanted cells.
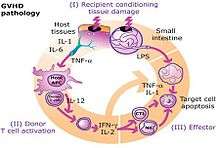
After bone marrow transplantation, T cells present in the graft, either as contaminants or intentionally introduced into the host, attack the tissues of the transplant recipient after perceiving host tissues as antigenically foreign. The T cells produce an excess of cytokines, including TNF-α and interferon-gamma (IFNγ). A wide range of host antigens can initiate graft-versus-host-disease, among them the human leukocyte antigens (HLA). However, graft-versus-host disease can occur even when HLA-identical siblings are the donors. HLA-identical siblings or HLA-identical unrelated donors often have genetically different proteins (called minor histocompatibility antigens) that can be presented by Major histocompatibility complex (MHC) molecules to the donor's T-cells, which see these antigens as foreign and so mount an immune response.[9]
Antigens most responsible for graft loss are HLA-DR (first six months), HLA-B (first two years), and HLA-A (long-term survival).[10]
While donor T-cells are undesirable as effector cells of graft-versus-host-disease, they are valuable for engraftment by preventing the recipient's residual immune system from rejecting the bone marrow graft (host-versus-graft). In addition, as bone marrow transplantation is frequently used to treat cancer, mainly leukemias, donor T-cells have proven to have a valuable graft-versus-tumor effect. A great deal of current research on allogeneic bone marrow transplantation involves attempts to separate the undesirable graft-vs-host-disease aspects of T-cell physiology from the desirable graft-versus-tumor effect.
Transfusion-associated GvHD
This type of GvHD is associated with transfusion of un-irradiated blood to immunocompromised recipients. It can also occur in situations in which the blood donor is homozygous and the recipient is heterozygous for an HLA haplotype. It is associated with higher mortality (80-90%) due to involvement of bone marrow lymphoid tissue, however the clinical manifestations are similar to GVHD resulting from bone marrow transplantation. Transfusion-associated GvHD is rare in modern medicine. It is almost entirely preventable by controlled irradiation of blood products to inactivate the white blood cells (including lymphocytes) within.[11]
Thymus transplantation
Thymus transplantation may be said to be able to cause a special type of GvHD because the recipient's thymocytes would use the donor thymus cells as models when going through the negative selection to recognize self-antigens, and could therefore still mistake own structures in the rest of the body for being non-self. This is a rather indirect GvHD because it is not directly cells in the graft itself that causes it but cells in the graft that make the recipient's T cells act like donor T cells. It can be seen as a multiple-organ autoimmunity in xenotransplantation experiments of the thymus between different species.[12] Autoimmune disease is a frequent complication after human allogeneic thymus transplantation, found in 42% of subjects over 1 year post transplantation.[13] However, this is partially explained by the fact that the indication itself, that is, complete DiGeorge syndrome, increases the risk of autoimmune disease.[14]
Prevention
- DNA-based tissue typing allows for more precise HLA matching between donors and transplant patients, which has been proven to reduce the incidence and severity of GvHD and to increase long-term survival.[15]
- The T-cells of umbilical cord blood (UCB) have an inherent immunological immaturity,[16] and the use of UCB stem cells in unrelated donor transplants has a reduced incidence and severity of GVHD.[17] The use of liver-derived hematopoietic stem cells to reconstitute bone marrow has the highest success rate according to recent studies.
- Methotrexate, cyclosporin and tacrolimus are common drugs used for GVHD prophylaxis.
- Graft-versus-host-disease can largely be avoided by performing a T-cell-depleted bone marrow transplant. However, these types of transplants come at a cost of diminished graft-versus-tumor effect, greater risk of engraftment failure, or cancer relapse,[18] and general immunodeficiency, resulting in a patient more susceptible to viral, bacterial, and fungal infection. In a multi-center study, disease-free survival at 3 years was not different between T cell-depleted and T cell-replete transplants.[19]
Treatment
Intravenously administered glucocorticoids, such as prednisone, are the standard of care in acute GvHD[7] and chronic GVHD.[20] The use of these glucocorticoids is designed to suppress the T-cell-mediated immune onslaught on the host tissues; however, in high doses, this immune-suppression raises the risk of infections and cancer relapse. Therefore, it is desirable to taper off the post-transplant high-level steroid doses to lower levels, at which point the appearance of mild GVHD may be welcome, especially in HLA mis-matched patients, as it is typically associated with a graft-versus-tumor effect.
