Macular edema
| Macular edema | |
|---|---|
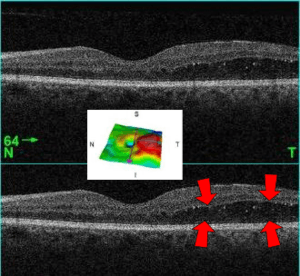 | |
| A 61-year-old man with medical history of type 2 diabetes that presents a macular edema, evidenced by an OCT (the edema marked with arrows). The central image is a 3D reconstruction of the retinal thickness (the edema is coloured in red). | |
| Classification and external resources | |
| Specialty | ophthalmology |
| ICD-10 | H35.8 |
| ICD-9-CM | 362.01, 362.53 |
Macular edema occurs when fluid and protein deposits collect on or under the macula of the eye (a yellow central area of the retina) and causes it to thicken and swell (edema). The swelling may distort a person's central vision, because the macula holds tightly packed cones that provide sharp, clear, central vision to enable a person to see detail, form, and color that is directly in the centre of the field of view.
Causes of macular edema
The causes of macular edema are numerous and different causes may be inter-related.
- It is commonly associated with diabetes. Chronic or uncontrolled diabetes type 2 can affect peripheral blood vessels including those of the retina which may leak fluid, blood and occasionally fats into the retina causing it to swell.[1]
- Age-related macular degeneration may cause macular edema. As individuals age there may be a natural deterioration in the macula which can lead to the depositing of drusen under the retina sometimes with the formation of abnormal blood vessels.[2]
- Replacement of the lens as treatment for cataract can cause pseudophakic macular edema. (‘pseudophakia’ means ‘replacement lens’) also known as Irvine-Gass syndrome The surgery involved sometimes irritates the retina (and other parts of the eye) causing the capillaries in the retina to dilate and leak fluid into the retina. Less common today with modern lens replacement techniques.[3]
- Chronic uveitis and intermediate uveitis can be a cause.[4]
- Blockage of a vein in the retina can cause engorgement of the other retinal veins causing them to leak fluid under or into the retina. The blockage may be caused, among other things, by atherosclerosis, high blood pressure and glaucoma.[5]
- A number of drugs can cause changes in the retina that can lead to macular edema. The effect of each drug is variable and some drugs have a lesser role in causation. The principal medication known to affect the retina are:- latanoprost, epinephrine, rosiglitazone, timolol and thiazolidinediones among others.[6][7]
- A few congenital diseases are known to be associated with macular edema for example retinitis pigmentosa and retinoschisis.[1]
Classification
Cystoid macular edema (CME) involves fluid accumulation in the outer plexiform layer secondary to abnormal perifoveal retinal capillary permeability. The edema is termed "cystoid" as it appears cystic; however, lacking an epithelial coating, it is not truly cystic. The etiology for CME can be remembered with the mnemonic "DEPRIVEN" (Diabetes, Epinepherine, Pars planitis, Retinitis pigmentosa, Irvine-Gass Syndrome, Venous occlusion, E2-Prostaglandin analogues, Nicotinic acid/Niacin).
Diabetic macular edema (DME) is similarly caused by leaking macular capillaries. DME is the most common cause of visual loss in both proliferative, and non-proliferative diabetic retinopathy.
Treatment
Macular edema sometimes occurs for a few days or weeks after cataract surgery, but most such cases can be successfully treated with NSAID or cortisone eye drops. Prophylactic use of Nonsteroidal anti-inflammatory drugs has been reported to reduce the risk of macular edema to some extent.[8]
In 2010 the US FDA approved the use of Lucentis intravitreal injections for macular edema.[9]
Iluvien, a sustained release intravitreal implant developed by Alimera Sciences, has been approved in Austria, Portugal and the U.K. for the treatment of vision impairment associated with chronic diabetic macular edema (DME) considered insufficiently responsive to available therapies. Additional EU country approvals are anticipated.[10]
In 2013 Lucentis by intravitreal injection was approved by the National Institute for Health and Care Excellence in the UK for the treatment of macular edema caused by diabetes[11] and/or retinal vein occlusion.[12]
On July 29, 2014, Eylea (aflibercept), an intravitreal injection produced by Regeneron Pharmaceuticals Inc., was approved to treat DME in the United States.[13]
Clinical trials
In 2005, steroids were investigated for the treatment of macular edema due to retinal blood vessel blockage such as CRVO and BRVO.[14]
Additional research
A 2014 Cochrane Systematic Review studied the effectiveness of two anti-VEGF treatments, ranibizumab and pegaptanib, on patients suffering from macular edema caused by CRVO.[15] Participants on both treatment groups showed a reduction in macular edema symptoms over six months.[15]
Another Cochrane Review examined the effectiveness and safety of two intravitreal steroid treatments, triamcinolone acetonide and dexamethasone, for patients with from CRVO-ME.[16] The results from one trial showed that patients treated with triamcinolone acetonide were significantly more likely to show improvements in visual acuity than those in the control group, though outcome data was missing for a large proportion of the control group. The second trial showed that patients treated with dexamethasone implants did not show improvements in visual acuity, compared to patients in the control group.
