Catecholamine

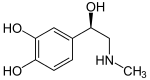
A catecholamine (/kætəˈkoʊləmin/) (CA) is a monoamine, an organic compound that has a catechol (benzene with two hydroxyl side groups at carbons 1 and 2) and a side-chain amine.[1]
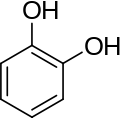
Catechol can be either a free molecule or a substituent of a larger molecule, where it represents a 1,2-dihydroxybenzene group.
Catecholamines are derived from the amino acid tyrosine, which is derived from dietary sources as well as synthesis of phenylalanine.[2] Catecholamines are water-soluble and are 50%-bound to plasma proteins in circulation.
Included among catecholamines are epinephrine (adrenaline), norepinephrine (noradrenaline), and dopamine. Release of the hormones epinephrine and norepinephrine from the adrenal medulla of the adrenal glands is part of the fight-or-flight response.[3]
Tyrosine is created from phenylalanine by hydroxylation by the enzyme phenylalanine hydroxylase. Tyrosine is also ingested directly from dietary protein. Catecholamine-secreting cells use several reactions to convert tyrosine serially to L-DOPA and then to dopamine. Depending on the cell type, dopamine may be further converted to norepinephrine or even further converted to epinephrine.[4]
Various stimulant drugs are catecholamine analogues.
Structure
Catecholamines have the distinct structure of a benzene ring with two hydroxyl groups, an intermediate ethyl chain, and a terminal amine group. Phenylethanolamines such as norepinephrine have a hydroxyl group on the ethyl chain.
Production and degradation
L-Phenylalanine is converted into L-tyrosine by an aromatic amino acid hydroxylase (AAAH) enzyme (phenylalanine 4-hydroxylase), with molecular oxygen (O2) and tetrahydrobiopterin as cofactors. L-Tyrosine is converted into L-DOPA by another AAAH enzyme (tyrosine 3-hydroxylase) with tetrahydrobiopterin, O2, and ferrous iron (Fe2+) as cofactors. L-DOPA is converted into dopamine by the enzyme aromatic L-amino acid decarboxylase (AADC), with pyridoxal phosphate as the cofactor. Dopamine itself is also used as precursor in the synthesis of the neurotransmitters norepinephrine and epinephrine. Dopamine is converted into norepinephrine by the enzyme dopamine β-hydroxylase (DBH), with O2 and L-ascorbic acid as cofactors. Norepinephrine is converted into epinephrine by the enzyme phenylethanolamine N-methyltransferase (PNMT) with S-adenosyl-L-methionine as the cofactor.
Location
Catecholamines are produced mainly by the chromaffin cells of the adrenal medulla and the postganglionic fibers of the sympathetic nervous system. Dopamine, which acts as a neurotransmitter in the central nervous system, is largely produced in neuronal cell bodies in two areas of the brainstem: the ventral tegmental area and the substantia nigra, the latter of which contains neuromelanin-pigmented neurons. The similarly neuromelanin-pigmented cell bodies of the locus ceruleus produce norepinephrine. Epinephrine is produced in small groups of neurons in the human brain which express its synthesizing enzyme, phenylethanolamine N-methyltransferase;[8] these neurons project from a nucleus that is adjacent (ventrolateral) to the area postrema and from a nucleus in the dorsal region of the solitary tract.[8]
Biosynthesis
Dopamine is the first catecholamine synthesized from DOPA. In turn, norepinephrine and epinephrine are derived from further metabolic modification of dopamine. The enzyme dopamine hydroxylase requires copper as a cofactor (not shown in the diagram) and DOPA decarboxylase requires PLP (not shown in the diagram). The rate limiting step in catecholamine biosynthesis through the predominant metabolic pathway is the hydroxylation of L-tyrosine to L-DOPA.
Catecholamine synthesis is inhibited by alpha-methyl-p-tyrosine (AMPT), which inhibits tyrosine hydroxylase.
Degradation
Catecholamines have a half-life of a few minutes when circulating in the blood. They can be degraded either by methylation by catechol-O-methyltransferases (COMT) or by deamination by monoamine oxidases (MAO).
MAOIs bind to MAO, thereby preventing it from breaking down catecholamines and other monoamines.
Function
Modality
Two catecholamines, norepinephrine and dopamine, act as neuromodulators in the central nervous system and as hormones in the blood circulation. The catecholamine norepinephrine is a neuromodulator of the peripheral sympathetic nervous system but is also present in the blood (mostly through "spillover" from the synapses of the sympathetic system).
High catecholamine levels in blood are associated with stress, which can be induced from psychological reactions or environmental stressors such as elevated sound levels, intense light, or low blood sugar levels.
Extremely high levels of catecholamines (also known as catecholamine toxicity) can occur in central nervous system trauma due to stimulation and/or damage of nuclei in the brainstem, in particular those nuclei affecting the sympathetic nervous system. In emergency medicine, this occurrence is widely known as catecholamine dump.
Extremely high levels of catecholamine can also be caused by neuroendocrine tumors in the adrenal medulla, a treatable condition known as pheochromocytoma.
High levels of catecholamines can also be caused by monoamine oxidase A (MAO-A) deficiency. As MAO-A is one of the enzymes responsible for degradation of these neurotransmitters, its deficiency increases the bioavailability of these neurotransmitters considerably. It occurs in the absence of pheochromocytoma, neuroendocrine tumors, and carcinoid syndrome, but it looks similar to carcinoid syndrome such as facial flushing and aggression.[9][10]
Effects
Catecholamines cause general physiological changes that prepare the body for physical activity (fight-or-flight response). Some typical effects are increases in heart rate, blood pressure, blood glucose levels, and a general reaction of the sympathetic nervous system. Some drugs, like tolcapone (a central COMT-inhibitor), raise the levels of all the catecholamines.
