Novichok agent
| Part of a series on | |||||
| Chemical agents | |||||
|---|---|---|---|---|---|
| Lethal agents | |||||
|
|||||
| Incapacitating agents | |||||
|
|||||
Novichok (Russian: новичок meaning "newcomer") is a series of nerve agents that were developed by the Soviet Union in the 1970s and 1980s.[1] Allegedly these are the most deadly nerve agents ever made, with some variants possibly five to eight times more potent than VX, though this has never been proven.[2][3] They belong to "third generation chemical weapons" designed as a part of Soviet "Foliant" program.[4] Initially designated K-84 and later renamed A-230, the Novichok family of analogs comprises more than a hundred structural variants.[5] Of all the variants the most promising, from a military standpoint, was A-232 (Novichok-5).[6]
Design objectives
These agents were designed to achieve three objectives:
- To be undetectable using standard NATO chemical detection equipment;
- To defeat NATO chemical protective gear;
- To be safer to handle.
Although all these objectives were claimed to have been achieved, this is yet to be proven.
Some of these agents are binary weapons, in which precursors for the nerve agents are mixed in a munition to produce the agent just prior to its use. Because the precursors are generally significantly less hazardous than the agents themselves, this technique makes handling and transporting the munitions a great deal simpler. Additionally, precursors to the agents are usually much easier to stabilize than the agents themselves, so this technique also made it possible to increase the shelf life of the agents. However this has the disadvantage that careless preparation or preparation by untrained individuals may produce a non-optimal agent. During the 1980s and 1990s, binary versions of several Soviet agents were developed and are designated as "Novichok" agents.
Disclosure
Extremely potent third-generation chemical weapons were developed in the Soviet Union and Russia from the 1970s until the early 1990s, according to a publication by two chemists, Lev Fedorov and Vil Mirzayanov in Moskovskiye Novosti weekly in 1992.[7] The publication appeared just on the eve of Russia's signing of the Chemical Weapons Convention. According to Mirzayanov, the Russian Military Chemical Complex (MCC) was using defense conversion money received from the West for development of a chemical warfare facility.[2][3] Mirzayanov made his disclosure out of environmental concerns. He was a head of a counter-intelligence department and performed measurements outside the chemical weapons facilities to make sure that foreign spies could not detect any traces of production. To his horror, the levels of deadly substances were 80 times greater than the maximum safe concentration.[3] (A full account by Mirzayanov is available online.[8])
The existence of Novichok agents was openly admitted by Russian military industrial complex authorities when they brought a treason case against Mirzayanov. According to expert witness testimonies prepared for the KGB by three scientists, novichok and other related chemical agents had indeed been produced and therefore the disclosure by Mirzayanov represented high treason.[9]
Vil Mirzayanov was arrested on October 22, 1992 and sent to Lefortovo prison for divulging state secrets. He was released later because "not one of the formulas or names of poisonous substances in the Moscow News article was new to the Soviet press, nor were locations ... of testing sites revealed."[3] According to Yevgenia Albats, "the real state secret revealed by Fyodorov and Mirzayanov was that generals had lied — and were still lying — to both the international community and their fellow citizens."[3] He now lives in the U.S.[10]
Description of Novichok agents
The first description of these agents was provided by Mirzayanov.[8] Dispersed in an ultra-fine powder instead of a gas or a vapor, they have unique qualities. A binary agent was then created that would mimic the same properties but would either be manufactured using materials legal under the CWT[10] or be undetectable by treaty regime inspections.[11] The most potent compounds from this family, novichok-5 and novichok-7, are supposedly around five to eight times more potent than VX.
