Pancreatic islets
| Pancreatic islets | |
|---|---|
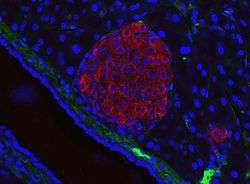 Pancreatic islet (mouse) in its typical proximity to a blood vessel; insulin in red, nuclei in blue. | |
 | |
| Details | |
| Identifiers | |
| Latin | insulae pancreaticae |
| TA | A05.9.01.019 |
| FMA | 16016 76489, 16016 |
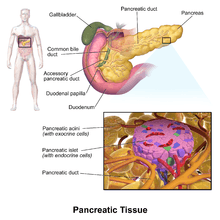
The pancreatic islets or islets of Langerhans are the regions of the pancreas that contain its endocrine (i.e., hormone-producing) cells, discovered in 1869 by German pathological anatomist Paul Langerhans.[2] New studies show that the pancreatic islets constitute approximately 4.5% of the pancreas volume[1] and receive 10-15% of its blood flow.[3] The islets of Langerhans are arranged in density routes throughout the healthy human pancreas.[1]
Structure
There are about 3 million islets distributed in the form of density routes throughout the pancreas of a healthy adult human,[1] each of which measures an average of about 0.1 mm (109 µm) in diameter.[1][4]:914 Each is separated from the surrounding pancreatic tissue by a thin fibrous connective tissue capsule which is continuous with the fibrous connective tissue that is interwoven throughout the rest of the pancreas.[4]:914 The combined mass of the islets is 2 grams.[1] Islets of Langerhans can also form super structures called Islet clusters which are composed of small islets that surround large blood vessels.[1] The roundness of islets along the pancreas has also been quantified through the Index of Sphericity method.[1] Thus, the islets closest to the spherical form are mainly found in the pancreas tail whereas the islets farthest from the spherical form are found in the pancreas neck.[1]
Histology
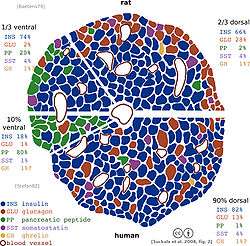
Hormones produced in the pancreatic islets are secreted directly into the blood flow by (at least) five types of cells. In rat islets, endocrine cell subsets are distributed as follows:[5]
- Alpha cells producing glucagon (20% of total islet cells)
- Beta cells producing insulin and amylin (≈70%)
- Delta cells producing somatostatin (<10%)
- PP cells (gamma cells) producing pancreatic polypeptide (<5%)
- Epsilon cells producing ghrelin (<1%)
It has been recognized that the cytoarchitecture of pancreatic islets differs between species.[6][7][8] In particular, while rodent islets are characterized by a predominant proportion of insulin-producing beta cells in the core of the cluster and by scarce alpha, delta and PP cells in the periphery, human islets display alpha and beta cells in close relationship with each other throughout the cluster.[6][8]
Islets can influence each other through paracrine and autocrine communication, and beta cells are coupled electrically to other beta cells (but not to other cell types).
Function
The paracrine feedback system of the pancreatic islets has the following structure:[9]
- Glucose/Insulin: activates beta cells and inhibits alpha cells
- Glycogen/Glucagon: activates alpha cells which activates beta cells and delta cells
- Somatostatin: inhibits alpha cells and beta cells
A large number of G protein-coupled receptors (GPCRs) regulate the secretion of insulin, glucagon and somatostatin from pancreatic islets,[10] and some of these GPCRs are the targets of drugs used to treat type-2 diabetes (ref GLP-1 receptor agonists, DPPIV inhibitors).b
Electrical activity
Electrical activity of pancreatic islets has been studied using patch clamp techniques. It has turned out that the behavior of cells in intact islets differs significantly from the behavior of dispersed cells.[11]
Clinical significance
Diabetes
The beta cells of the pancreatic islets secrete insulin, and so play a significant role in diabetes. It is thought that they are destroyed by immune assaults. However, there are also indications that beta cells have not been destroyed but have only become non-functional.
Transplantation
Restoration of metabolic control via the transplantation of pancreatic islets is an appealing approach. This can be achieved by transplantation of the pancreas as vascularized organ or of isolated pancreatic islets (islet cell clusters). Islet transplantation has emerged as a viable option for the treatment of insulin requiring diabetes in the early 1970s with steady progress over the last three decades.[12]
Islet transplantation has the possibility of restoring beta cell function from diabetes, offering an alternative to a complete pancreas transplantation or an artificial pancreas.