Research
There are a large number of clinical trials either ongoing or recently completed in the investigation of graft-versus-host disease treatment and prevention.[21]
On May 17, 2012, Osiris Therapeutics announced that Canadian health regulators approved Prochymal, its drug for acute graft-versus host disease in children who have failed to respond to steroid treatment. Prochymal is the first stem cell drug to be approved for a systemic disease.[22]
In January 2016, Mesoblast released results of a Phase2 clinical trial on 241 children with acute Graft-versus-host disease, that was not responsive to steroids,.[23] Survival rate was 82% (vs 39% of controls) for those who showed some improvement after 1 month, and in the long term 72% (vs 18% of controls) for those that showed little effect after 1 month.
See also
References
- ↑ Paczesny, S.; Levine, J.E.; Hogan, J.; Crawford, J.; Braun, T.M.; Wang, H.; Faca, V.; Zhang, Q.; et al. (2009). "Elafin is a Biomarker of Graft Versus Host Disease of the Skin". Biology of Blood and Marrow Transplantation. 15 (2): 13–4. doi:10.1016/j.bbmt.2008.12.039.
- 1 2 Spiryda, L; Laufer, MR; Soiffer, RJ; Antin, JA (2003). "Graft-versus-host disease of the vulva and/or vagina: Diagnosis and treatment". Biology of Blood and Marrow Transplantation. 9 (12): 760–5. doi:10.1016/j.bbmt.2003.08.001. PMID 14677115.
- ↑ Elad, Sharon; Zadik, Yehuda; Zeevi, Itai; Miyazaki, Akihiro; De Figueiredo, Maria A. Z.; Or, Reuven (2010). "Oral Cancer in Patients After Hematopoietic Stem-Cell Transplantation: Long-Term Follow-Up Suggests an Increased Risk for Recurrence". Transplantation. 90 (11): 1243–4. doi:10.1097/TP.0b013e3181f9caaa. PMID 21119507.
- ↑ Martino R, Romero P, Subirá M, Bellido M, Altés A, Sureda A, Brunet S, Badell I, Cubells J, Sierra J (1999). "Comparison of the classic Glucksberg criteria and the IBMTR Severity Index for grading acute graft-versus-host disease following HLA-identical sibling stem cell transplantation. International Bone Marrow Transplant Registry". Bone Marrow Transplantation (journal). 24 (3): 283–287. doi:10.1038/sj.bmt.1701899. PMID 10455367.
- ↑ Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, Martin P, Chien J, Przepiorka D, Couriel D, Cowen EW, Dinndorf P, Farrell A, Hartzman R, Henslee-Downey J, Jacobsohn D, McDonald G, Mittleman B, Rizzo JD, Robinson M, Schubert M, Schultz K, Shulman H, Turner M, Vogelsang G, Flowers ME (2005). "National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report". BIOLOGY OF BLOOD AND MARROW TRANSPLANTATION. 11 (12): 945–956. doi:10.1016/j.bbmt.2005.09.004. PMID 16338616.
- ↑ "Improved Management of Graft-Versus-Host Disease". National Marrow Donor Program.
- 1 2 Goker, H; Haznedaroglu, IC; Chao, NJ (2001). "Acute graft-vs-host disease Pathobiology and management". Experimental Hematology. 29 (3): 259–77. doi:10.1016/S0301-472X(00)00677-9. PMID 11274753.
- ↑ Lee, Stephanie J.; Vogelsang, Georgia; Flowers, Mary E.D. (2003). "Chronic graft-versus-host disease". Biology of Blood and Marrow Transplantation. 9 (4): 215–33. doi:10.1053/bbmt.2003.50026. PMID 12720215.
- ↑ Taylor CJ, Bolton EM, Bradley JA (2011). "Immunological considerations for embryonic and induced pluripotent stem cell banking". Philosophical Transactions of the Royal Society B. 366 (1575): 2312–2322. doi:10.1098/rstb.2011.0030. PMC 3130422
 . PMID 21727137.
. PMID 21727137. - ↑ Solomon S, Pitossi F, Rao MS (2015). "Banking on iPSC--is it doable and is it worthwhile". STEM CELL REVIEWS. 11 (1): 1–10. doi:10.1007/s12015-014-9574-4. PMC 4333229
 . PMID 25516409.
. PMID 25516409. - ↑ Moroff, G; Leitman, SF; Luban, NL (1997). "Principles of blood irradiation, dose validation, and quality control". Transfusion. 37 (10): 1084–92. doi:10.1046/j.1537-2995.1997.371098016450.x. PMID 9354830.