One of the key manufacturing sites was a chemical research institute in what is now Uzbekistan,[11] and small, experimental batches of the weapons may have been tested on the nearby Ustyurt plateau.[11]
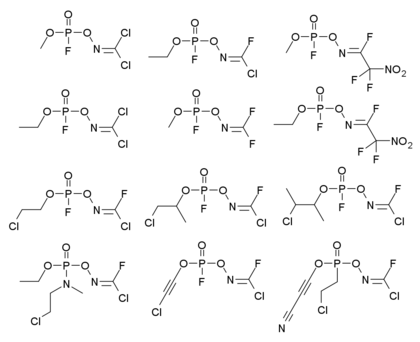
Two broad families of organophosphorus agents have been claimed to be Novichok agents. First are a group of organophosphorus compounds with an attached dihaloformaldoxime group, with the general formula shown below, where R = alkyl, alkoxy, alkylamino, or fluorine and X = halogen (F, Cl, Br) or pseudohalogen such as C≡N. These compounds are extensively documented in Soviet literature of the time, but it is unclear whether they are all members of the potent "Novichok" compounds.[12][13][14][15][16][17][18][19]

Some examples of the first group of compounds reported in the literature are shown below;
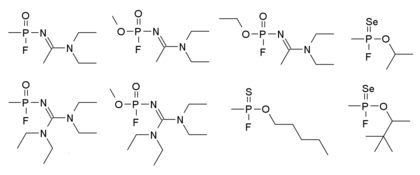
Mirzayanov gives somewhat different structures for Novichok agents in his autobiography, as shown below. He makes clear that a large number of compounds were made, and many of the less potent derivatives reported in the open literature as new organophosphate insecticides, so that the secret chemical weapons program could be disguised as legitimate pesticide research.[20]
Effects
As nerve agents, the Novichok agents belong to the class of organophosphate acetylcholinesterase inhibitors. These chemical compounds inhibit the enzyme acetylcholinesterase, preventing the normal breakdown of the neurotransmitter acetylcholine. Acetylcholine concentrations then increase at neuromuscular junctions to cause involuntary contraction of all muscles. This then leads to respiratory and cardiac arrest (as the victim's heart and diaphragm muscles no longer function normally) and finally death from heart failure or suffocation as copious fluid secretions fill the victim's lungs. [21]
The use of a fast-acting peripheral anticholinergic drug such as atropine can block the receptors where acetylcholine acts to prevent poisoning (as is the treatment for poisoning by other acetylcholinesterare inhibitors). Atropine, however, is difficult to administer safely, because its effective dose for nerve agent poisoning is close to the dose at which patients suffer severe side effects such as changes in heart rate and thickening of the bronchial secretions which fill the lungs of someone suffering nerve agent poisoning, so that suctioning of these secretion and other advanced life support techniques may be necessary in addition to administration of atropine to treat nerve agent poisoning. [21]
In the treatment of nerve agent poisoning, atropine is most often administered along with pralidoxime, which reactivates acetylcholinesterase which has been inactivated by phosphorylation by an organophosphorous nerve agent and relieves the respiratory muscle paralysis caused by some nerve agents. Pralidoxime is not effective in reactivating acetylcholine inhibited by some older nerve agents such as soman [21] or the Novichok nerve agents, described in the literature as being up to 8 times more toxic than nerve agent VX. [22]
See also
References
- ↑ Tucker, J. B.; War of Nerves; Anchor Books; New York; 2006; pp 232-233.
- 1 2 Vadim J. Birstein. The Perversion Of Knowledge: The True Story of Soviet Science. Westview Press (2004) ISBN 0-8133-4280-5
- 1 2 3 4 5 Yevgenia Albats and Catherine A. Fitzpatrick. The State Within a State: The KGB and Its Hold on Russia — Past, Present, and Future, 1994. ISBN 0-374-18104-7 (see pages 325-328)
- ↑ Tucker, J. B.; War of Nerves; Anchor Books; New York; 2006; pp 231.
- ↑ Tucker, J. B.; War of Nerves; Anchor Books; New York; 2006; pp 233.
- ↑ Tucker, J. B.; War of Nerves; Anchor Books; New York; 2006; pp 253.
- ↑ Fedorov, Lev and Vil Mirzayanov, "Poisoned Politics," Moskovskiye Novosti weekly No. 39, 1992. Much of this information was published earlier in the newspaper "Top Secret" run by Artyom Borovik in September 1991. However the KGB did not arrest Mirzayanov earlier due to political turmoil in Russia at this time, according to a book by Yevgenia Albats.
- 1 2 Vil Mirzayanov "Dismantling the Soviet/Russian Chemical Weapons Complex: An Insider's View" Chemical Weapons Disarmament in Russia: Problems and Prospects (Washington, D.C.: Henry L. Stimson Center, 1995), page 21.
- ↑ "the talk [by Mirzayanov] about binary weapons was no more than a verbal construct, an argument ex adverso, and only the MCC [Russian Military Chemical Complex] could corroborate or refute this natural assumption. By entangling V. S. Mirzayanov in investigation, the MCC confirmed the stated hypothesis, advancing it to the ranks of proven facts."