Because the beta cells in the pancreatic islets are selectively destroyed by an autoimmune process in type 1 diabetes, clinicians and researchers are actively pursuing islet transplantation as a means of restoring physiological beta cell function in patients with type 1 diabetes.[13][14]
Recent clinical trials have shown that insulin independence and improved metabolic control can be reproducibly obtained after transplantation of cadaveric donor islets into patients with unstable type 1 diabetes.[14]
Islet transplantation for type 1 diabetes currently requires potent immunosuppression to prevent host rejection of donor islets.[15]
An alternative source of beta cells, such insulin-producing cells derived from adult stem cells or progenitor cells would contribute to overcoming the shortage of donor organs for transplantation. The field of regenerative medicine is rapidly evolving and offers great hope for the nearest future. However, type 1 diabetes is the result of the autoimmune destruction of beta cells in the pancreas. Therefore, an effective cure will require a sequential, integrated approach that combines adequate and safe immune interventions with beta cell regenerative approaches.[16]
Another potential source of beta cells may be xenotransplantation. The most likely source for xenogeneic islets for transplantation into human under evaluation is the pig pancreas. Interestingly, human and porcine insulin differ only for one amino acid, and insulin extracted from porcine pancreata has been used for the treatment of patients with diabetes before the development of recombinant human insulin technology. Several studies in small and large animals models have shown that transplantation of islet cells across species is possible. However, several problems need to be overcome for porcine islet transplantation to become a viable clinical option. The immunogenicity of xenogeneic tissues may be different from and even stronger than allogeneic tissues. For instance, Galalpha1-3Galbeta1-4GlcNAc (alpha galactosidase, alpha-Gal) expressed on porcine cells represents a major barrier to xenotransplantation being the target of preformed antibodies present in human blood.
Remarkable progress has been recorded in the development of genetically modified pigs lacking or overexpressing molecules that may improve acceptance of transplanted tissues across into humans. Pigs lacking alpha-Gal or overexpressing human decay accelerating factor (hDAF), amongst others, have been generated to study the impact on transplanted outcome in nonhuman primate models. Another possible antigenic target is the Hanganutziu-Deichter antigen, a sialic acid found in pigs and not humans, which may contribute to immunogenicity of porcine islets. Another limitation is the risk for transmission of zoonotic infections from pigs to humans, particularly from porcine endogenous retro-viruses (PERV). Amongst the approaches proposed to overcome islet xenorejection is immunoisolation of the clusters using encapsulation techniques that may shield them from immune attack. Studies in rodents and large animals have shown great promise that justify cautious optimism for the near future. Nonrandomized, uncontrolled pilot clinical trials are ongoing in subject with insulin-requiring diabetes to test the efficacy of encapsulation techniques to protect xenogeneic islets in the absence of chronic anti-rejection drugs.
Additional images
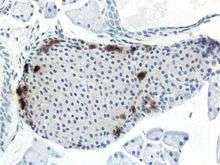 Mouse islet immunostained for pancreatic polypeptide
Mouse islet immunostained for pancreatic polypeptide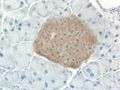 Mouse islet immunostained for insulin
Mouse islet immunostained for insulin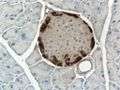 Mouse islet immunostained for glucagon
Mouse islet immunostained for glucagon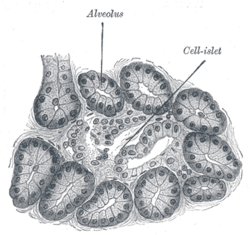 Illustration of dog pancreas. 250x.
Illustration of dog pancreas. 250x.
See also
References
- 1 2 3 4 5 6 7 8 9 Ionescu-Tirgoviste, Constantin; Gagniuc, Paul A.; Gubceac, Elvira; Mardare, Liliana; Popescu, Irinel; Dima, Simona; Militaru, Manuella (2015-09-29). "A 3D map of the islet routes throughout the healthy human pancreas". Scientific Reports. 5. doi:10.1038/srep14634. PMC 4586491
 . PMID 26417671.
. PMID 26417671. - ↑ Langerhans P (1869). "Beitrage zur mikroscopischen anatomie der bauchspeichel druse". Inaugural-dissertation. Berlin: Gustav Lange.
