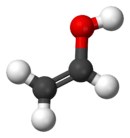
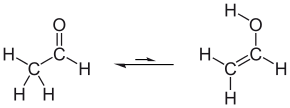Vinyl alcohol
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Ethenol | |||
| Other names
Hydroxyethene Hydroxyethylene | |||
| Identifiers | |||
| 557-75-5 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEMBL | ChEMBL76101 | ||
| ChemSpider | 10726 | ||
| ECHA InfoCard | 100.008.350 | ||
| PubChem | 11199 | ||
| |||
| |||
| Properties | |||
| C2H4O | |||
| Molar mass | 44.053 g/mol | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
Vinyl alcohol, also called ethenol (IUPAC name), is an alcohol. It is not to be confused with the drinking alcohol, ethanol. With the formula CH2CHOH, vinyl alcohol is an isomer of acetaldehyde and ethylene oxide.
Tautomerization of acetaldehyde to vinyl alcohol
Under normal conditions, vinyl alcohol converts (tautomerizes) to acetaldehyde:
At room temperature, acetaldehyde (H3CC(O)H) is more stable than vinyl alcohol (H2C=CHOH) by 42.7 kJ/mol:[1]
- H2C=CHOH → H3CC(O)H ΔH298,g = −42.7 kJ/mol
This keto-enol tautomerization has a high barrier and does therefore not occur near room temperature. This tautomerization can be catalyzed via photochemical process. These findings suggest that the keto-enol tautomerization is a viable route under atmospheric and stratospheric conditions, relevant to a role for vinyl alcohol in the production of organic acids in the atmosphere.[2][3]
Relationship to polyvinylalcohol
Because of the instability of vinyl alcohol, the thermoplastic polyvinyl alcohol (PVA or PVOH) is made indirectly by polymerization of vinyl acetate followed by hydrolysis of the ester bonds (Ac = acetyl, HOAc = acetic acid):
- n CH2=CHOAc → (CH2−CHOAc)n
- (CH2−CHOAc)n + n H2O → (CH2−CHOH)n + n HOAc
As a ligand
Several metal complexes are known that contain vinyl alcohol as a ligand. One example is Pt(acac)(η2-C2H3OH)Cl.[4]
Occurrence in interstellar medium
Vinyl alcohol was discovered in the molecular cloud Sagittarius B using the 12-meter radio telescope at the Kitt Peak National Observatory.[5] This vinyl alcohol being stable in the (dilute) interstellar medium shows that its tautomerization is not unimolecular.
See also
References
- ↑ R.D. Johnson III. "CCCBDB NIST Standard Reference Database.". Retrieved 2014-08-30.
- ↑ Heazlewood, B. R.; Maccarone, A. T.; Andrews, D. U.; Osborn, D. L.; Harding, L. B.; Klippenstein, S. J.; Jordan, M. J. T.; Kable, S. H. "Near-threshold H/D exchange in CD3CHO photodissociation." Nat. Chem. 2011, 3, 443−448. doi:10.1038/nchem.1052
- ↑ Andrews, D. U.; Heazlewood, B. R.; Maccarone, A. T.; Conroy, T.; Payne, R. J.; Jordan, M. J. T.; Kable, S. H. "Photo-tautomerization of acetaldehyde to vinyl alcohol: A potential route to tropospheric acids." Science 2012, 337, 1203−1206. doi:10.1126/science.1220712
- ↑ F. A. Cotton, J. N. Francis, B. A. Frenz, M. Tsutsui "Structure of a dihapto(vinyl alcohol) complex of platinum(II)" Journal of the American Chemical Society, 1973, volume 95, p. 2483-6. doi:10.1021/ja00789a011
- ↑ "Scientists Toast the Discovery of Vinyl Alcohol in Interstellar Space". National Radio Astronomy Observatory. 2001-10-01. Retrieved 2006-12-20.