- 1 2 David Hoffman (August 16, 1998). "Wastes of War: Soviets Reportedly Built Weapon Despite Pact". The Washington Post. Retrieved 2007-07-20.
- 1 2 3 Louise Hidalgo (August 9, 1999). "US dismantles chemical weapons". BBC News Online. Retrieved 2007-07-20.
- ↑ Kruglyak Yu L, Malekin SI, Martynov IV. Phosphorylated oximes. XII. Reactions of 2-halophospholanes with dichlorofluoronitrosomethane. Zhurnal Obshchei Khimii. 1972; 42(4):811-14.
- ↑ Raevskii OA, Chapysheva NV, Ivanov AN, Sokolov VB, Martynov IV. Effect of Alkyl Substituents in Phosphorylated Oximes. Zhurnal Obshchei Khimii. 1987; 57(12):2720-2723
- ↑ Raevskii OA, Grigor'ev V Yu, Solov'ev VP, Ivanov AN, Sokolov VB, Martynov IV. Electron-Donor Functions of Ethyl Methylchloroformimino Methylphosphonate. Zhurnal Obshchei Khimii. 1987; 57(9):2073-2078
- ↑ Makhaeva GF, Filonenko IV, Yankovskaya VL, Fomicheva SB, Malygin VV. Comparative studies of O,O-dialkyl-O-chloromethylchloroformimino phosphates: interaction with neuropathy target esterase and acetylcholinesterase. Neurotoxicology. 1998 Aug-Oct;19(4-5):623-8. PMID 9745921
- ↑ Raevskiĭ OA, Chistiakov VV, Agabekian RS, Sapegin AM, Zefirov NS. Formation of models of the interaction between organophosphate compound structure and their ability to inhibit cholinesterase. Bioorganicheskaia Khimiia. 1990 Nov;16(11):1509-22. PMID 2096825
- ↑ Ivanov IuIa, Sokolov VB, Epishina TA, Martynov IV. O-substituted alkylchloroformoximes as substrates and inhibitors of cholinesterases. Doklady Akademii Nauk SSSR. 1990;310(5):1253-5. PMID 2354654
- ↑ Malygin VV, Sokolov VB, Richardson RJ, Makhaeva GF. Quantitative structure-activity relationships predict the delayed neurotoxicity potential of a series of O-alkyl-O-methylchloroformimino phenylphosphonates. Journal of Toxicology and Environmental Health Part A. 2003 Apr 11;66(7):611-25. PMID 12746136
- ↑ Steven L. Hoenig. Compendium of Chemical Warfare Agents. Springer New York, 2007. ISBN 978-0-387-34626-7
- ↑ Vil S Mirzayanov. State Secrets. An Insider's Chronicle of the Russian Chemical Weapons Program. (2009) pp142-145, 179-180. ISBN 978-1-4327-2566-2
- 1 2 3 Meridian Medical Technologies , Inc. (30 September 2009). "LABEL: DUODOTE- atropine and pralidoxime chloride". National Institutes of Health, Bethesda, MD, USA. Retrieved 9 April 2016.
- ↑ Gupta, Ramesh C (ed) (2015). Handbook of Toxicology of Chemical Warfare Agents. Cambridge, MA, USA: Academic Press. pp. 339–340. ISBN 9780128004944.
External links
- Chemical Weapons in Russia: History, Ecology, Politics by Lev Fedorov, Moscow, Center of Ecological Policy of Russia, 27 July 1994
- Vil Mirzayanov "Dismantling the Soviet/Russian Chemical Weapons Complex: An Insider's View" Chemical Weapons Disarmament in Russia: Problems and Prospects (Washington, D.C.: Henry L. Stimson Center, 1995).
- Russian chemical weapons and by Federation of American Scientists
- Vil Mirzayanov, a scientist honored by the AAAS Science and Human Rights Program in 1995
- The Chemical Weapons Coverup, by J. Michael Waller, The Wall Street Journal, February 13, 1997
- Ken Alibek and Steven Handelman (1999), Biohazard: The Chilling True Story of the Largest Covert Biological Weapons Program in the World – Told from Inside by the Man Who Ran It, Random House, ISBN 0-385-33496-6.