Evidence also suggests that intravitreal injections and implantation of steroids inside the eye can result in improved visual outcomes for patients with chronic or refractory diabetic macular edema.[17]
See also
References
- 1 2 "What Causes Macular Edema". American Academy of Ophthalmology. Retrieved 30 January 2016.
- ↑ "What is Age-Related Macular Degeneration?". American Academy of Ophthalmology. Retrieved 30 January 2016.
- ↑ Laly, David R (5 March 2014). "Pseudophakic Cystoid Macular Edema". Review of Ophthalmology. Retrieved 30 January 2016.
- ↑ "Complications of Uveitis". Her Majesty's Government, UK. 27 January 2015. Retrieved 30 January 2016.
- ↑ Lusby, Franklin W (8 May 2014). "Retinal Vein Occlusion". Medline Plus. US Library of Medicine. Retrieved 30 January 2016.
- ↑ Abaasi, Omar (11 June 2009). "Common Medications That May Be Toxic To The Retina". Review of Ophthalmology. Retrieved 30 January 2016.
- ↑ "Medication Cautions in Macular Degeneration". American Macular Degeneration Foundation. Retrieved 30 January 2016.
- ↑ Lim, Blanche X.; Lim, Chris Hl; Lim, Dawn K.; Evans, Jennifer R.; Bunce, Catey; Wormald, Richard. "Prophylactic non-steroidal anti-inflammatory drugs for the prevention of macular oedema after cataract surgery". The Cochrane Database of Systematic Reviews 2016. 11: CD006683. doi:10.1002/14651858.CD006683.pub3. ISSN 1469-493X. PMID 27801522.
- ↑ "GEN | News Highlights: FDA Green-Lights Genentech's Lucentis for Macular Edema following Retinal Vein Occlusion". Genengnews.com. Retrieved 2012-12-15.
- ↑ "Iluvien gains marketing authorization in Portugal for chronic DME". OSN SuperSite, June 7, 2012. Retrieved June 20, 2012
- ↑ "Ranibizumab for treating diabetic macular oedema". NICE Guidance. NICE. February 2013.
- ↑ "Ranibizumab for treating visual impairment caused by macular oedema secondary to retinal vein occlusion". NICE Guidance. NICE. May 2013. Retrieved 30 January 2016.
- ↑ http://files.shareholder.com/downloads/REGN/3361382769x0x772028/698a12f7-a8dc-4f89-b9e2-f8134ae6f3de/REGN_News_2014_7_29_General_Releases.pdf
- ↑ "Steroid Injections vs. Standard Treatment for Macular Edema Due to Retinal Blood Vessel Blockage - Full Text View". ClinicalTrials.gov. Retrieved 2012-12-15.
- 1 2 Braithwaite T, Nanji AA, Greenberg PB (2010). "Anti-vascular endothelial growth factor for macular edema secondary to central retinal vein occlusion". Cochrane Database Syst Rev. 10: CD007325. doi:10.1002/14651858.CD007325.pub2. PMID 20927757.
- ↑ Gewaily D, Muthuswamy K, Greenberg PB (2015). "Intravitreal steroids versus observation for macular edema secondary to central retinal vein occlusion". Cochrane Database Syst Rev. 9: CD007324. doi:10.1002/14651858.CD007324.pub3. PMID 26352007.
- ↑ Grover DA, Li T, Chong CC (2008). "Intravitreal steroids for macular edema in diabetes". Cochrane Database Syst Rev. 1: CD005656. doi:10.1002/14651858.CD005656.pub2. PMC 3804331
 . PMID 18254088.
. PMID 18254088.