- ↑ Xia, G; Goebels, J; Rutgeerts, O; Vandeputte, M; Waer, M (2001). "Transplantation tolerance and autoimmunity after xenogeneic thymus transplantation". Journal of immunology. 166 (3): 1843–54. doi:10.4049/jimmunol.166.3.1843. PMID 11160231.
- ↑ Markert, M. Louise; Devlin, Blythe H.; McCarthy, Elizabeth A.; Chinn, Ivan K.; Hale, Laura P. (2008). "Thymus Transplantation". In Lavini, Corrado; Moran, Cesar A.; Morandi, Uliano; et al. Thymus Gland Pathology: Clinical, Diagnostic, and Therapeutic Features. pp. 255–67. doi:10.1007/978-88-470-0828-1_30. ISBN 978-88-470-0827-4.
- ↑ Markert, M. L.; Devlin, B. H.; Alexieff, M. J.; Li, J.; McCarthy, E. A.; Gupton, S. E.; Chinn, I. K.; Hale, L. P.; et al. (2007). "Review of 54 patients with complete DiGeorge anomaly enrolled in protocols for thymus transplantation: Outcome of 44 consecutive transplants". Blood. 109 (10): 4539–47. doi:10.1182/blood-2006-10-048652. PMC 1885498
 . PMID 17284531.
. PMID 17284531. - ↑ Morishima, Y.; Sasazuki, T; Inoko, H; Juji, T; Akaza, T; Yamamoto, K; Ishikawa, Y; Kato, S; et al. (2002). "The clinical significance of human leukocyte antigen (HLA) allele compatibility in patients receiving a marrow transplant from serologically HLA-A, HLA-B, and HLA-DR matched unrelated donors". Blood. 99 (11): 4200–6. doi:10.1182/blood.V99.11.4200. PMID 12010826.
- ↑ Grewal, S. S.; Barker, JN; Davies, SM; Wagner, JE (2003). "Unrelated donor hematopoietic cell transplantation: Marrow or umbilical cord blood?". Blood. 101 (11): 4233–44. doi:10.1182/blood-2002-08-2510. PMID 12522002.
- ↑ Laughlin, Mary J.; Barker, Juliet; Bambach, Barbara; Koc, Omer N.; Rizzieri, David A.; Wagner, John E.; Gerson, Stanton L.; Lazarus, Hillard M.; et al. (2001). "Hematopoietic Engraftment and Survival in Adult Recipients of Umbilical-Cord Blood from Unrelated Donors". New England Journal of Medicine. 344 (24): 1815–22. doi:10.1056/NEJM200106143442402. PMID 11407342.
- ↑ Hale, G; Waldmann, H (1994). "Control of graft-versus-host disease and graft rejection by T cell depletion of donor and recipient with Campath-1 antibodies. Results of matched sibling transplants for malignant diseases". Bone marrow transplantation. 13 (5): 597–611. PMID 8054913.
- ↑ Wagner, John E; Thompson, John S; Carter, Shelly L; Kernan, Nancy A; Unrelated Donor Marrow Transplantation Trial (2005). "Effect of graft-versus-host disease prophylaxis on 3-year disease-free survival in recipients of unrelated donor bone marrow (T-cell Depletion Trial): A multi-centre, randomised phase II–III trial". The Lancet. 366 (9487): 733–41. doi:10.1016/S0140-6736(05)66996-6. PMID 16125590.
- ↑ Menillo, S A; Goldberg, S L; McKiernan, P; Pecora, A L (2001). "Intraoral psoralen ultraviolet a irradiation (PUVA) treatment of refractory oral chronic graft-versus-host disease following allogeneic stem cell transplantation". Bone Marrow Transplantation. 28 (8): 807–8. doi:10.1038/sj.bmt.1703231. PMID 11781637.
- ↑ search of clinicaltrials.gov for Graft-versus-host disease
- ↑ "World's First Stem-Cell Drug Approval Achieved in Canada". The National Law Review. Drinker Biddle & Reath LLP. 2012-06-12. Retrieved 2012-07-01.
- ↑ "Increased Survival Using MSB Cells In Children With aGVHD". Retrieved 22 Feb 2016.
Further reading
- Ferrara JLM, Deeg HJ, Burakoff SJ. Graft-Vs.-Host Disease: Immunology, Pathophysiology, and Treatment. Marcel Dekker, 1990 ISBN 0-8247-9728-0
- Polsdorfer, JR Gale Encyclopedia of Medicine: Graft-vs.-host disease