- ↑ Functional Anatomy of the Endocrine Pancreas
- 1 2 Sleisenger, edited by Mark Feldman, Lawrence S. Friedman, Lawrence J. Brandt; consulting editor, Marvin H. (2009). Sleisenger & Fordtran's gastrointestinal and liver disease pathophysiology, diagnosis, management (9th ed.). St. Louis, Mo.: MD Consult. ISBN 978-1-4160-6189-2.
- ↑ Elayat AA; el-Naggar MM; Tahir M; Bassam dahrouj (1995). "An immunocytochemical and morphometric study of the rat pancreatic islets". Journal of Anatomy. 186. (Pt 3) (Pt 3): 629–37. PMC 1167020
 . PMID 7559135.
. PMID 7559135. - 1 2 Brissova M, Fowler MJ, Nicholson WE, Chu A, Hirshberg B, Harlan DM, Powers AC (2005). "Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy". Journal of Histochemistry and Cytochemistry. 53 (9): 1087–97. doi:10.1369/jhc.5C6684.2005. PMID 15923354.
- ↑ Ichii H, Inverardi L, Pileggi A, Molano RD, Cabrera O, Caicedo A, Messinger S, Kuroda Y, Berggren PO, Ricordi C (2005). "A novel method for the assessment of cellular composition and beta-cell viability in human islet preparations". American Journal of Transplantation. 5 (7): 1635–45. doi:10.1111/j.1600-6143.2005.00913.x. PMID 15943621.
- 1 2 Cabrera O, Berman DM, Kenyon NS, Ricordi C, Berggren PO, Caicedo A (2006). "The unique cytoarchitecture of human pancreatic islets has implications for islet cell function". Proceedings of the National Academy of Sciences of the United States of America. 103 (7): 2334–9. doi:10.1073/pnas.0510790103. ISSN 1091-6490. PMC 1413730
 . PMID 16461897.
. PMID 16461897. - ↑ Wang, Michael B.; Bullock, John; Boyle, Joseph R. (2001). Physiology. Hagerstown, MD: Lippincott Williams & Wilkins. p. 391. ISBN 0-683-30603-0.
- ↑ "An atlas and functional analysis of G-protein coupled receptors in human islets of Langerhans.Amisten S, Salehi A, Rorsman P, Jones PM, Persaud SJ., Pharmacol Ther. 2013 May 18. PMID 23694765
- ↑ Pérez-Armendariz M, Roy C, Spray DC, Bennett MV (1991). "Biophysical properties of gap junctions between freshly dispersed pairs of mouse pancreatic beta cells". Biophysical Journal. 59 (1): 76–92. doi:10.1016/S0006-3495(91)82200-7. PMC 1281120
 . PMID 2015391.
. PMID 2015391. - ↑ Piemonti L, Pileggi A (2013). "25 Years of the Ricordi Automated Method for Islet Isolation". CellR4. 1 (1): 8–22.
- ↑ Meloche RM (2007). "Transplantation for the treatment of type 1 diabetes". World Journal of Gastroenterology. 13 (47): 6347–55. doi:10.3748/wjg.13.6347. PMID 18081223.
- 1 2 Hogan A, Pileggi A, Ricordi C (2008). "Transplantation: current developments and future directions; the future of clinical islet transplantation as a cure for diabetes". Frontiers in Bioscience. 13 (13): 1192–205. doi:10.2741/2755. PMID 17981623.
- ↑ Chatenoud L (2008). "Chemical immunosuppression in islet transplantation—friend or foe?". New England Journal of Medicine. 358 (11): 1192–3. doi:10.1056/NEJMcibr0708067. ISSN 0028-4793. PMID 18337609.
- ↑ Pileggi A, Cobianchi L, Inverardi L, Ricordi C (2006). "Overcoming the challenges now limiting islet transplantation: a sequential, integrated approach". Annals of the New York Academy of Sciences. 1079 (1): 383–98. doi:10.1196/annals.1375.059. ISSN 0077-8923. PMID 17130583.
External links
- "The Islets of Langerhans", Karolinska Institutet, Sweden
- "Islets"
- Islet Society
- MeSH A03.734.414
- "Pancreas, human – H&E", Blue Histology – Accessory Digestive Glands, School of Anatomy and Human Biology,