Function in plants
"They have been found in 44 plant families, but no essential metabolic function has been established for them. They are precursors of benzo[c]phenanthridine alkaloids, which are the active principal ingredients of many medicinal plant extracts. CAs have been implicated to have a possible protective role against insect predators, injuries, and nitrogen detoxification. They have been shown to promote plant tissue growth, somatic embryogenesis from in vitro cultures, and flowering. CAs inhibit indole-3-acetic acid oxidation and enhance ethylene biosynthesis. They have also been shown to enhance synergistically various effects of gibberellins."[11]
See also
References
- ↑ Fitzgerald, P. A. (2011). "Chapter 11. Adrenal Medulla and Paraganglia". In Gardner, D. G.; Shoback, D. Greenspan’s Basic & Clinical Endocrinology (9th ed.). New York: McGraw-Hill. Retrieved October 26, 2011.
- ↑ Purves, D.; Augustine, G. J.; Fitzpatrick, D.; Hall, W. C.; LaMantia, A. S.; McNamara, J. O.; White, L. E., eds. (2008). Neuroscience (4th ed.). Sinauer Associates. pp. 137–8. ISBN 978-0-87893-697-7.
- ↑ "Catecholamines". Health Library. San Diego: University of California.
- ↑ Joh, T. H.; Hwang, O. (1987). "Dopamine Beta-Hydroxylase: Biochemistry and Molecular Biology". Annals of the New York Academy of Sciences. 493: 342–350. doi:10.1111/j.1749-6632.1987.tb27217.x. PMID 3473965.
- ↑ Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacol. Ther. 125 (3): 363–375. doi:10.1016/j.pharmthera.2009.11.005. PMID 19948186.
- ↑ Lindemann L, Hoener MC (May 2005). "A renaissance in trace amines inspired by a novel GPCR family". Trends Pharmacol. Sci. 26 (5): 274–281. doi:10.1016/j.tips.2005.03.007. PMID 15860375.
- ↑ Wang X, Li J, Dong G, Yue J (February 2014). "The endogenous substrates of brain CYP2D". Eur. J. Pharmacol. 724: 211–218. doi:10.1016/j.ejphar.2013.12.025. PMID 24374199.
The highest level of brain CYP2D activity was found in the substantia nigra (Bromek et al., 2010). The in vitro and in vivo studies have shown the contribution of the alternative CYP2D-mediated dopamine synthesis to the concentration of this neurotransmitter although the classic biosynthetic route to dopamine from tyrosine is active. CYP2D6 protein level is approximately 40% lower in the frontal cortex, cerebellum, and hippocampus in PD patients, even when controlling for CYP2D6 genotype (Mann et al., 2012). ... Tyramine levels are especially high in the basal ganglia and limbic system, which are thought to be related to individual behavior and emotion (Yu et al., 2003c). Studies have demonstrated that dopamine is formed from p-tyramine as well as m-tyramine via tyramine 3-hydroxylation or 4-hydroxylation by rat CYP2D2, 2D4, and 2D18 as well as human CYP2D6. ... Both rat CYP2D and human CYP2D6 have a higher affinity for m-tyramine compared with p-tyramine for the generation of dopamine. Rat CYP2D isoforms (2D2/2D4/2D18) are less efficient than human CYP2D6 for the generation of dopamine from p-tyramine. The Km values of the CYP2D isoforms are as follows: CYP2D6 (87–121 μm) ≈ CYP2D2 ≈ CYP2D18 > CYP2D4 (256 μm) for m-tyramine and CYP2D4 (433 μm) > CYP2D2 ≈ CYP2D6 > CYP2D18 (688 μm) for p-tyramine (Bromek et al., 2010; Thompson et al., 2000).
- 1 2 Kitahama K, Pearson J, Denoroy L, Kopp N, Ulrich J, Maeda T, Jouvet M (1985). "Adrenergic neurons in human brain demonstrated by immunohistochemistry with antibodies to phenylethanolamine-N-methyltransferase (PNMT): discovery of a new group in the nucleus tractus solitarius". Neurosci. Lett. 53 (3): 303–308. PMID 3885079.
- ↑ Manor, I.; Tyano, S.; Mel, E.; Eisenberg, J.; Bachner-Melman, R.; Kotler, M.; Ebstein, R. P. (2002). "Family-Based and Association Studies of Monoamine Oxidase A and Attention Deficit Hyperactivity Disorder (ADHD): Preferential Transmission of the Long Promoter-Region Repeat and its Association with Impaired Performance on a Continuous Performance Test (TOVA)". Molecular Psychiatry. 7 (6): 626–632. doi:10.1038/sj.mp.4001037. PMID 12140786.
- ↑ Brunner, H. G. (1996). "MAOA Deficiency and Abnormal Behaviour: Perspectives on an Association". Ciba Foundation Symposium. 194: 155–164; discussion 164–167. PMID 8862875.
- ↑ Kuklin, A. I.; Conger, B. V. (1995). "Catecholamines in Plants". Journal of Plant Growth Regulation. 14 (2): 91–97. doi:10.1007/BF00203119.
External links
- Catecholamines at the US National Library of Medicine Medical Subject Headings (MeSH)