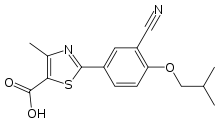
Febuxostat
 | |
| Clinical data | |
|---|---|
| Trade names | Uloric, Adenuric, Atenurix, Feburic, Goturic, Goutex. Generic in several countries.[1] |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a609020 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | M04AA03 (WHO) |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~49% absorbed |
| Protein binding | ~99% to albumin |
| Metabolism | via CYP1A2, 2C8, 2C9, UGT1A1, 1A3, 1A9, 2B7 |
| Biological half-life | ~5-8 hours |
| Excretion | Urine (~49% mostly as metabolites, 3% as unchanged drug); feces (~45% mostly as metabolites, 12% as unchanged drug) |
| Identifiers | |
| |
| CAS Number |
144060-53-7 |
| PubChem (CID) | 134018 |
| IUPHAR/BPS | 6817 |
| DrugBank |
DB04854 |
| ChemSpider |
118173 |
| UNII |
101V0R1N2E |
| KEGG |
D01206 |
| ChEMBL |
CHEMBL1164729 |
| ECHA InfoCard | 100.207.329 |
| Chemical and physical data | |
| Formula | C16H16N2O3S |
| Molar mass | 316.374 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Febuxostat (INN; trade names Adenuric in Europe and New Zealand, Uloric in the US, Goturic in Latin America, Feburic in Japan) is a drug that inhibits xanthine oxidase, thus reducing production of uric acid in the body. It is used in the treatment of chronic gout and hyperuricemia.
Febuxostat was discovered by scientists at the Japanese pharmaceutical company Teijin in 1998. Teijin partnered the drug with TAP Pharmaceuticals in the US and Ipsen in Europe. Ipsen obtained marketing approval for febuxostat from the European Medicines Agency in April 2008, Takeda obtained FDA approval in February 2009, and Teijin obtained approval from the Japanese "Pharmaceuticals and Medical Devices Agency" in 2011.
Medical uses
Febuxostat is used to treat chronic gout and hyperuricemia.[2] National Institute for Health and Clinical Excellence concluded that febuxostat is more effective than standard doses of allopurinol, but not more effective than higher doses of allopurinol.[2]

Febuxostat is in the US pregnancy category C; there are no adequate and well-controlled studies in pregnant women.[3]
Side effects
The adverse effects associated with febuxostat therapy include nausea, diarrhea, arthralgia, headache, increased hepatic serum enzyme levels and rash.[3][4]
Drug interactions
Febuxostat is contraindicated with concomitant use of theophylline and chemotherapeutic agents, namely azathioprine and 6-mercaptopurine, because it could increase blood plasma concentrations of these drugs, and therefore their toxicity.[3][5]
Mechanism of action
Febuxostat is a non-purine-selective inhibitor of xanthine oxidase.[3] It works by non-competitively blocking the molybdenum pterin center which is the active site on xanthine oxidase. Xanthine oxidase is needed to successively oxidize both hypoxanthine and xanthine to uric acid. Hence, febuxostat inhibits xanthine oxidase, therefore reducing production of uric acid. Febuxostat inhibits both oxidized as well as reduced form of xanthine oxidase because of which febuxostat cannot be easily displaced from the molybdenum pterin site.[4]
History
Febuxostat was discovered by scientists at the Japanese pharmaceutical company Teijin in 1998.[6] Teijin partnered the drug with TAP Pharmaceuticals in the US and Ipsen in Europe.[7][8][9]
Ipsen obtained marketing approval for febuxostat from the European Medicines Agency in April 2008,[10] Takeda obtained FDA approval in February 2009,[11][12] and Teijin obtained approval from the Japanese authorities in 2011.[13] Ipsen exclusively licensed its European rights to Menarini in 2009.[14] Teijin partnered with Astellas for distribution in China and southeast Asia.[15][16]
Society and culture
Cost
In the UK, NICE has found that febuxostat has a higher cost/benefit ratio than allopurinol and on that basis recommended febuxostat as a second-line drug for people who cannot use allopurinol.[2]
Trade names
Febuxostat is marketed as Adenuric in Europe and New Zealand, Uloric in the US, Goturic and Goutex in Latin America, Feburic in Japan, and is generic in several countries and is available by many names in those countries.[1]
References
- 1 2 Drugs.com Drugs.com international names for febuxostat Page accessed June 25, 2015
- 1 2 3 Febuxostat for the management of hyperuricaemia in people with gout (TA164) Chapter 4. Consideration of the evidence
- 1 2 3 4 Uloric label Updated February, 2009.
- 1 2 Love BL, Barrons R, Veverka A, Snider KM (2010). "Urate-lowering therapy for gout: focus on febuxostat". Pharmacotherapy. 30 (6): 594–608. doi:10.1592/phco.30.6.594. PMID 20500048.
- ↑ Ashraf Mozayani; Lionel Raymon (2011). Handbook of Drug Interactions: A Clinical and Forensic Guide. Springer Science+Business Media.
- ↑ Teijin Febuxostat Story Page accessed June 25, 2015
- ↑ Tomlinson B. Febuxostat (Teijin/Ipsen/TAP). Curr Opin Investig Drugs. 2005 Nov;6(11):1168-78. PMID 16312139
- ↑ Bruce Japsen for the Chicago Tribune. August 17, 2006. FDA puts gout treatment on hold
- ↑ Note: TAP Pharmaceuticals was a joint venture between Abbott Laboratories and Takeda that was dissolved in 2008 per this press release: Takeda, Abbott Announce Plans to Conclude TAP Joint Venture
- ↑ "Adenuric (febuxostat) receives marketing authorisation in the European Union" (PDF). Retrieved 2008-05-28.
- ↑ "Uloric Approved for Gout". U.S. News and World Report. Retrieved 2009-02-16.
- ↑ Teijin and Takeda. February 14, 2009 Press release: ULORIC® (TMX-67, febuxostat) Receives FDA Approval for the Chronic Management of Hyperuricemia in Patients with Gout
- ↑ Teijin. January 21, 2011 Press release: TMX-67 (febuxostat) Approved in Japan
- ↑ Genetic Engineering News. October 2009. Menarini to Market Takeda/Ipsen Gout Therapy in 41 European Countries
- ↑ First Word Pharma. April 1st, 2010 Teijin Pharma and Astellas Pharma enter into agreement for marketing rights of TMX-67 in China and Hong Kong
- ↑ Research Views. Aug 11 2011 Teijin Pharma Enters Into Distribution Agreement With Astellas Pharma For Febuxostat