NS5B
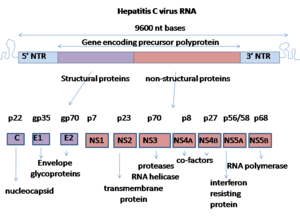
Nonstructural protein 5B (NS5B) is a viral protein found in the hepatitis C virus (HCV).[1] It has the key function of replicating the HCV's viral RNA by using the viral positive RNA strand as its template and catalyzes the polymerization of ribonucleoside triphosphates (rNTP) during RNA replication.[2][3][4] Several crystal structures of NS5B polymerase in several crystalline forms have been determined based on the same consensus sequence BK (HCV-BK, genotype 1).[5] The structure can be represented by a right hand shape with fingers, palm, and thumb. The encircled active site, unique to NS5B, is contained within the palm structure of the protein. Recent studies on NS5B protein genotype 1b strain J4’s (HC-J4) structure indicate a presence of an active site where possible control of nucleotide binding occurs and initiation of de-novo RNA synthesis. De-novo adds necessary primers for initiation of RNA replication.[6] Current research attempts to bind structures to this active site to alter its functionality in order to prevent further viral RNA replication.[7]
Drugs targeting NS5B
- Beclabuvir, currently in clinical trials
- Dasabuvir (Viekira Pak), non-nucleoside/nucleotide analog, approved by FDA in December 2014 (only in combination with ombitasvir, paritaprevir, and ritonavir)
- Deleobuvir, development terminated
- Filibuvir, development terminated
- Radalbuvir, currently in clinical trials
- Setrobuvir, development terminated
- Sofosbuvir (Sovaldi), nucleotide analog, approved by the FDA in December 2013
References
- ↑ Gehring S, Gregory SH, Wintermeyer P, Aloman C, Wands JR (December 2008). "Generation of Immune Responses Against HCV Using Dendritic Cells Containing NS5 Protein-Coated Microparticles". Clin. Vaccine Immunol. 16 (2): 163–71. doi:10.1128/CVI.00287-08. PMC 2643538
 . PMID 19091993.
. PMID 19091993. - ↑ Jin, Z; Leveque, V; Ma, H; Johnson, K. A.; Klumpp, K (2012). "Assembly, purification, and pre-steady-state kinetic analysis of active RNA-dependent RNA polymerase elongation complex". Journal of Biological Chemistry. 287 (13): 10674–83. doi:10.1074/jbc.M111.325530. PMC 3323022
 . PMID 22303022.
. PMID 22303022. - ↑ Moradpour, D; Penin, F; Rice, CM (2007). "Replication of hepatitis C virus". Nature reviews. Microbiology. 5 (6): 453–63. doi:10.1038/nrmicro1645. PMID 17487147.
- ↑ Rigat, K.; Wang, Y.; Hudyma, T. W.; Ding, M.; Zheng, X.; Gentles, R. G.; Beno, B. R.; Gao, M.; Roberts, S. B. (2010). "Ligand-induced changes in hepatitis C virus NS5B polymerase structure". Antiviral Research. 88 (2): 197–206. doi:10.1016/j.antiviral.2010.08.014. PMID 20813137.
- ↑ Biswal, B. K.; Cherney, M. M.; Wang, M.; Chan, L.; Yannopoulos, C. G.; Bilimoria, D.; Nicolas, O.; Bedard, J.; James, M. N. (2005). "Crystal structures of the RNA-dependent RNA polymerase genotype 2a of hepatitis C virus reveal two conformations and suggest mechanisms of inhibition by non-nucleoside inhibitors". The Journal of Biological Chemistry. 280 (18): 18202–18210. doi:10.1074/jbc.M413410200. PMID 15746101.
- ↑ O'Farrell, D; Trowbridge, R; Rowlands, D; Jäger, J (2003). "Substrate complexes of hepatitis C virus RNA polymerase (HC-J4): Structural evidence for nucleotide import and de-novo initiation". Journal of Molecular Biology. 326 (4): 1025–35. doi:10.1016/s0022-2836(02)01439-0. PMID 12589751.
- ↑ Biswal, BK; Wang, M; Cherney, MM; Chan, L; Yannopoulos, CG; Bilimoria, D; Bedard, J; James, MN (2006). "Non-nucleoside inhibitors binding to hepatitis C virus NS5B polymerase reveal a novel mechanism of inhibition". Journal of Molecular Biology. 361 (1): 33–45. doi:10.1016/j.jmb.2006.05.074. PMID 16828488.